Advanced Low-Temperature Synthesis of Cefdinir for Commercial Scale-Up of Complex Beta-Lactams
Advanced Low-Temperature Synthesis of Cefdinir for Commercial Scale-Up of Complex Beta-Lactams
The pharmaceutical industry is constantly seeking robust manufacturing pathways for third-generation cephalosporins, and the preparation method detailed in patent CN101565427B represents a significant leap forward in the production of Cefdinir. This specific intellectual property outlines a refined synthetic strategy that addresses the longstanding challenges of yield optimization and impurity control inherent in beta-lactam antibiotic manufacturing. By shifting away from traditional high-temperature hydrolysis and hazardous solvent systems, this technology enables the production of high-purity cephalosporin intermediates with superior consistency. For a reliable API intermediate supplier, adopting such methodologies is crucial for meeting the stringent quality standards required by global regulatory bodies. The core innovation lies in the precise manipulation of reaction thermodynamics, specifically through low-temperature acylation and hydrolysis steps that preserve the delicate beta-lactam ring structure.
Furthermore, the integration of green chemistry principles, such as the substitution of chlorinated solvents with recoverable alcohols and esters, aligns perfectly with modern environmental compliance mandates. This approach not only mitigates the ecological footprint of the manufacturing process but also simplifies the downstream purification workflows. As we delve deeper into the technical specifics, it becomes evident that this patent offers a viable solution for the commercial scale-up of complex beta-lactams, ensuring a steady supply of critical antibiotic ingredients. The ability to control the reaction profile so precisely allows manufacturers to minimize waste and maximize the efficiency of raw material utilization. Consequently, this technology stands as a benchmark for next-generation antibiotic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefdinir has been plagued by inefficiencies associated with traditional active ester methods and nitrosating agent routes, which often suffer from suboptimal yields and rigorous reaction conditions. As illustrated in the prior art reaction schemes, conventional Route 1 relies on the condensation of active intermediates which, while stable, incurs high production costs due to the expense of reagents and the complexity of the purification steps. Moreover, Route 2, which utilizes nitrosating agents to form the thiazole ring, is characterized by strict reaction conditions that are difficult to maintain on a large industrial scale, leading to inconsistent batch quality. A major drawback across these legacy methods is the reliance on solvents like tetrahydrofuran (THF) or methylene chloride, which pose significant environmental hazards and require complex recovery systems to meet safety regulations.
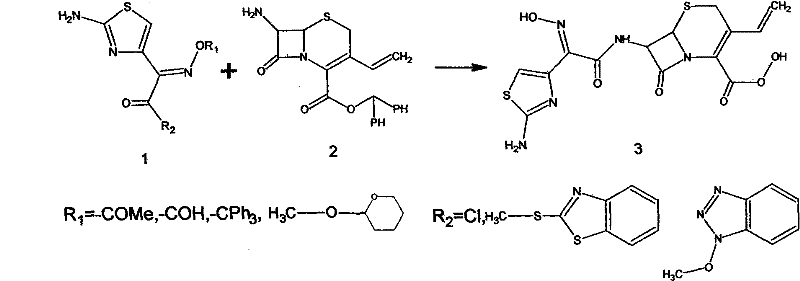
Additionally, the hydrolysis steps in conventional processes are typically conducted at elevated temperatures, which inadvertently promotes the degradation of the sensitive beta-lactam core and the formation of polymeric impurities. This thermal instability results in a lower overall yield, often hovering around 88%, and necessitates extensive downstream processing to remove these stubborn byproducts. The accumulation of such impurities not only affects the final potency of the drug but also complicates the crystallization process, making it difficult to achieve the particle size distribution required for optimal bioavailability. For procurement managers, these inefficiencies translate into higher raw material consumption and increased waste disposal costs, eroding the profit margins of the final pharmaceutical product. Therefore, there is a critical need for a method that can operate under milder conditions to preserve the integrity of the molecular structure.
