Advancing Phenolic Glycoside Synthesis with Organic Thiourea Catalysis for Commercial Scale-up
The landscape of carbohydrate chemistry is continually evolving to meet the rigorous demands of modern pharmaceutical development, particularly in the synthesis of complex oligosaccharides and glycoconjugates. A significant breakthrough in this domain is documented in Chinese Patent CN113121619B, which introduces a novel method for the glycosidation of phenolic hydroxyl groups catalyzed by organic thiourea. This technology addresses a longstanding challenge in the field: the efficient and stereoselective formation of glycosidic bonds with phenolic acceptors, which are notoriously poor nucleophiles compared to aliphatic alcohols. The invention utilizes glycosyl N-phenyl-trifluoroacetyl imine esters as donors and employs a specific Kass reagent as the catalyst to drive the reaction under remarkably mild conditions. This approach not only enhances reaction efficiency but also provides superior control over stereochemistry, making it a highly valuable tool for the production of bioactive molecules used in new drug and vaccine development.
For procurement managers and supply chain leaders seeking a reliable pharmaceutical intermediate supplier, understanding the underlying robustness of this synthetic route is critical. The patent outlines a process that avoids the pitfalls of traditional methods, offering a pathway to cost reduction in pharmaceutical intermediate manufacturing through simplified operational parameters and higher material throughput. By leveraging this advanced catalytic system, manufacturers can achieve consistent quality in high-purity phenolic glycosides, ensuring a stable supply chain for downstream API production. The versatility of the method allows for the accommodation of various protecting groups and sugar moieties, facilitating the commercial scale-up of complex glycoconjugates without the need for exotic or prohibitively expensive reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of phenolic glycosidic bonds has been fraught with difficulties due to the intrinsic chemical properties of phenols. Unlike aliphatic alcohols, phenolic hydroxyl groups exhibit significantly lower nucleophilicity, making them reluctant partners in glycosylation reactions. Conventional strategies often rely on strong Lewis or Brønsted acids, such as triflic acid (TfOH), to activate the glycosyl donor. However, these harsh acidic conditions frequently lead to undesirable side reactions, including the hydrolysis of the glycosyl donor, which drastically reduces the overall yield of the desired product. Furthermore, achieving stereocontrol is exceptionally challenging when using strong acids, as the reaction often proceeds through an oxonium ion intermediate that can be attacked from either face, resulting in a mixture of alpha and beta anomers. This lack of selectivity necessitates cumbersome purification steps to isolate the desired stereoisomer, thereby increasing production costs and extending lead times. For substrates lacking neighboring group participation, such as 2-deoxy sugars, these conventional methods often fail entirely or produce unacceptable ratios of by-products, limiting their utility in synthesizing complex natural products.
The Novel Approach
In stark contrast to the aggressive nature of traditional acid catalysis, the method disclosed in patent CN113121619B utilizes a neutral organic thiourea catalyst, specifically the Kass reagent, to facilitate the glycosylation process. This innovative approach operates under mild conditions, typically at temperatures between 20°C and 50°C, which preserves the integrity of sensitive functional groups on both the donor and acceptor molecules. The use of glycosyl N-phenyl-trifluoroacetyl imine esters as donors, particularly those equipped with a Pico (2-methylenepyridine) directing group, synergizes effectively with the thiourea catalyst to enhance reactivity and stereoselectivity. Experimental data from the patent highlights the superiority of this method; for instance, the glycosylation of a glucose donor with 4-methoxyphenol yielded the desired product in 94% yield with an impressive beta/alpha ratio of 8:1. In comparison, the same reaction catalyzed by TfOH resulted in a mere 67% yield and a poor stereoselectivity ratio of 3:1. This dramatic improvement underscores the potential of organic thiourea catalysis to revolutionize the synthesis of phenolic glycosides, offering a cleaner, more efficient, and highly selective alternative for industrial applications.
