Advanced Visible-Light Mediated Synthesis of Heterocyclic Sulfones and Sulfonamides for Commercial Production
The landscape of organic synthesis for high-value heterocyclic scaffolds is undergoing a significant transformation, driven by the urgent need for greener, safer, and more efficient manufacturing protocols. Patent CN109503472B introduces a groundbreaking preparation method for heterocyclic sulfone and heterocyclic sulfonamide compounds that fundamentally shifts away from hazardous traditional reagents. This technology leverages visible light photocatalysis, utilizing fluorescein as an organocatalyst to facilitate a radical coupling reaction between iodo-aromatic heterocyclic compounds and thiourea dioxide. For R&D Directors and Procurement Managers seeking a reliable pharmaceutical intermediates supplier, this patent represents a critical opportunity to optimize supply chains. By operating under mild conditions without strong corrosives, the process not only enhances operational safety but also ensures the production of high-purity heterocyclic sulfones with minimal byproduct formation, addressing key pain points in the manufacturing of complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial synthesis of sulfone and sulfonamide motifs has relied heavily on the oxidation of sulfides or the nucleophilic substitution of sulfonyl chlorides with amines. These legacy methods present substantial challenges for modern chemical manufacturing, particularly regarding safety and environmental compliance. The use of sulfonyl chlorides requires stringent moisture control and generates corrosive hydrogen chloride gas as a byproduct, necessitating expensive scrubbing systems and specialized corrosion-resistant reactor materials. Furthermore, the oxidation steps often involve strong oxidizing agents that lack chemoselectivity, leading to the over-oxidation of sensitive functional groups and the generation of complex impurity profiles that are difficult and costly to remove. These factors collectively drive up the cost reduction in fine chemical manufacturing and pose significant risks to supply chain continuity due to the handling of hazardous materials.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a visible-light mediated radical coupling strategy that operates at room temperature, effectively bypassing the need for harsh oxidants or corrosive chlorinating agents. The process begins with the generation of aromatic heterocyclic sodium sulfinate through a photocatalytic cycle involving thiourea dioxide and sodium hydroxude, which serves as a stable and safe sulfur dioxide equivalent. This intermediate is then seamlessly converted into the final sulfone or sulfonamide via alkylation or amidation in a second step. This two-step sequence not only simplifies the operational workflow but also drastically improves the atom economy and safety profile of the synthesis. By eliminating the most dangerous reagents from the process flow, manufacturers can achieve substantial cost savings in terms of waste treatment and equipment maintenance, while simultaneously ensuring a more robust and reliable supply of critical chemical building blocks.
Mechanistic Insights into Fluorescein-Catalyzed Radical Coupling
The core innovation of this technology lies in the precise mechanistic pathway enabled by the fluorescein photocatalyst under visible light irradiation. Upon excitation by a standard fluorescent lamp (20-30W), fluorescein enters an excited state that facilitates single electron transfer (SET) processes. Thiourea dioxide reacts with sodium hydroxide to generate reductive sulfur dioxide anions, which then interact with the excited photocatalyst to produce sulfur dioxide negative charge free radicals. Concurrently, the iodo-aromatic heterocyclic compound undergoes reduction via the reduced fluorescein species to generate a heterocyclic carbon free radical. The convergence of these two radical species results in a highly efficient radical coupling reaction, forming the aromatic heterocyclic sodium sulfinate intermediate with exceptional selectivity. This mechanism avoids the high-energy transition states typical of thermal reactions, thereby preserving sensitive functional groups on the heterocyclic ring.
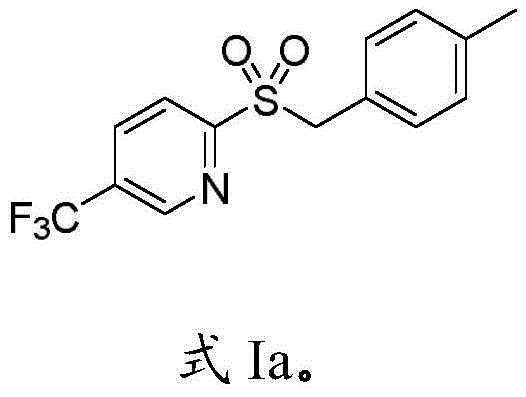
Following the formation of the sulfinate intermediate, the pathway diverges based on the desired final functionality, offering versatile access to both sulfones and sulfonamides. For sulfone synthesis, the sodium sulfinate undergoes an alkylation reaction with halogenated compounds or diaryl iodonium salts, often catalyzed by tetrabutylammonium iodide and potassium iodide to enhance reactivity. Alternatively, for sulfonamide production, the intermediate reacts with amine organic compounds in the presence of N-chlorosuccinimide. This amidation process is carefully controlled through a two-stage temperature protocol, starting at low temperatures to form the sulfinyl chloride in situ before warming to room temperature for coupling with the amine. This nuanced control over the reaction environment ensures that the final products, such as the representative structures shown in the patent examples, are obtained with high purity and yield, minimizing the formation of regioisomers or over-alkylated byproducts that often plague conventional methods.
