Advanced Three-Step Synthesis of Minodronic Acid for Commercial Pharmaceutical Production
Advanced Three-Step Synthesis of Minodronic Acid for Commercial Pharmaceutical Production
The pharmaceutical industry is constantly seeking more efficient and safer pathways for the production of critical therapeutic agents, particularly in the realm of osteoporosis treatment where bisphosphonates play a pivotal role. Patent CN102344463A introduces a groundbreaking methodology for the preparation of 1-hydroxy-2-(imidazo[1,2-a]pyridin-3-yl)ethylidene-1,1-bisphosphonic acid, commonly known as Minodronic Acid, which addresses significant limitations found in prior art. This novel approach streamlines the synthetic route from a cumbersome six-step process down to a highly efficient three-step sequence, fundamentally altering the economic and safety profile of manufacturing this potent third-generation nitrogen-containing heterocyclic bisphosphonate. By eliminating the reliance on highly toxic sodium cyanide and simplifying downstream processing, this technology offers a robust solution for reliable pharmaceutical intermediate supplier networks aiming to enhance their portfolio with safer, cost-effective APIs. The strategic implementation of this synthesis not only mitigates environmental hazards but also ensures a consistent supply of high-purity material essential for clinical efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Minodronic Acid has been plagued by significant operational challenges and safety concerns that hindered its widespread industrial adoption. Prior art, such as the methods disclosed in EP0354806 and various academic journals, typically relied on a six-step synthetic pathway that was not only labor-intensive but also fraught with peril due to the utilization of sodium cyanide. The use of such highly toxic substances necessitates rigorous safety protocols, specialized waste treatment facilities, and complex containment systems, all of which drive up the capital expenditure and operational costs for manufacturing plants. Furthermore, the multi-step nature of these conventional routes inherently leads to cumulative yield losses at each stage, resulting in lower overall productivity and higher material costs. The difficulty in purifying intermediates generated during these lengthy sequences often leads to impurity profiles that are challenging to control, potentially compromising the quality of the final active pharmaceutical ingredient and requiring extensive chromatographic purification.
The Novel Approach
In stark contrast to the legacy methodologies, the innovative process detailed in the patent data utilizes a direct and concise three-step strategy that bypasses the need for hazardous cyanide reagents entirely. This modern approach initiates with a nucleophilic addition cyclization between 2-aminopyridine and a specific crotonate derivative, establishing the core heterocyclic structure in a single, high-efficiency operation. By condensing the synthetic timeline, the process drastically reduces the number of isolation and purification events, thereby minimizing material loss and solvent consumption. The elimination of toxic reagents simplifies the regulatory compliance landscape and reduces the burden on environmental health and safety departments, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing. Additionally, the final phosphorylation step is optimized to proceed under moderate conditions using toluene as a solvent, which facilitates easier product recovery and ensures that the final compound meets the stringent purity requirements demanded by global regulatory bodies without excessive reprocessing.
Mechanistic Insights into the Three-Step Cyclization and Phosphorylation
The core of this technological advancement lies in the precise orchestration of nucleophilic attacks and subsequent functional group transformations that build the complex bisphosphonate architecture. The first stage involves the reaction of 2-aminopyridine with trans-4-methoxy-2-butenoyl aldehyde (Formula II) in acetonitrile, where the amino group acts as a nucleophile attacking the electron-deficient alkene system to form the imidazo[1,2-a]pyridine ring system (Formula III). This cyclization is critical as it establishes the nitrogenous aromatic heterocyclic scaffold that is essential for the biological activity of the final drug, and the use of acetonitrile promotes the necessary solubility and reaction kinetics for high conversion. Following the formation of the ester intermediate, a hydrolysis step under alkaline conditions using sodium hydroxide cleaves the ethyl ester group to yield the free acetic acid derivative (Formula IV), preparing the molecule for the final phosphonation. The ultimate step employs phosphorus trichloride and phosphorous acid in a toluene medium to introduce the geminal bisphosphonic acid moiety, a reaction that requires careful temperature control to manage the exothermic nature of phosphorus halide chemistry while ensuring complete substitution.
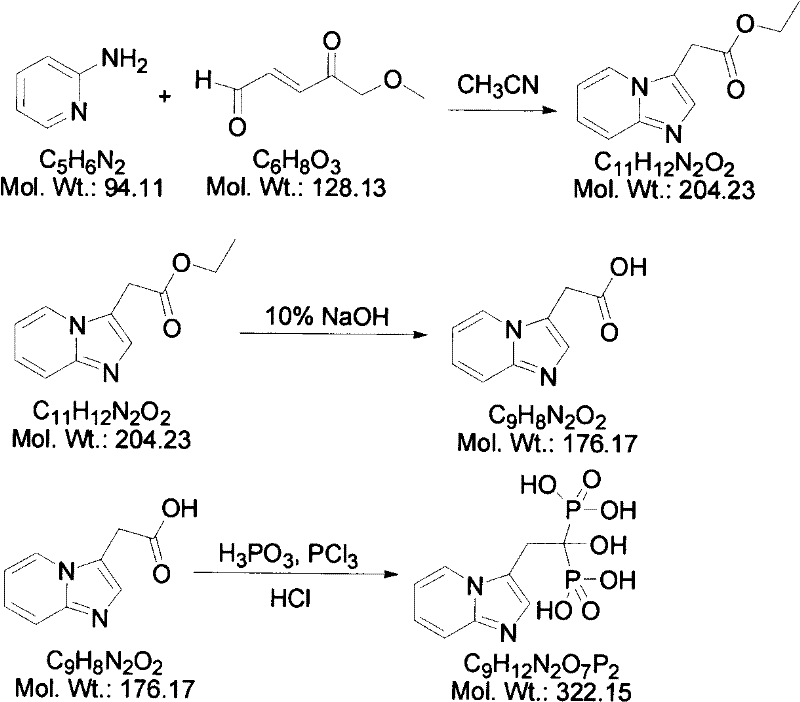
Controlling the impurity profile throughout this three-stage sequence is paramount for ensuring the safety and efficacy of the final osteoporosis medication. The patent specifies that the initial cyclization yields a yellow needle-like crystal which, upon hydrolysis, converts to a solid acid with a sharp melting point, indicating high structural integrity before the final phosphorylation. During the phosphorylation phase, the reaction mixture is subjected to acid hydrolysis using 6N hydrochloric acid, a step designed to quench any unreacted phosphorus species and hydrolyze intermediate phosphonate esters into the desired free acid form. The subsequent recrystallization from 1N hydrochloric acid serves as a powerful purification tool, effectively removing inorganic salts and organic byproducts that may have formed during the vigorous reaction conditions. Analytical data confirms that this rigorous purification protocol results in a final product with an HPLC purity of 99.7%, demonstrating the method's superior capability to produce high-purity pharmaceutical intermediates compared to older, less controlled synthetic routes.