The Novel Approach
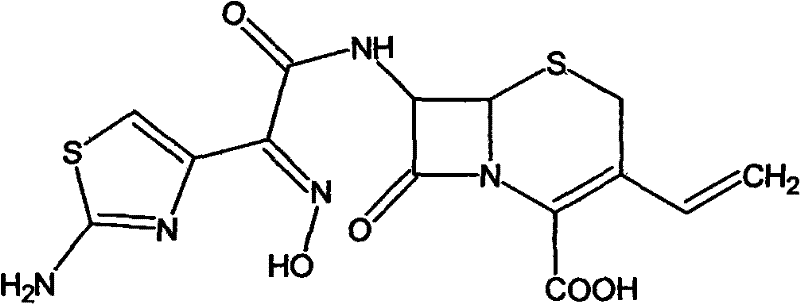
The novel approach presented in patent CN101565427B fundamentally reengineers the synthesis pathway by introducing a low-temperature reaction technique that dramatically enhances both yield and product quality. This method utilizes 7-amino-3-vinyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid (7-AVCA) reacting with a specific active ester in a solvent system comprising water and recoverable organic solvents like methanol, ethanol, or ethyl acetate. By maintaining the reaction temperature between -10°C and 30°C, the process effectively suppresses the kinetic energy that leads to unwanted side reactions, thereby preserving the stereochemical integrity of the molecule. The subsequent hydrolysis is performed under alkaline conditions at temperatures ranging from -10°C to 10°C, a stark contrast to the pyrohydrolysis of the past, which prevents the thermal degradation of the cephem nucleus.
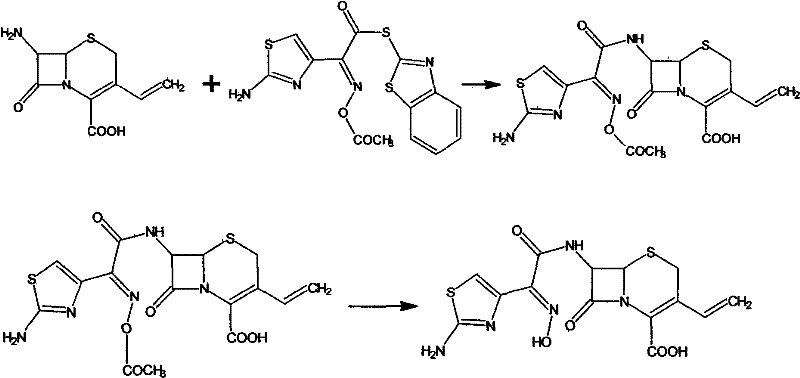
Furthermore, the strategic selection of solvents allows for easy recovery and reuse, significantly reducing the volume of three-wastes drain and lowering the overall environmental pollution associated with the manufacturing process. The result is a substantial improvement in reaction yield, which has been documented to increase from 88% in traditional methods to an impressive 95% with this new protocol. This enhancement in efficiency is coupled with a marked improvement in purity, with the final product consistently testing greater than 97% purity and exceeding 99% by HPLC analysis. For a reliable API intermediate supplier, this level of control translates directly into a more predictable supply chain and reduced risk of batch rejection. The simplicity of the hydrolysis and crystallization process further ensures that the method is highly suitable for large-scale industrial production without compromising on quality.
Mechanistic Insights into Low-Temperature Acylation and Hydrolysis
The mechanistic success of this synthesis relies heavily on the kinetic control exerted during the acylation of the 7-AVCA nucleus with the (Z)-2-(2-aminothiazole-4-yl)-2-acetoxyimino thioacetic acid ester. At low temperatures, the nucleophilic attack of the amino group on the activated carbonyl of the ester is highly selective, minimizing the formation of regioisomers or N-acylated byproducts that can occur at higher thermal energies. The presence of an organic base, such as triethylamine or tri-n-butylamine, facilitates the deprotonation of the amino group, enhancing its nucleophilicity without inducing elimination reactions that could compromise the vinyl group at the C-3 position. This delicate balance is crucial because the vinyl group is essential for the oral bioavailability of the final Cefdinir molecule, and its preservation is a key indicator of process robustness. The solvent mixture of water and organic modifiers creates a homogeneous phase that ensures efficient mass transfer while stabilizing the transition state of the acylation reaction.
Following the formation of the protected intermediate, the hydrolysis mechanism is equally critical, as it involves the cleavage of the ester protecting group to reveal the free carboxylic acid without opening the beta-lactam ring. By employing a strong base solution like sodium hydroxide or potassium hydroxide at temperatures between -10°C and 10°C, the hydrolysis proceeds via a nucleophilic acyl substitution that is fast enough to be practical but slow enough to avoid ring strain-induced opening. This low-temperature regime effectively quenches the thermal energy that would otherwise drive the polymerization of the beta-lactam rings, a common failure mode in cephalosporin synthesis. The rapid quenching and pH adjustment to the isoelectric point allow for the immediate precipitation of the product, locking it into a stable crystalline form before degradation pathways can initiate. This precise control over the hydrolysis kinetics is the primary driver behind the observed increase in yield and the superior impurity profile of the final active pharmaceutical ingredient.
