Scalable Metal-Free Synthesis of Heterocyclic Sulfones for Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Heterocyclic Sulfones for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, sustainable, and cost-effective methodologies for constructing complex molecular architectures, particularly sulfone-containing heterocycles which are prevalent in bioactive molecules. Patent CN111072555A introduces a groundbreaking approach to synthesizing heterocyclic sulfone organic compounds by leveraging a metal-free visible-light photocatalytic system. This innovation utilizes diethyl 2,6-dimethyl-1,4-dihydro-3,5-pyridinedicarboxylate, commonly known as HEH, as an organic photoredox catalyst to drive the cross-coupling of heterocyclic aryl halides with aryl sulfinates. For R&D directors and procurement managers, this technology represents a significant paradigm shift away from traditional transition-metal catalysis, offering a pathway to high-purity intermediates with reduced regulatory burdens regarding heavy metal residues. The process operates under mild conditions, specifically at room temperature using blue LED irradiation, which drastically lowers energy consumption and enhances operational safety compared to thermal methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfone compounds has relied heavily on transition metal catalysts such as palladium or copper, often requiring elevated temperatures that can degrade sensitive functional groups and lead to complex impurity profiles. Alternative oxidative methods involving thioethers frequently suffer from poor selectivity, generating unwanted byproducts like sulfoxides and necessitating difficult purification steps that lower overall yield. Furthermore, advanced photocatalytic methods utilizing ruthenium or iridium complexes, while effective, introduce substantial cost pressures due to the scarcity and high price of these noble metals. From a supply chain perspective, reliance on these precious metals creates vulnerability to market volatility and geopolitical supply disruptions. Additionally, the stringent requirements for removing trace metal contaminants from final API intermediates add significant time and cost to the downstream processing, making these conventional routes less attractive for large-scale commercial manufacturing where efficiency and purity are paramount.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles by employing an organic small molecule, HEH, as a visible light reduction catalyst, effectively eliminating the need for any auxiliary transition metal catalyst or noble metal photosensitizer. This metal-free strategy not only simplifies the reaction workup by removing the necessity for metal scavenging steps but also ensures a cleaner product profile suitable for sensitive pharmaceutical applications. The reaction utilizes a simple setup involving a blue LED light source (10W, wavelength 460-485 nm) and proceeds at room temperature, demonstrating remarkable compatibility with a wide range of functional groups including trifluoromethyl and cyano substituents on pyridine and quinoline rings. By solving the challenge of inducing aryl halides with higher reduction potentials using visible light, this approach opens new avenues for synthesizing complex heterocyclic sulfones that were previously difficult to access efficiently.

Mechanistic Insights into HEH-Catalyzed Visible Light Reduction
The core of this technological advancement lies in the unique redox properties of the HEH catalyst, which acts as a potent hydride donor and single-electron reducer upon photoexcitation. Under the irradiation of blue LEDs, the HEH molecule enters an excited state capable of transferring an electron to the heterocyclic aryl halide substrate, generating a reactive radical intermediate that facilitates the cross-coupling with the sulfinate species. This mechanism bypasses the high activation energy barriers typically associated with aryl halide activation, allowing the reaction to proceed smoothly at ambient temperatures without the need for harsh thermal conditions. The use of an inorganic base, preferably cesium carbonate, further stabilizes the reaction environment and promotes the formation of the sulfone bond with high selectivity. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction parameters for specific substrates, as it highlights the importance of light intensity and catalyst loading in driving the catalytic cycle to completion.
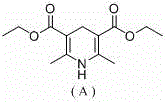
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity landscape of the final product. Traditional metal-catalyzed reactions often leave behind trace amounts of palladium, copper, or nickel, which are strictly regulated in pharmaceutical ingredients due to their toxicity. By utilizing an organic catalyst like HEH, the risk of heavy metal contamination is virtually eliminated, thereby simplifying the quality control process and reducing the need for expensive purification technologies such as preparative HPLC or specialized resin treatments. This results in a cleaner crude product and a more streamlined isolation process, directly contributing to higher overall yields and reduced production costs. Furthermore, the mild reaction conditions minimize the formation of thermal degradation byproducts, ensuring that the structural integrity of sensitive heterocyclic cores, such as those found in quinolines and thiophenes, is preserved throughout the synthesis.