How to Synthesize Minodronic Acid Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to maximize yield and safety while maintaining the integrity of the sensitive bisphosphonate functionality. The process begins with the reflux of reactants in acetonitrile followed by a specific workup involving petroleum ether to isolate the intermediate ester, which is then directly subjected to alkaline hydrolysis without the need for extensive drying or purification. The final phosphorylation step demands precise temperature ramping from 50°C to 110°C over several hours to ensure complete reaction of the phosphorus reagents, followed by an overnight hydrolysis period to fully convert the species to the target acid. For a comprehensive understanding of the specific molar ratios, solvent volumes, and detailed operational procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Perform nucleophilic addition of 2-aminopyridine and trans-4-methoxy-2-butenoyl aldehyde in acetonitrile to form the imidazo[1,2-a]pyridine ester intermediate.
- Hydrolyze the ester intermediate under alkaline conditions using sodium hydroxide to obtain the corresponding acetic acid derivative.
- Conduct phosphorylation using phosphorus trichloride and phosphorous acid in toluene, followed by acid hydrolysis and recrystallization to yield the final bisphosphonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this cyanide-free synthetic route offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By reducing the number of synthetic steps from six to three, the process inherently lowers the consumption of raw materials, solvents, and energy, leading to a significant reduction in the overall cost of goods sold. The elimination of sodium cyanide not only removes the high costs associated with handling and disposing of hazardous waste but also mitigates the risk of supply chain disruptions caused by strict regulatory controls on toxic precursors. This streamlined approach enhances supply chain reliability by simplifying the manufacturing workflow, reducing the potential for batch failures, and shortening the production cycle time, which allows for more responsive inventory management. Furthermore, the use of common industrial solvents like toluene and acetonitrile ensures that raw material sourcing remains stable and cost-effective, avoiding the volatility associated with specialty reagents.
- Cost Reduction in Manufacturing: The consolidation of the synthetic pathway into three robust steps eliminates the cumulative yield losses typical of longer sequences, thereby maximizing the output per unit of input material. Removing the requirement for expensive hazardous waste treatment associated with cyanide disposal further drives down operational expenditures, allowing for more competitive pricing strategies in the global market. The simplified purification process reduces the demand for costly chromatography resins and extensive solvent exchanges, contributing to substantial cost savings in the final processing stages. These efficiencies collectively enable a more economical production model that can withstand market fluctuations while maintaining healthy profit margins for producers.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-aminopyridine and common phosphorus reagents ensures a stable supply base that is less susceptible to geopolitical or logistical bottlenecks. By avoiding restricted substances, manufacturers can operate with greater flexibility across different jurisdictions, reducing the administrative burden and lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions means that production can be scaled up or down with minimal re-optimization, providing the agility needed to meet fluctuating market demands for osteoporosis treatments. This stability is crucial for long-term supply agreements where consistency and continuity of supply are paramount for downstream drug formulation.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing standard reactor equipment and moderate temperatures that do not require exotic high-pressure or cryogenic infrastructure. The absence of toxic cyanide significantly lowers the environmental footprint of the manufacturing process, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. Waste streams are easier to treat and neutralize, reducing the liability and cost associated with environmental compliance and permitting. This green chemistry approach not only safeguards the environment but also enhances the corporate reputation of manufacturers as responsible stewards of chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced Minodronic Acid synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing frameworks.
Q: How does this new method improve upon traditional Minodronic Acid synthesis?
A: Traditional methods often involve six complex steps and utilize highly toxic sodium cyanide, posing severe safety and environmental risks. This patented process reduces the synthesis to only three steps, completely eliminates the use of cyanide, and significantly simplifies purification, resulting in higher overall yields and reduced production costs.
Q: What is the expected purity of the final product using this route?
A: The process is designed to achieve high purity levels suitable for pharmaceutical applications. Experimental data from the patent indicates that the final product can reach an HPLC purity of 99.7% after recrystallization, meeting stringent quality specifications for active pharmaceutical ingredients.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the methodology is specifically optimized for industrial suitability. By using common solvents like acetonitrile and toluene, avoiding hazardous reagents, and reducing the number of unit operations, the process offers excellent scalability and operational safety for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Minodronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging the efficiencies of this three-step cyanide-free process, we can offer our clients a reliable Minodronic Acid supplier partnership that balances cost, quality, and speed.
We invite forward-thinking pharmaceutical companies to collaborate with us to explore the full potential of this optimized manufacturing route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your supply chain objectives and drive value for your osteoporosis drug portfolio.