How to Synthesize Cefdinir Efficiently
The synthesis of Cefdinir via this patented route involves a streamlined sequence of acylation, extraction, and low-temperature hydrolysis that is designed for operational simplicity and high throughput. The process begins with the dissolution of 7-AVCA and the active ester in a mixed solvent system, followed by the controlled addition of an organic base to initiate the coupling reaction under strictly monitored thermal conditions. Once the intermediate is formed and isolated through pH adjustment and extraction, it undergoes a mild alkaline hydrolysis to remove the protecting groups, followed by decolorization and final crystallization. The detailed standardized synthetic steps, including specific molar ratios, stirring times, and filtration parameters, are outlined in the comprehensive guide below to ensure reproducibility and compliance with GMP standards.
- React 7-AVCA with Cefdinir active methyl ester in a solvent system of water and alcohol/ketone/ester at temperatures between -10°C and 30°C using an organic base.
- Adjust pH to isolate the intermediate, extract byproducts, and decolorize the aqueous layer using activated carbon.
- Hydrolyze the intermediate using a strong base solution at low temperatures (-10°C to 10°C), followed by pH adjustment and crystallization to obtain high-purity Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this low-temperature synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. By eliminating the need for hazardous solvents like methylene chloride and replacing them with easily recoverable alcohols and esters, the process drastically simplifies the solvent recovery infrastructure, leading to substantial cost savings in utility and waste management. The reduction in reaction temperature also lowers the energy consumption required for heating and cooling cycles, contributing to a leaner and more sustainable manufacturing operation. Furthermore, the enhanced stability of the reaction intermediates reduces the risk of batch failures, ensuring a more consistent and reliable supply of critical antibiotic intermediates to downstream formulation partners. This reliability is paramount in the pharmaceutical sector, where supply disruptions can have cascading effects on patient access to essential medications.
- Cost Reduction in Manufacturing: The shift to recoverable solvents such as ethyl acetate and ethanol eliminates the high costs associated with the disposal and replacement of single-use chlorinated solvents, directly impacting the bottom line. Additionally, the increase in reaction yield from historical baselines to nearly 95% means that less raw material is required to produce the same amount of finished product, optimizing the cost of goods sold. The simplified purification process, which avoids complex chromatographic separations often needed to remove high-temperature impurities, further reduces the operational expenditure per kilogram of output. These cumulative efficiencies create a robust economic model that supports competitive pricing strategies in the global generic antibiotic market.
- Enhanced Supply Chain Reliability: The use of commodity chemicals like methanol, acetone, and sodium hydroxide ensures that the supply chain is not vulnerable to the shortages or price volatility often seen with specialized reagents. The mild reaction conditions reduce the wear and tear on reactor vessels and piping, extending the lifespan of capital equipment and minimizing unplanned maintenance downtime. By achieving a purity profile that consistently exceeds pharmacopeial standards, the need for reprocessing or re-crystallization is virtually eliminated, speeding up the release of batches for shipment. This operational fluidity allows suppliers to respond more agilely to fluctuations in market demand, securing their position as a trusted partner for major pharmaceutical companies.
- Scalability and Environmental Compliance: The process is inherently scalable because the heat management requirements are less demanding than exothermic high-temperature reactions, allowing for larger batch sizes without the risk of thermal runaway. The significant reduction in three-wastes drain, particularly the minimization of halogenated organic waste, ensures full compliance with increasingly stringent environmental regulations across different jurisdictions. This eco-friendly profile not only mitigates regulatory risk but also enhances the corporate social responsibility standing of the manufacturing entity. Consequently, the technology supports the long-term sustainability goals of the organization while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Cefdinir synthesis method, based on the specific data points and beneficial effects described in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or capacity expansion projects. The answers provided reflect the empirical results observed during the optimization of the low-temperature hydrolysis and solvent recovery systems.
Q: What are the primary advantages of the low-temperature hydrolysis method for Cefdinir?
A: The low-temperature hydrolysis method prevents product degradation and polymerization common in high-temperature processes, increasing yield from approximately 88% to 95% and ensuring purity greater than 97%.
Q: Which solvents are preferred in this green synthesis route?
A: The process utilizes easily recoverable alcohols, ketones, or esters such as methanol, ethanol, acetone, and ethyl acetate, replacing hazardous solvents like tetrahydrofuran (THF) or methylene chloride.
Q: How does this method impact environmental compliance?
A: By using recyclable solvents and reducing the generation of high-temperature impurities, the method significantly lowers three-waste drainage and reduces overall environmental pollution compared to conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN101565427B to deliver high-quality antibiotic intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of low-temperature synthesis are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of Cefdinir meets the highest international standards for potency and impurity profiles. Our commitment to process excellence allows us to offer a supply of high-purity cephalosporin intermediates that supports the development of safe and effective generic medications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized manufacturing process can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient synthesis route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition to a more reliable and cost-effective sourcing strategy.
