Scalable Asymmetric Total-Synthesis of Chiral Huperzine A for Commercial Pharmaceutical Production
Scalable Asymmetric Total-Synthesis of Chiral Huperzine A for Commercial Pharmaceutical Production
The global demand for effective treatments against neurodegenerative disorders, particularly Alzheimer's disease, has placed significant pressure on the supply chain of critical active pharmaceutical ingredients (APIs). Among these, Huperzine A, a potent and reversible acetylcholinesterase inhibitor extracted from Huperzia serrata, stands out as a molecule of immense therapeutic value. However, natural extraction is plagued by extremely low content (approximately 0.01%) and the slow growth cycle of the source plant, making synthetic routes essential for meeting market needs. Patent CN101333190A discloses a groundbreaking asymmetric total-synthesis method for chiral Huperzine A that addresses these supply bottlenecks. This innovative approach utilizes 1,4-dihydro-spiro[4,5]-8-decanone as a starting material and leverages chiral ammonia, specifically Cinchona alkaloids, to promote a tandem asymmetric Michael addition/aldol condensation reaction. By shifting away from traditional extraction and older synthetic methodologies, this technology offers a viable pathway for the reliable production of high-purity pharmaceutical intermediates required for next-generation neurological therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active Huperzine A has been fraught with significant technical and economic challenges that hindered its widespread commercial adoption. Early representative methods, such as those reported by Kozikowski, relied heavily on the use of chiral auxiliaries like (-)-8-phenyl menthol. While effective in inducing asymmetry, this approach required large usage quantities of the auxiliary, leading to inflated raw material costs and complex downstream processing. Furthermore, these traditional routes often involved tediously long reaction sequences with relatively low overall productive rates. The necessity for chromatographic separation of non-enantiomer mixtures and the use of sterically hindered esters that resisted hydrolysis added further layers of complexity. Additionally, other prior art methods utilized harsh Lewis acid catalysts such as titanium tetrachloride, which pose significant environmental and safety hazards in a manufacturing setting. These factors combined to create a high barrier to entry for cost reduction in pharmaceutical intermediates manufacturing, limiting the availability of affordable, high-quality Huperzine A for clinical applications.
The Novel Approach
The methodology outlined in the patent data presents a paradigm shift by introducing a more effective and practical stereoselective synthesis route. Instead of relying on stoichiometric chiral auxiliaries or toxic heavy metals, this novel approach employs chiral ammonia, such as Cinchona alkaloids, to catalyze the key bond-forming steps. The process begins with the conversion of 1,4-dihydro-spiro[4,5]-8-decanone into a beta-keto ester through a streamlined sequence involving hydroxymethylation and quinazoline formation. The core innovation lies in the use of (-)-cinchovatin to promote the tandem asymmetric Michael addition/aldol condensation of these beta-keto esters with methyl acrolein. This organocatalytic strategy not only simplifies the reaction setup but also demonstrates good enantioselectivity, with maximum enantiomeric excess reaching significant levels. By eliminating the need for expensive chiral pools like phenyl menthol and avoiding the waste streams associated with heavy metal catalysts, this new route fundamentally alters the economic landscape of Huperzine A production, offering a cleaner and more sustainable alternative for industrial synthesis.
Mechanistic Insights into Cinchona Alkaloid-Promoted Asymmetric Catalysis
The heart of this synthetic breakthrough is the sophisticated application of organocatalysis to construct the complex bridged ring system characteristic of Huperzine A. The mechanism involves a carefully orchestrated tandem reaction where the beta-keto ester acts as the acceptor in an asymmetric Michael addition, followed immediately by an intramolecular aldol condensation. The chiral environment provided by the Cinchona alkaloid, specifically (-)-cinchovatin in the preferred embodiment, effectively induces asymmetry during the nucleophilic attack on methyl acrolein. This step is critical as it establishes the multiple stereocenters required for the biological activity of the final molecule. The reaction proceeds under mild conditions, typically at low temperatures such as -10°C, which helps in controlling the stereochemical outcome and minimizing side reactions. The resulting mixture of diastereomers is then subjected to transformation using methanesulfonyl chloride (MsCl) and triethylamine, followed by elimination reactions to refine the ring structure. This mechanistic pathway avoids the racemization issues often seen in harsher chemical environments, ensuring that the chiral integrity of the molecule is maintained throughout the synthesis.
