Advanced Ruthenium Catalysis for Green Nitrile Synthesis and Commercial Scale-Up
The chemical industry is currently witnessing a paradigm shift towards greener, atom-economical synthesis routes, particularly in the production of high-value nitrile intermediates which are foundational to pharmaceuticals and agrochemicals. Patent CN110294689B, filed in 2021, introduces a groundbreaking methodology utilizing specifically designed ruthenium metal complexes to catalyze the direct dehydrogenation of primary amines into nitrile compounds. This innovation addresses critical pain points in traditional synthetic chemistry by eliminating the need for hazardous cyanating agents and strong oxidants, replacing them with a clean catalytic cycle that releases hydrogen gas as the sole byproduct. For R&D directors and process chemists, this represents a significant leap forward in sustainable manufacturing, offering a pathway to high-purity nitriles with exceptional selectivity and minimal environmental footprint. The technology not only simplifies the purification process but also aligns with global regulatory trends demanding safer chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of nitrile compounds has relied heavily on methods that pose severe safety and environmental challenges, creating substantial liabilities for supply chain managers and EHS officers. Traditional routes often involve the Sandmeyer reaction using aryl diazonium salts or the nucleophilic substitution of halides with inorganic cyanides like sodium cyanide or potassium cyanide. These processes are fraught with difficulties, including the handling of extremely toxic reagents that require specialized containment facilities and rigorous waste treatment protocols to prevent ecological damage. Furthermore, alternative oxidative methods using transition metals often necessitate stoichiometric amounts of strong oxidants, leading to poor atom economy and the generation of large volumes of hazardous waste streams that drive up disposal costs. The harsh reaction conditions associated with these legacy methods frequently result in lower selectivity, producing complex impurity profiles that complicate downstream purification and reduce overall process efficiency.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a homogeneous catalytic system based on ruthenium complexes to achieve direct dehydrogenation, fundamentally altering the economic and safety profile of nitrile production. This method operates under relatively mild thermal conditions, typically between 60°C and 110°C, using common organic solvents like toluene, which facilitates easier scale-up and reactor compatibility. By avoiding the use of external oxidants and toxic cyanide sources, the process inherently reduces the risk of workplace accidents and eliminates the regulatory burden associated with hazardous material transport and storage. The reaction mechanism is driven by the efficient removal of two hydrogen molecules from the amine substrate, resulting in a clean transformation where the only byproduct is molecular hydrogen, a valuable resource that can be captured for energy or other chemical syntheses. This shift from a waste-generating oxidation process to a hydrogen-releasing dehydrogenation process exemplifies the principles of green chemistry, offering a sustainable solution for the manufacture of complex organic intermediates.
Mechanistic Insights into Ruthenium-Catalyzed Dehydrogenation
The core of this technological advancement lies in the precise design of the ruthenium metal complex catalysts, which feature pyridine amide N,N-bidentate ligands coordinated to the ruthenium center. As illustrated in the structural diagrams, these complexes (designated as Formula I and II in the patent) incorporate specific substituents such as phenyl or isopropyl groups on the amide nitrogen, which fine-tune the electronic and steric environment around the metal center to optimize catalytic activity. The presence of arene ligands like benzene or p-cymene further stabilizes the complex, ensuring robustness under the reaction conditions required for amine activation. The catalytic cycle likely involves the coordination of the primary amine to the ruthenium center, followed by beta-hydride elimination steps that sequentially remove hydrogen atoms to form the imine intermediate and finally the nitrile product. The high stability of these catalysts allows them to maintain activity over extended reaction times, typically 16 to 32 hours, ensuring complete conversion of the substrate.
