Advanced Green Synthesis of Nitrile Compounds for Commercial Pharmaceutical Manufacturing
Advanced Green Synthesis of Nitrile Compounds for Commercial Pharmaceutical Manufacturing
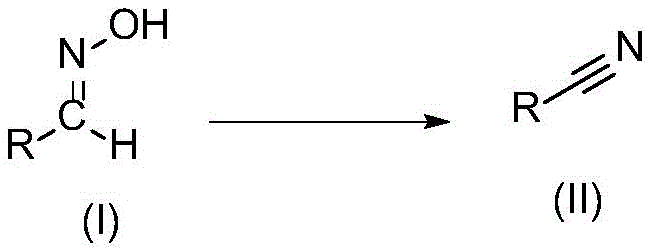
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, safer, and more cost-effective synthetic methodologies. Patent CN115322116B, published in May 2023, introduces a groundbreaking preparation technology for nitrile compounds that addresses critical pain points in the production of pharmaceutical and agrochemical intermediates. This innovation leverages a novel fluorosulfonyl imidazolium salt as a highly efficient promoter to facilitate the dehydration of aldoximes into nitriles. Unlike traditional methods that rely on toxic cyanide sources or harsh gaseous reagents, this protocol operates under remarkably mild conditions, typically between 25°C and 50°C, and achieves completion in as little as 10 minutes. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards sustainable manufacturing that does not compromise on yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the cyano group has been fraught with significant safety and operational challenges that impact both cost and supply chain stability. Classical approaches often involve the use of stoichiometric amounts of highly toxic cyanide salts, such as zinc cyanide or copper cyanide, which pose severe environmental hazards and require rigorous waste management protocols. Furthermore, recent literature highlights methods employing transition metal catalysts like nickel chloride with specialized ligands, which not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final API. Other reported strategies utilize gaseous reagents like sulfuryl fluoride (SO2F2), a potent greenhouse gas that requires specialized equipment for safe handling and poses regulatory compliance issues. These conventional pathways often necessitate elevated temperatures exceeding 60°C and prolonged reaction times up to 16 hours, leading to higher energy consumption and reduced throughput in large-scale reactors.
The Novel Approach
In stark contrast, the technology disclosed in CN115322116B utilizes a stable, solid fluorosulfonyl imidazolium salt to drive the conversion of aldoximes to nitriles via a beta-elimination mechanism. This approach eliminates the need for transition metals and toxic cyanide sources entirely. The reaction is characterized by its exceptional efficiency, proceeding rapidly at ambient to slightly elevated temperatures. The use of a solid promoter simplifies logistics and storage compared to handling hazardous gases. Moreover, the workup procedure is drastically simplified; the promoter and byproducts can be removed simply by washing with water, avoiding complex chromatographic purifications. This streamlined process significantly enhances cost reduction in nitrile manufacturing by reducing both raw material expenses and downstream processing time.
Mechanistic Insights into Fluorosulfonyl Imidazolium Salt Promoted Dehydration
The core of this technological advancement lies in the unique reactivity of the fluorosulfonyl imidazolium salt, structurally defined in the patent as Formula A. This reagent acts as a powerful electrophilic activator for the hydroxyl group of the aldoxime substrate. Upon interaction with the base, the fluorosulfonyl group facilitates the formation of a reactive intermediate that undergoes rapid beta-elimination to release water and form the carbon-nitrogen triple bond. The presence of the triflate counterion and the specific imidazolium cation structure ensures high solubility and reactivity in common organic solvents such as acetonitrile, dichloromethane, and ethyl acetate. This mechanistic pathway avoids the formation of stable metal-complex intermediates, thereby preventing the entrapment of product within catalyst coordination spheres which often leads to yield loss in metal-catalyzed systems.
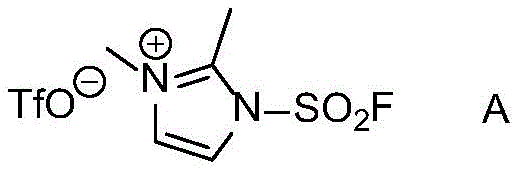
From an impurity control perspective, this metal-free mechanism offers distinct advantages for producing high-purity nitrile compounds. The absence of transition metals means there is no risk of residual nickel, palladium, or copper in the final product, which is a critical specification for pharmaceutical ingredients. The primary byproduct of the reaction is the corresponding imidazole derivative, which is highly water-soluble and easily separated during the aqueous extraction phase. This clean reaction profile minimizes the formation of complex side products that are difficult to separate, ensuring that the crude product often meets stringent purity specifications with minimal additional processing. The broad substrate scope demonstrated in the patent, covering electron-rich and electron-deficient aromatic aldoximes, confirms the robustness of this mechanistic approach across diverse chemical spaces.