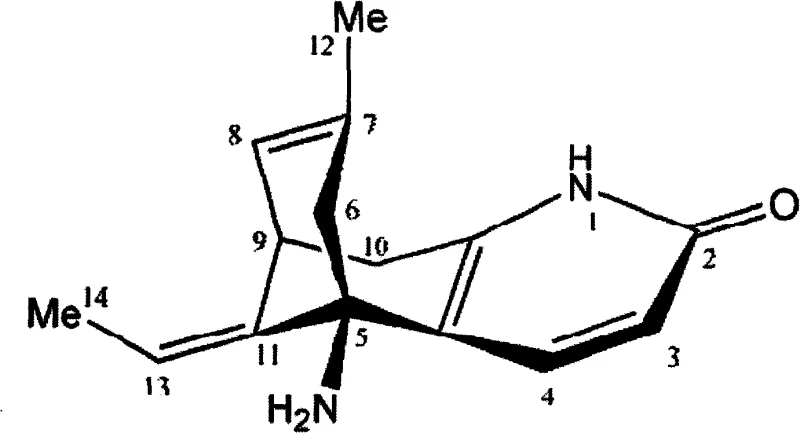
Controlling the impurity profile in the synthesis of complex alkaloids like Huperzine A is paramount for regulatory approval and patient safety. The described process incorporates specific purification strategies to manage diastereomeric impurities generated during the asymmetric induction phase. Although the initial Michael-Aldol reaction yields a mixture of diastereomers, the subsequent chemical transformations and recrystallization steps are designed to enrich the desired optical isomer. For instance, the intermediate tricyclic compounds can be recrystallized to achieve optical purity samples with greater than 99% enantiomeric excess (ee). This high level of purity is crucial because the biological activity of Huperzine A is highly stereospecific, with the (-)-enantiomer exhibiting significantly higher potency than its counterpart. The ability to remove unwanted isomers through physical means like recrystallization, rather than relying solely on difficult chromatographic separations at the final stage, represents a major advantage in process chemistry. It ensures that the final API meets stringent purity specifications while maintaining a robust and scalable manufacturing workflow.
How to Synthesize (-)-Huperzine A Efficiently
The synthesis of (-)-Huperzine A via this patented route involves a logical sequence of transformations starting from readily available spiro-ketones. The process is divided into the preparation of the key beta-keto ester intermediate followed by the asymmetric construction of the core ring system. Detailed operational parameters, including specific molar ratios, temperature controls, and solvent systems, are critical for reproducing the high yields and selectivity reported in the patent embodiments. The following guide outlines the standardized synthesis steps derived from the experimental data, providing a roadmap for process chemists to implement this technology in a pilot or production environment.
- Synthesize the beta-keto ester intermediate via hydroxymethylation, quinazoline formation, and ketal elimination starting from 1,4-dihydro-spiro[4,5]-8-decanone.
- Perform the key tandem asymmetric Michael addition/aldol condensation using (-)-cinchovatin and methyl acrolein to establish chirality.