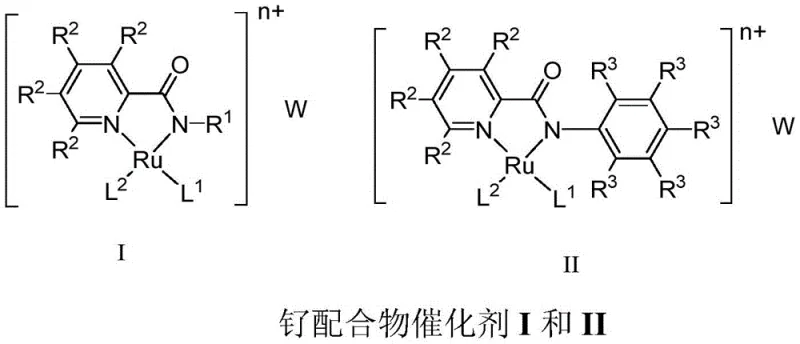
Understanding the mechanistic nuances is crucial for controlling impurity profiles, a key concern for quality assurance teams in pharmaceutical manufacturing. The high selectivity of the ruthenium catalyst minimizes side reactions such as over-oxidation or polymerization, which are common pitfalls in amine chemistry. The use of cesium carbonate as a base in the reaction mixture plays a pivotal role in facilitating the deprotonation steps necessary for the catalytic cycle while maintaining a pH environment that suppresses the formation of unwanted byproducts. By carefully controlling the molar ratios of the catalyst, base, and substrate, manufacturers can achieve yields as high as 95% for model substrates like benzylamine, demonstrating the precision of this chemical transformation. This level of control ensures that the final nitrile product meets stringent purity specifications required for downstream applications in drug synthesis, reducing the need for extensive chromatographic purification.
How to Synthesize Nitrile Compounds Efficiently
The practical implementation of this synthesis route involves a straightforward two-stage process: first, the preparation of the active ruthenium catalyst, and second, the catalytic dehydrogenation reaction itself. The catalyst preparation is conducted in anhydrous isopropanol under inert atmosphere, reacting the ligand with a ruthenium precursor at moderate temperatures to ensure high yield and purity of the active species. Once the catalyst is isolated and dried, it is introduced into the main reaction vessel containing the primary amine substrate and base in dry toluene. The reaction is then heated to the optimal temperature range and stirred under a flowing nitrogen environment to drive the equilibrium towards nitrile formation by removing the generated hydrogen.
- Prepare the ruthenium catalyst by reacting a pyridine amide N,N-bidentate ligand with a metal ruthenium compound in anhydrous isopropanol at 45-90°C for 5-25 hours.
- In a dry toluene solvent under nitrogen protection, mix cesium carbonate and the prepared ruthenium complex catalyst, then add the primary amine substrate.
- Heat the reaction mixture to 60-110°C and stir for 16-32 hours to achieve dehydrogenation, followed by solvent removal and purification to isolate the target nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed technology offers compelling strategic advantages that extend beyond mere technical feasibility to impact the bottom line and operational resilience. The elimination of highly toxic cyanide reagents drastically simplifies the supply chain logistics, removing the need for specialized hazardous material vendors and reducing the insurance and compliance costs associated with storing dangerous chemicals. Furthermore, the simplicity of the catalyst preparation and the use of commodity solvents like toluene and isopropanol ensure that raw material sourcing remains stable and cost-effective, shielding production schedules from volatility in specialty reagent markets. The robust nature of the catalyst also implies longer shelf-life and easier handling, reducing waste due to degradation and ensuring consistent batch-to-batch performance.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the workflow and the reduction in waste management overhead. By removing the requirement for expensive and hazardous oxidants and cyanating agents, the direct material costs are significantly lowered, while the absence of toxic waste streams reduces the financial burden of environmental compliance and disposal. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall mass balance and effective yield of the process. The ability to generate hydrogen as a useful byproduct rather than a waste gas further adds potential value, as this gas can be recycled within the plant for other hydrogenation processes or utilized for energy generation.
- Enhanced Supply Chain Reliability: The broad substrate scope of this catalytic system, which accommodates various substituted benzylamines, heterocyclic amines, and aliphatic chains, provides supply chain teams with greater flexibility in sourcing raw materials.