Mechanistic Insights into Organic Thiourea-Catalyzed Glycosylation
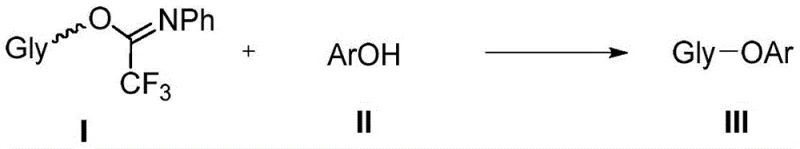
The success of this glycosylation method lies in the unique mechanistic role played by the Kass reagent, a bis-thiourea derivative. Unlike traditional promoters that generate highly reactive ionic species, the Kass reagent functions primarily through hydrogen bonding interactions. The thiourea moieties act as dual hydrogen bond donors, coordinating with the leaving group of the glycosyl donor (the trifluoroacetimidate) to facilitate its departure. This activation mode lowers the energy barrier for the formation of the glycosyl cation equivalent without generating a highly acidic environment. The presence of the Pico directing group on the C-6 position of the sugar donor further refines this process. As illustrated in the donor structures below, the picolinoyl group can participate in remote stereodirecting effects, guiding the incoming phenolic nucleophile to attack from the preferred face of the sugar ring. This synergy between the catalyst and the protecting group strategy allows for exceptional control over the anomeric configuration, even in the absence of traditional C-2 neighboring group participation.
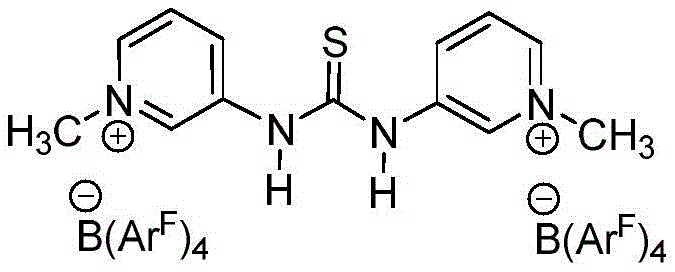
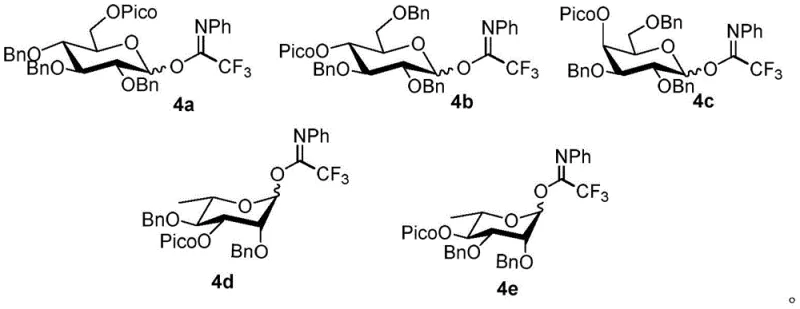
Furthermore, the structural diversity of the glycosyl donors compatible with this system is extensive, ranging from glucose and galactose to rhamnose and rare sugars. The patent details the preparation and utilization of various donors, such as compounds 4a through 4e, which feature different arrangements of benzyl and picolinoyl protecting groups. These structural variations are not merely academic; they are critical for tuning the electronic and steric environment of the anomeric center to match the specific requirements of the phenolic acceptor. For example, electron-deficient phenols, which are even less nucleophilic than their electron-rich counterparts, react efficiently with these activated donors to produce O-aryl glycosides with yields reaching up to 100% and stereoselectivity ratios exceeding 10:1. This mechanistic robustness ensures that the process is not limited to simple model systems but is applicable to the synthesis of complex, biologically relevant molecules, including flavonoid glycosides and glycosyl amino acids, which are essential building blocks in medicinal chemistry.
How to Synthesize Phenolic Glycosides Efficiently
The practical implementation of this glycosylation technology is straightforward and amenable to standard laboratory and pilot plant operations. The general procedure involves the preparation of the glycosyl N-phenyl-trifluoroacetyl imine ester donor, which is typically synthesized from the corresponding hemiacetal using N-phenyl-trifluoroacetimidoyl chloride and a base like cesium carbonate. Once the donor is prepared, it is combined with the phenolic acceptor in an anhydrous organic solvent, such as dichloromethane (DCM) or toluene. The mixture is subjected to azeotropic distillation with toluene to remove trace moisture, which is crucial for preventing donor hydrolysis. Following drying, the Kass catalyst is added in a catalytic amount (0.2 to 0.3 equivalents), and the reaction is allowed to proceed at room temperature or with mild heating. The progress is monitored via thin-layer chromatography (TLC), and upon completion, the product is isolated through standard workup procedures involving aqueous washes and column chromatography. For a detailed, step-by-step guide on executing this synthesis with specific reagents and conditions, please refer to the standardized protocol below.