How to Synthesize Heterocyclic Sulfones Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls as outlined in the patent embodiments. The standard protocol involves mixing the heterocyclic aryl halide, sodium benzene sulfinate, inorganic base, and HEH catalyst in dimethyl sulfoxide (DMSO) under an inert atmosphere to prevent oxidative side reactions. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining heterocyclic aryl halide, sodium benzene sulfinate, inorganic base (Cs2CO3), and HEH catalyst in DMSO solvent under inert gas protection.
- Irradiate the reaction mixture with a 10W blue LED (wavelength 460-485 nm) while stirring at room temperature for 24 hours to facilitate the cross-coupling reaction.
- Quench the reaction with water, extract with ethyl acetate, dry the organic phase, and purify the crude product via silica gel chromatography to obtain the target sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this HEH-catalyzed process offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of expensive noble metal catalysts like ruthenium and iridium translates directly into significant raw material cost savings, insulating the manufacturing process from the volatile pricing trends of the precious metals market. Moreover, the simplified downstream processing required to remove metal residues reduces the consumption of auxiliary chemicals and solvents, further driving down the cost of goods sold (COGS). The ability to run reactions at room temperature also implies a drastic reduction in energy consumption compared to thermal processes, aligning with global sustainability goals and reducing the facility's carbon footprint. These factors combined create a more resilient and cost-efficient supply chain capable of delivering high-quality intermediates with greater reliability.
- Cost Reduction in Manufacturing: The substitution of costly transition metal catalysts with the inexpensive organic molecule HEH removes a major cost driver from the bill of materials. Additionally, the avoidance of metal scavenging resins and the reduction in purification steps lower the overall operational expenditure, making the process economically superior for large-scale production runs where margin compression is a constant concern.
- Enhanced Supply Chain Reliability: By relying on readily available organic reagents and common inorganic bases rather than scarce noble metals, manufacturers can secure a more stable supply of critical inputs. This diversification of the supply base mitigates the risk of production stoppages caused by shortages of specialized catalysts, ensuring consistent delivery schedules for downstream pharmaceutical customers who depend on uninterrupted supply chains.
- Scalability and Environmental Compliance: The mild, metal-free nature of this reaction simplifies waste management and disposal, as there are no heavy metal contaminants to treat in the effluent streams. This ease of waste handling facilitates smoother regulatory approvals and faster scale-up from laboratory to commercial production, allowing companies to bring new products to market more rapidly while maintaining strict adherence to environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their own portfolios.
Q: What are the advantages of using HEH over traditional transition metal catalysts?
A: HEH eliminates the need for expensive and toxic transition metals like palladium or ruthenium, significantly reducing raw material costs and simplifying the removal of metal residues, which is critical for pharmaceutical compliance.
Q: Does this method require high temperatures or specialized equipment?
A: No, the reaction proceeds efficiently at room temperature under standard blue LED irradiation, removing the need for energy-intensive heating systems and allowing for safer, more scalable operations.
Q: What types of heterocyclic substrates are compatible with this synthesis?
A: The method demonstrates broad substrate scope, successfully coupling various heterocyclic aryl halides including pyridines, quinolines, and thiophenes bearing functional groups like trifluoromethyl and cyano groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free photocatalysis in modern pharmaceutical manufacturing and are fully equipped to leverage this technology for your specific project needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of heterocyclic sulfone intermediate meets the highest international standards, free from the heavy metal impurities that plague traditional synthesis methods.
We invite you to engage with our technical procurement team to discuss how this innovative HEH-catalyzed route can be integrated into your supply chain to achieve substantial cost savings and efficiency gains. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits specific to your target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique requirements, positioning your organization at the forefront of sustainable and efficient chemical manufacturing.
