Advanced Chemical Synthesis of Gastrodin and Analogous Phenolic Glycosides for Commercial Scale-up
Introduction to Advanced Glycoside Synthesis Technology
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with environmental sustainability, a challenge effectively addressed by the chemical synthesis process detailed in patent CN1428345A. This groundbreaking technology focuses on the preparation of gastrodin and its analogous phenolic glycosides, which are critical active ingredients in various therapeutic formulations known for their sedative and hypnotic properties. Unlike traditional methods that rely on hazardous reagents, this novel approach utilizes industrially accessible materials such as acetic anhydride and acetyl bromide to achieve hydroxyl protection and hemiacetal bromination simultaneously. By fundamentally redesigning the activation strategy for the sugar moiety, the process mitigates the severe safety risks associated with red phosphorus and elemental bromine, thereby establishing a new benchmark for a reliable pharmaceutical intermediates supplier aiming to modernize their production capabilities. The innovation lies not just in the reagents but in the holistic optimization of reaction conditions that ensure high yields while maintaining stringent safety protocols essential for modern GMP facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of gastrodin and related glycosides has been plagued by significant safety and environmental drawbacks inherent to the Koenigs-Knorr reaction variants used in early methodologies. Traditional routes typically necessitate the use of red phosphorus and elemental bromine to generate the reactive glycosyl bromide intermediates, a practice that poses severe threats to production personnel through potential exposure to toxic fumes and corrosive substances. Furthermore, the generation of phosphorus-containing waste streams creates a substantial burden on wastewater treatment facilities, leading to elevated operational costs and complex regulatory compliance issues for manufacturers. The reliance on heavy metal promoters such as silver or mercury salts in alternative classical methods further exacerbates the problem by introducing expensive reagents that require rigorous removal steps to meet pharmaceutical purity standards. These legacy processes often suffer from low overall yields and inconsistent batch-to-batch reproducibility, making them increasingly untenable for cost reduction in API manufacturing where efficiency and safety are paramount concerns for stakeholders.
The Novel Approach
In stark contrast to these archaic techniques, the process described in CN1428345A introduces a streamlined and safer methodology that leverages acetic anhydride combined with acetyl bromide or phosphorus tribromide for the dual purpose of protection and activation. This strategic shift eliminates the need for elemental bromine and red phosphorus entirely, thereby removing the associated occupational health hazards and simplifying the waste management profile of the synthesis. The new route operates under mild conditions, typically maintaining reaction temperatures below 40°C, which significantly reduces energy consumption and minimizes the risk of thermal runaway incidents common in exothermic halogenation reactions. By utilizing readily available industrial chemicals, the process enhances supply chain reliability and reduces dependency on specialized or controlled reagents that might face procurement bottlenecks. This modernization of the synthetic pathway represents a pivotal advancement for any organization seeking the commercial scale-up of complex phenolic glycosides, offering a viable path to high-purity products with a drastically reduced environmental footprint.
Mechanistic Insights into Acetyl-Bromide Mediated Glycosylation
The core of this technological breakthrough resides in the efficient conversion of free sugars into reactive bromoacetyl glycosyl compounds through a concerted acetylation and bromination mechanism. In the first stage, the hydroxyl groups of the sugar substrate, such as glucose or galactose, are rapidly acetylated by acetic anhydride, often catalyzed by trace amounts of perchloric acid, forming a peracetylated intermediate that stabilizes the sugar ring. Subsequently, the anomeric hydroxyl group is selectively substituted with a bromine atom using acetyl bromide or phosphorus tribromide, generating the crucial alpha-bromo donor species required for glycosidic bond formation. This transformation proceeds with exceptional efficiency, reportedly achieving total yields exceeding 95% across a wide variety of monosaccharide and disaccharide substrates, demonstrating the versatility of the method. The use of acetyl bromide serves a dual function by providing both the acetyl group for protection and the bromide ion for activation, streamlining the reagent inventory and simplifying the reaction workup procedures significantly.
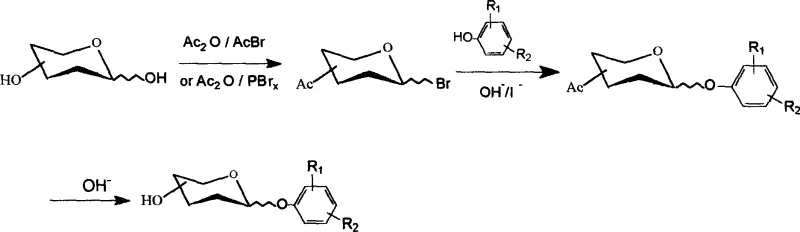
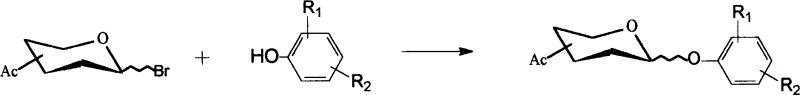
Following the activation of the sugar donor, the subsequent condensation with phenolic acceptors is facilitated by a unique solvent system comprising acetone and water, which balances the solubility of both organic and inorganic components. The addition of catalytic amounts of iodide ions, typically from potassium iodide, plays a critical role in enhancing the nucleophilicity of the phenolic oxygen and promoting the displacement of the anomeric bromide via an SN2-like mechanism. This iodide-catalyzed glycosylation allows the reaction to proceed smoothly at room temperature, avoiding the harsh conditions that often lead to sugar degradation or anomerization side reactions. The careful optimization of the acetone-to-water ratio ensures that hydrolysis of the reactive bromide is minimized while maintaining a homogeneous reaction medium that supports rapid kinetics. This mechanistic elegance results in the formation of the desired beta-glycosidic linkage with high stereoselectivity, which is essential for the biological activity of the final gastrodin analogues.