- Prepare the glycosyl N-phenyl-trifluoroacetyl imine ester donor and the phenolic acceptor by azeotropic dehydration with toluene.
- Dissolve the reactants in dry dichloromethane (DCM) and add the Kass catalyst (0.2-0.3 equivalents) under an inert atmosphere.
- Stir the reaction mixture at room temperature or mild heating (20-50°C) for 16-36 hours, monitoring progress via TLC until completion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this organic thiourea-catalyzed glycosylation method offers substantial strategic advantages for pharmaceutical and fine chemical manufacturers. The shift from harsh acid catalysis to neutral organocatalysis fundamentally alters the cost structure and risk profile of the production process. By eliminating the need for strong acids and the associated corrosion-resistant equipment, facilities can reduce capital expenditure and maintenance costs. Moreover, the mild reaction conditions minimize the degradation of sensitive starting materials, leading to higher effective yields and reduced raw material consumption. This efficiency translates directly into improved margins and a more competitive pricing structure for the final intermediates. Additionally, the simplified purification requirements, driven by higher stereoselectivity and fewer by-products, streamline the manufacturing workflow, allowing for faster batch turnover and increased production capacity.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal promoters and strong acids significantly reduces the cost of goods sold (COGS). Traditional methods often require expensive scavengers to remove metal residues to meet regulatory standards for pharmaceutical ingredients. In contrast, the organic thiourea catalyst is metal-free, simplifying the purification process and reducing the burden on waste treatment systems. The high atom economy and reduced need for extensive chromatographic separation further contribute to substantial cost savings, making the production of complex glycosides more economically viable on a large scale.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more reliable supply of critical intermediates. Because the reaction tolerates a wide range of functional groups and proceeds under mild conditions, the risk of batch failure due to side reactions is markedly decreased. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers. Furthermore, the starting materials, including the glycosyl donors and phenolic acceptors, are generally commercially available or easily synthesized from bulk chemicals, reducing dependency on scarce or volatile supply chains.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often introduces safety and environmental challenges, particularly when handling hazardous reagents. The neutral nature of the Kass reagent and the moderate temperatures required for this reaction make it inherently safer and easier to scale. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations and corporate sustainability goals. By adopting this greener chemistry approach, companies can enhance their environmental, social, and governance (ESG) profiles while ensuring long-term operational compliance and community acceptance.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common questions regarding the scope and limitations of the organic thiourea-catalyzed glycosylation method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for optimizing reaction conditions and predicting outcomes for novel substrates. Whether you are exploring new drug candidates or optimizing existing synthetic routes, the following information provides a foundational understanding of what to expect from this advanced catalytic system.
Q: Why is organic thiourea catalysis superior to traditional acid catalysis for phenolic glycosylation?
A: Traditional strong acid catalysts like TfOH often require harsh conditions that lead to donor hydrolysis and poor stereoselectivity, especially with weakly nucleophilic phenols. The organic thiourea (Kass reagent) operates under neutral, mild conditions, significantly improving yields and beta-selectivity while minimizing side reactions.
Q: Can this method be applied to 2-deoxy sugars lacking neighboring group participation?
A: Yes, the patent demonstrates excellent results with 2-deoxy sugars and rare sugars where traditional neighboring group participation is impossible. The use of Pico directing groups on the donor combined with the Kass catalyst ensures high stereoselectivity even in these challenging substrates.
Q: What are the typical reaction conditions for this glycosylation process?
A: The reaction typically proceeds in organic solvents such as DCM, toluene, or acetonitrile at temperatures ranging from 20°C to 50°C. The molar ratio of donor to acceptor is generally 1.5:1 to 2:1, with a catalyst loading of 0.1 to 0.3 equivalents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenolic Glycosides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in patent CN113121619B for the next generation of glycosylated therapeutics. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory methods into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We are committed to delivering high-purity phenolic glycosides that meet the most stringent purity specifications required by global regulatory agencies. Our state-of-the-art rigorous QC labs employ advanced analytical techniques to verify the identity, potency, and impurity profile of every batch, guaranteeing the quality and consistency of our supply.
We invite you to collaborate with us to leverage this cutting-edge glycosylation technology for your specific application needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your project, demonstrating how our optimized processes can reduce your overall development costs. Please contact our technical procurement team today to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your custom synthesis requirements. Together, we can accelerate the development of life-saving medicines and bring innovative glycosylated products to market faster and more efficiently.