How to Synthesize Nitrile Compounds Efficiently
The implementation of this synthesis route is designed for operational simplicity, making it highly attractive for process chemists aiming to scale up production. The general procedure involves charging a reactor with the aldoxime starting material, the fluorosulfonyl imidazolium salt promoter, and a selected solvent. A base, such as triethylamine, potassium carbonate, or DBU, is then added to initiate the reaction. The mixture is stirred at mild temperatures, typically between 25°C and 50°C, for a short duration ranging from 10 to 100 minutes depending on the specific substrate. Following the reaction, the mixture is subjected to a straightforward aqueous workup involving extraction with ethyl acetate and washing with water and brine. The detailed standardized synthesis steps for specific substrates are outlined in the guide below.
- Prepare the reaction mixture by combining the aldoxime substrate, fluorosulfonyl imidazolium salt promoter, and a suitable organic solvent such as acetonitrile or dichloromethane.
- Add a base such as triethylamine, potassium carbonate, or DBU to the mixture to facilitate the beta-elimination process.
- Stir the reaction at mild temperatures between 25°C and 50°C for 10 to 100 minutes, followed by aqueous workup to isolate the high-purity nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits regarding cost, safety, and reliability. The elimination of expensive transition metal catalysts and toxic cyanide reagents directly reduces the bill of materials. Furthermore, the simplified workup procedure, which relies on basic aqueous washing rather than complex purification techniques, significantly lowers utility costs and waste disposal fees. The use of stable solid reagents enhances supply chain resilience by removing the dependency on hazardous gas cylinders or sensitive organometallic complexes that require cold chain logistics. This robustness ensures consistent production schedules and reduces the risk of shutdowns due to reagent availability issues.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of high-cost inputs and the optimization of processing time. By avoiding precious metal catalysts, the raw material cost per kilogram of product is substantially lowered. Additionally, the rapid reaction kinetics allow for higher reactor turnover rates, effectively increasing plant capacity without capital expenditure. The simplified isolation process reduces solvent consumption and labor hours associated with purification, contributing to overall margin improvement.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents is inherently less risky than managing toxic gases or air-sensitive metals. The fluorosulfonyl imidazolium salt is shelf-stable and easy to transport, mitigating logistical bottlenecks. This stability ensures that production can continue uninterrupted even during supply chain disruptions that might affect specialized reagents. The broad availability of aldoxime precursors further secures the upstream supply chain, allowing for flexible sourcing strategies.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable at mild temperatures and the absence of hazardous gas evolution. This makes it ideal for commercial scale-up of complex intermediates in multi-ton quantities. From an environmental standpoint, the avoidance of cyanide and greenhouse gases aligns with increasingly strict global regulations, reducing the compliance burden and potential liability associated with hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical application of this method for industrial partners.
Q: What are the safety advantages of this nitrile synthesis method compared to traditional cyanide routes?
A: This method completely avoids the use of highly toxic cyanide salts and hazardous gases like sulfuryl fluoride, utilizing stable solid reagents instead.
Q: Does this process require expensive transition metal catalysts?
A: No, the process is metal-free, eliminating the need for costly nickel or palladium catalysts and simplifying downstream purification.
Q: What is the typical reaction time and temperature for this transformation?
A: The reaction proceeds efficiently at mild temperatures ranging from 25°C to 50°C and typically completes within 10 to 100 minutes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrile Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting innovative synthetic technologies to deliver superior value to our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green nitrile synthesis are realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of nitrile intermediate meets the highest standards required for pharmaceutical and agrochemical applications. We are committed to leveraging such advanced patents to enhance our portfolio of high-purity nitrile compounds.
We invite you to collaborate with us to evaluate the feasibility of this route for your specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how we can optimize your supply chain for reducing lead time for high-purity nitriles while maintaining uncompromised quality.