How to Synthesize Gastrodin Efficiently
The practical implementation of this synthesis route involves a logical sequence of operations designed for maximum throughput and minimal waste generation in a pilot or production plant setting. Operators begin by preparing the bromoacetyl glycosyl donor through the controlled addition of acetic anhydride and the brominating agent to the sugar substrate, ensuring strict temperature control to prevent exotherms. Once the activated donor is isolated or prepared in situ, it is introduced into the condensation reactor containing the phenolic acceptor, base, and iodide catalyst dissolved in the optimized acetone-water mixture. Reaction progress is monitored via thin-layer chromatography (TLC) to determine the precise endpoint, after which the protected glycoside precipitates or is extracted for the final deprotection step. Detailed standardized synthesis steps see the guide below.
- Synthesize bromoacetyl glycosyl compounds by reacting sugars with acetic anhydride and acetyl bromide or phosphorus tribromide at temperatures below 40°C.
- Condense the bromoacetyl glycosyl compound with a phenolic compound in an acetone-water solvent system using catalytic potassium iodide and sodium hydroxide.
- Remove acetyl protecting groups using sodium methoxide in methanol or sodium ethoxide in ethanol to obtain the final phenolic glycoside product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis technology offers compelling advantages that directly address the pain points of cost volatility and supply discontinuity often faced in the fine chemical sector. By substituting hazardous and regulated reagents like red phosphorus with commodity chemicals such as acetic anhydride, manufacturers can secure a more stable supply chain that is less susceptible to regulatory shutdowns or transportation restrictions. The elimination of heavy metal catalysts removes the need for expensive and time-consuming purification steps dedicated to residual metal removal, which translates into significant operational savings and faster batch release times. Furthermore, the mild reaction conditions reduce the demand for specialized high-pressure or high-temperature equipment, allowing for the utilization of standard glass-lined reactors that are commonplace in existing facilities. These factors collectively contribute to a more resilient manufacturing model that can adapt quickly to market demands without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The transition to non-toxic, industrially abundant reagents fundamentally alters the cost structure of producing phenolic glycosides by removing the premium associated with handling hazardous materials. Eliminating the need for specialized waste treatment for phosphorus and bromine residues lowers the overhead costs related to environmental compliance and disposal fees significantly. Additionally, the high yields reported in the initial activation step (>95%) ensure that raw material utilization is maximized, reducing the effective cost per kilogram of the intermediate. The simplified workup procedures, which often involve straightforward filtration and washing rather than complex chromatographic separations, further drive down labor and solvent costs, making the process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: Sourcing acetic anhydride and simple phenols is far more predictable than procuring red phosphorus or elemental bromine, which are often subject to strict government controls and security monitoring. This shift allows procurement managers to diversify their supplier base and negotiate better terms due to the commoditized nature of the new input materials. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures or safety incidents, ensuring a consistent flow of high-purity gastrodin to downstream customers. This reliability is crucial for maintaining long-term contracts with pharmaceutical clients who require guaranteed delivery timelines to support their own drug formulation schedules.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scalability, with patent examples demonstrating successful execution in batch sizes ranging from laboratory scale up to 500 kilograms. The absence of toxic heavy metals and volatile halogens simplifies the environmental impact assessment, facilitating faster regulatory approvals for new production lines in regions with strict emission standards. The aqueous-organic solvent system is easier to recover and recycle compared to purely organic systems, aligning with green chemistry principles and corporate sustainability goals. This scalability ensures that the technology can grow with demand, supporting reducing lead time for high-purity glycosides as market volume increases without requiring disproportionate capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for gastrodin and its analogues. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these specifics is vital for evaluating the feasibility of adopting this technology within your existing manufacturing infrastructure.
Q: How does this process improve safety compared to traditional gastrodin synthesis?
A: This process eliminates the use of highly toxic red phosphorus and elemental bromine, replacing them with industrially available acetic anhydride and acetyl bromide, significantly reducing environmental pollution and worker safety risks.
Q: What are the optimal conditions for the glycosidic condensation step?
A: The optimal conditions involve an acetone-to-water ratio of 2:1, a raw material ratio of bromoacetyl sugar to phenol of 2:3, and the use of 0.02 equivalents of potassium iodide as a catalyst at room temperature.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process is suitable for industrial production with batch sizes ranging from 1 to 500 kilograms, offering short production cycles and low costs due to readily available raw materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gastrodin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis technology in delivering high-value pharmaceutical intermediates to the global market. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards, guaranteeing that the gastrodin and phenolic glycosides we produce meet the exacting requirements of modern drug development. We leverage our deep technical expertise to optimize every parameter of this process, from reagent sourcing to final crystallization, maximizing yield and minimizing environmental impact for our clients.
We invite forward-thinking pharmaceutical companies to collaborate with us to harness the benefits of this safer and more efficient synthetic route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our capabilities align with your strategic sourcing goals. Together, we can accelerate the availability of essential therapeutic agents while upholding the highest standards of safety and sustainability in chemical manufacturing.