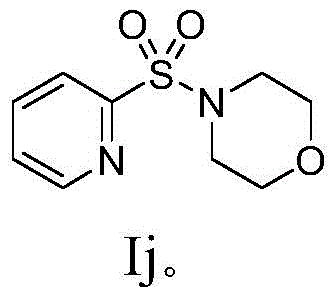
How to Synthesize Heterocyclic Sulfone Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters to maximize efficiency and reproducibility on a larger scale. The process initiates by mixing the iodo-aromatic heterocyclic substrate with thiourea dioxide, sodium hydroxide, and the fluorescein catalyst in dimethyl sulfoxide (DMSO) under a nitrogen or inert gas atmosphere. Visible light irradiation is maintained for approximately 16 to 18 hours at room temperature to ensure complete conversion to the sulfinate intermediate. Once this first stage is complete, the reaction mixture can be directly utilized for the subsequent transformation without isolation, which streamlines the workflow and reduces material loss. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures required to achieve the reported yields of up to 83%, are outlined in the technical guide below for immediate reference by process chemists.
- Conduct a radical coupling reaction between an iodo-aromatic heterocyclic compound, thiourea dioxide, and sodium hydroxide under visible light irradiation using fluorescein as a catalyst to form aromatic heterocyclic sodium sulfinate.
- Maintain a protective atmosphere and react the resulting sodium sulfinate intermediate with a group donor compound, such as a halogenated compound for alkylation or an amine for amidation.
- Quench the reaction with saturated brine, extract with ethyl acetate, and purify the final heterocyclic sulfone or sulfonamide via column chromatography and solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers profound strategic advantages beyond mere technical feasibility. The elimination of strong corrosive substances and heavy metal catalysts from the synthesis pathway directly translates to a simplified regulatory compliance landscape and reduced liability. Traditional methods often require extensive purification steps to remove trace metals or acidic byproducts, which adds time and cost to the manufacturing cycle. By contrast, this mild, organocatalytic approach produces fewer byproducts and utilizes reagents that are easier to handle and store, thereby enhancing supply chain reliability. The ability to operate at room temperature also significantly reduces energy consumption compared to high-temperature thermal processes, contributing to a lower carbon footprint and aligning with global sustainability goals that are increasingly important for multinational corporations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction workflow and the reduction in hazardous waste disposal costs. By avoiding the use of expensive and dangerous sulfonyl chlorides, manufacturers eliminate the need for specialized corrosion-resistant equipment and complex gas scrubbing systems. Furthermore, the high selectivity of the radical coupling reaction minimizes the formation of difficult-to-separate impurities, which reduces the load on downstream purification units like column chromatography or crystallization. This streamlined process flow allows for a more efficient use of raw materials and labor, resulting in substantial cost savings that can be passed down the supply chain, making the final API intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability and transportation restrictions of hazardous chemicals. The reagents used in this novel method, such as thiourea dioxide and fluorescein, are stable, commercially available, and not subject to the same strict transportation regulations as corrosive acids or toxic heavy metals. This stability ensures that production schedules are less likely to be disrupted by logistical delays or regulatory hurdles. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or pressure, leading to more consistent batch-to-batch quality. This reliability is crucial for maintaining the steady flow of high-purity pharmaceutical intermediates required for continuous drug manufacturing operations.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden challenges related to heat transfer and safety. The visible-light mediated nature of this reaction, combined with its operation at ambient temperature, makes it inherently safer and easier to scale than exothermic oxidation reactions. The reduced generation of hazardous waste simplifies the environmental permitting process and lowers the long-term operational costs associated with waste treatment facilities. As environmental regulations become stricter globally, having a manufacturing process that is inherently green and compliant provides a significant competitive advantage, ensuring long-term viability and reducing the risk of future shutdowns due to non-compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or for sourcing these intermediates from external partners. The insights provided here cover aspects ranging from reaction scalability to impurity control, offering a comprehensive overview of the method's practical applications in the fine chemical industry.
Q: What are the primary safety advantages of this photocatalytic method over traditional sulfonyl chloride routes?
A: This method eliminates the need for strongly corrosive sulfonyl chlorides and irritant sulfurization reagents, significantly reducing EHS risks and equipment corrosion costs associated with traditional sulfone synthesis.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the reaction operates at room temperature under visible light with common solvents like DMSO, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates without requiring extreme thermal conditions.
Q: What level of purity can be expected from this synthetic route?
A: Experimental data indicates that the process yields products with high purity, often exceeding 96% by HPLC, due to the mild reaction conditions which minimize side reactions and byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the pharmaceutical and fine chemical industries. Our team of expert chemists has extensively evaluated the photocatalytic route described in CN109503472B and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the high quality of heterocyclic sulfones and sulfonamides produced via this method, guaranteeing that every batch meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this innovative technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to not just a product, but a comprehensive solution that includes specific COA data and route feasibility assessments to ensure your supply chain is optimized for efficiency and resilience. Contact us today to discuss how we can support your development goals with high-quality, sustainably produced chemical intermediates.