- Execute functional group transformations including mesylation, elimination, Wittig reaction, and deprotection to yield final optically pure Huperzine A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive, specialized chiral auxiliaries with widely available Cinchona alkaloids, the dependency on niche suppliers is reduced, thereby enhancing supply chain reliability. Furthermore, the elimination of heavy metal catalysts like titanium tetrachloride removes the need for costly and time-consuming metal scavenging steps, which are often required to meet residual metal limits in pharmaceutical products. This streamlining of the process directly translates to reduced operational complexity and lower waste disposal costs. The robustness of the reaction conditions also suggests a higher tolerance for scale-up, reducing the risk of batch failures during commercial production. Consequently, this technology enables a more predictable and cost-effective supply of high-purity pharmaceutical intermediates, securing the continuity of supply for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this synthesis route is driven by the substitution of stoichiometric chiral reagents with catalytic amounts of organocatalysts. In previous methods, the requirement for large quantities of (-)-8-phenyl menthol represented a significant portion of the raw material cost. By shifting to a catalytic system using Cinchona alkaloids, the consumption of high-value chiral sources is minimized. Additionally, the avoidance of harsh reagents reduces the wear and tear on reactor equipment and lowers the costs associated with neutralizing hazardous waste streams. The simplified purification protocol, which relies on recrystallization rather than extensive chromatography for final purification, further decreases solvent consumption and processing time. These factors collectively contribute to a leaner manufacturing process with a significantly improved cost structure, allowing for competitive pricing in the global market for neurological therapeutics.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals and broadly available natural product derivatives as starting materials. The precursor 1,4-dihydro-spiro[4,5]-8-decanone and Cinchona alkaloids are sourced from established supply chains, reducing the risk of bottlenecks associated with custom-synthesized chiral auxiliaries. Moreover, the process conditions are less sensitive to moisture and air compared to organometallic routes, which simplifies logistics and storage requirements for reagents. This robustness ensures that production schedules can be maintained with greater consistency, even in varying operational environments. For supply chain heads, this means a more dependable source of critical intermediates, mitigating the risk of stockouts that could disrupt the production of finished dosage forms for patients suffering from Alzheimer's disease and myasthenia gravis.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this organocatalytic route aligns well with modern green chemistry principles. The absence of toxic heavy metals eliminates a major regulatory hurdle regarding heavy metal residues in the final API, simplifying the validation process for regulatory filings. The reaction steps are amenable to large-scale batch processing, as demonstrated by the successful execution of multi-gram syntheses in the patent examples. The waste profile is cleaner, consisting primarily of organic byproducts that are easier to treat than heavy metal sludge. This environmental compatibility not only reduces the carbon footprint of the manufacturing process but also ensures compliance with increasingly stringent environmental regulations globally. For manufacturers looking to expand capacity, this route offers a clear path to commercial scale-up of complex pharmaceutical intermediates without the need for specialized containment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the process capabilities and advantages over legacy methods. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: What are the limitations of previous Huperzine A synthesis methods compared to this patent?
A: Previous methods, such as the Kozikowski route, relied on expensive chiral auxiliaries like (-)-8-phenyl menthol in large quantities and involved tedious separation steps. Other routes utilized harsh Lewis acids like titanium tetrachloride. This new method utilizes readily available Cinchona alkaloids for organocatalysis, avoiding heavy metals and simplifying the purification process.
Q: How is high optical purity achieved in this synthesis route?
A: High optical purity is achieved through a tandem asymmetric Michael addition/aldol condensation promoted by chiral amines like (-)-cinchovatin. While the initial reaction yields a mixture of diastereomers, subsequent recrystallization steps allow for the isolation of intermediates with greater than 99% enantiomeric excess (ee).
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the route is designed for scalability. It avoids the use of stoichiometric amounts of expensive chiral auxiliaries and toxic heavy metal catalysts. The use of robust reagents and standard purification techniques like dynamic axial compression chromatography (DAC) facilitates scale-up from laboratory to industrial production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Huperzine A Supplier
The technological advancements detailed in patent CN101333190A represent a significant leap forward in the manufacture of chiral Huperzine A, yet translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of monitoring every step of this complex synthesis, ensuring that stringent purity specifications are met consistently. We understand the critical nature of neurological APIs and the absolute necessity for batch-to-batch consistency. Our team of expert chemists is ready to optimize the Cinchona alkaloid-catalyzed steps to maximize yield and enantiomeric excess, delivering a product that exceeds the expectations of global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to secure a stable supply of this vital intermediate. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this new synthetic route can optimize your overall COGS. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of chiral synthesis and bring your Alzheimer's therapeutic projects to market faster and more efficiently.