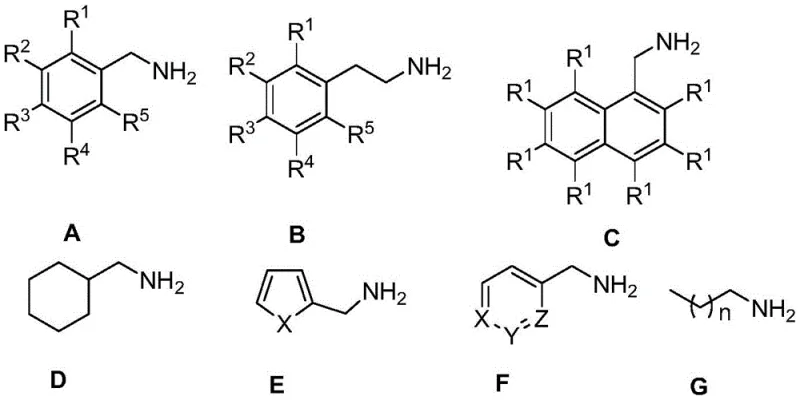 This versatility means that manufacturers are not locked into a single, rigid synthetic route but can adapt to the availability of different amine precursors without re-engineering the entire process. The mild reaction conditions also reduce the stress on production equipment, lowering maintenance frequency and extending the operational life of reactors, which contributes to more reliable long-term production capacity and shorter lead times for high-purity nitrile intermediates.
This versatility means that manufacturers are not locked into a single, rigid synthetic route but can adapt to the availability of different amine precursors without re-engineering the entire process. The mild reaction conditions also reduce the stress on production equipment, lowering maintenance frequency and extending the operational life of reactors, which contributes to more reliable long-term production capacity and shorter lead times for high-purity nitrile intermediates. - Scalability and Environmental Compliance: From a scalability perspective, the homogeneous nature of the catalysis and the use of standard organic solvents make the transition from laboratory scale to commercial production seamless and predictable. The process avoids the exothermic risks associated with strong oxidants, allowing for safer operation in large-scale reactors without the need for complex cooling systems or emergency quenching protocols. Environmentally, the process aligns perfectly with modern sustainability goals by adhering to green chemistry principles, producing no heavy metal waste or toxic effluents, and generating only benign hydrogen gas. This eco-friendly profile facilitates easier permitting and regulatory approval in jurisdictions with strict environmental laws, ensuring uninterrupted operations and enhancing the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the operational parameters and capabilities of this ruthenium-catalyzed system. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for process assessment. Understanding these details is essential for integrating this method into existing manufacturing workflows and maximizing its potential benefits.
Q: What are the safety advantages of this ruthenium-catalyzed method compared to traditional cyanation?
A: Unlike traditional methods that require highly toxic cyanating reagents like potassium cyanide or explosive strong oxidants, this catalytic dehydrogenation process uses benign primary amines and produces only hydrogen gas as a byproduct, significantly eliminating environmental pollution and safety hazards.
Q: What is the expected yield for benzylamine conversion using this technology?
A: According to the patent data, the conversion of benzylamine to benzonitrile using the specific ruthenium complex catalysts (such as Cat.A) can achieve yields of up to 95%, demonstrating high efficiency and selectivity.
Q: Can this catalytic system handle diverse substrate structures beyond simple benzylamines?
A: Yes, the catalyst system exhibits broad substrate applicability, successfully catalyzing the dehydrogenation of various primary amines including substituted benzylamines, heterocyclic amines, and aliphatic amines, making it versatile for complex intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrile Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ruthenium-mediated dehydrogenation described in CN110294689B for the production of high-value nitrile intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nitrile compound delivered meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with our technical team to explore how this green synthesis route can be tailored to your specific product portfolio and cost targets. By engaging with us, you can request a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this catalytic method for your supply chain. We encourage you to contact our technical procurement team today to obtain specific COA data for our nitrile intermediates and to discuss detailed route feasibility assessments that will secure your supply of critical chemical building blocks for the future.
