Revolutionizing Cefdinir Production: A Technical Analysis of Mild Acylation and Purification Strategies
Revolutionizing Cefdinir Production: A Technical Analysis of Mild Acylation and Purification Strategies
The pharmaceutical industry continuously seeks robust manufacturing pathways for third-generation cephalosporins, particularly Cefdinir, due to its broad-spectrum efficacy and favorable safety profile. Patent CN103319503A introduces a transformative preparation method that addresses critical bottlenecks in traditional synthesis, specifically focusing on the acylation of 7-amino-3-vinyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid (7-AVCA). This technical disclosure outlines a sophisticated approach utilizing an active ester strategy in a dichloromethane and tri-n-butylamine system, diverging significantly from legacy processes that rely on hazardous solvents and harsh acidic conditions. By optimizing the reaction environment to maintain temperatures between 20°C and 30°C, the method ensures exceptional stability of the beta-lactam ring while achieving molar yields exceeding 80% and product purity greater than 99%. For R&D directors and process engineers, this patent represents a pivotal shift towards greener, more controllable chemistry that minimizes degradation products and simplifies isolation protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefdinir has been plagued by significant operational hazards and purity challenges inherent to older methodologies. Traditional routes often employ chloride methods or active ester methods that necessitate the use of tosic acid and large volumes of diethyl ether as an anti-solvent to induce crystallization. The reliance on diethyl ether introduces severe safety risks due to its extremely low flash point, creating a high potential for fire and explosion during large-scale production. Furthermore, the removal of tosic acid requires high-temperature vacuum distillation, which subjects the thermally sensitive cephalosporin core to stress, potentially leading to ring opening or isomerization. Additionally, the use of anti-solvents frequently results in rapid, uncontrolled supersaturation, causing the coprecipitation of impurities and the formation of unstable agglomerates that compromise the genetic morphology of the crystals. These factors collectively result in inconsistent batch quality, lower overall yields typically ranging between 20% and 50% in less optimized variations, and substantial environmental burdens associated with solvent recovery and waste treatment.
The Novel Approach
In stark contrast, the methodology described in CN103319503A leverages a refined active ester coupling in a dichloromethane system buffered by tri-n-butylamine, effectively circumventing the pitfalls of previous generations. This novel approach eliminates the requirement for hazardous anti-solvents entirely, thereby removing the risk of uncontrolled precipitation and enhancing the safety coefficient of the production environment. The reaction proceeds under remarkably mild conditions, maintaining a temperature range of 20°C to 30°C, which preserves the integrity of the vinyl group and the beta-lactam ring. By utilizing tri-n-butylamine, the process avoids the corrosive effects of strong mineral acids during the coupling phase, reducing equipment maintenance costs and extending reactor lifespan. The subsequent purification strategy involves a unique phosphate salt precipitation step, which offers superior selectivity compared to direct crystallization, allowing for the effective removal of various impurities before the final deprotection. This streamlined workflow not only simplifies the operational steps but also facilitates solvent recycling, particularly for dichloromethane, aligning with modern principles of sustainable chemical manufacturing.
Mechanistic Insights into Tri-n-butylamine Mediated Acylation
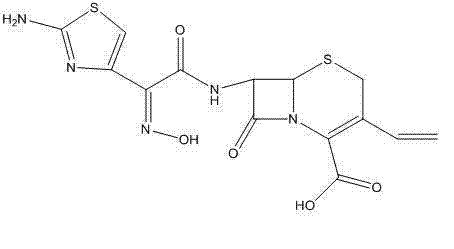
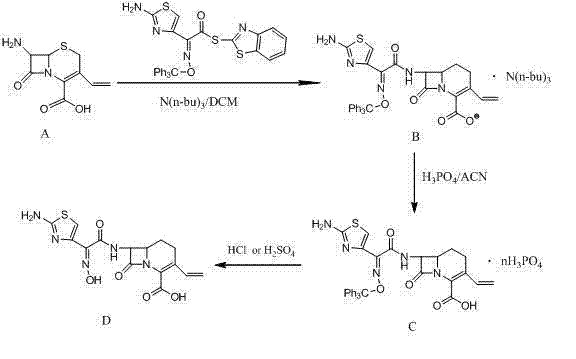
The core of this technological advancement lies in the precise control of the acylation mechanism using the (Z)-2-(2-amino-4-thiazolyl)-2-(trityloxyimino)thioacetic acid S-2-benzothiazolyl ester. In this system, tri-n-butylamine acts not merely as a base but as a crucial buffering agent that modulates the nucleophilicity of the 7-AVCA amino group without inducing excessive basicity that could trigger beta-lactam hydrolysis. The reaction kinetics are optimized by maintaining a molar ratio of tri-n-butylamine to 7-AVCA between 2:1 and 3:1, ensuring complete conversion of the starting material while minimizing side reactions. The dichloromethane solvent provides an ideal medium for solubilizing both the polar amino acid substrate and the lipophilic active ester, facilitating efficient molecular collisions. Crucially, the absence of protic solvents or strong acids during this stage prevents premature cleavage of the trityl protecting group, ensuring that the Z-isomer configuration of the oxime side chain is retained throughout the coupling process. This stereochemical fidelity is paramount for the biological activity of the final antibiotic, as the E-isomer possesses significantly reduced potency.
Following the acylation, the purification mechanism employs a sophisticated acid-base manipulation to isolate the intermediate. By introducing phosphoric acid into the acetonitrile solution of the crude ester, the process selectively precipitates the Cefdinir trityl ester as a phosphate salt. This step is mechanistically distinct from simple salting out; it exploits the specific solubility differences between the desired phosphate salt and neutral organic impurities or unreacted active ester by-products. The phosphate salt formation acts as a rigorous filtering event, trapping the target molecule in a solid lattice while leaving contaminants in the supernatant. Subsequent hydrolysis of this purified salt using a weak base like sodium bicarbonate at controlled temperatures of 10°C to 20°C ensures gentle removal of the trityl group. The final gradient crystallization, where pH is carefully adjusted from 3.5-4.0 down to 2.4-2.5, allows for the controlled growth of crystals, preventing the inclusion of mother liquor impurities and yielding a product with a defined particle size distribution suitable for downstream formulation.
How to Synthesize Cefdinir Efficiently
The implementation of this synthesis route requires strict adherence to the optimized parameters regarding solvent ratios and temperature controls to maximize yield and purity. The process begins with the preparation of the reaction mixture in a dry environment to prevent hydrolysis of the active ester, followed by a controlled addition of reagents to manage exotherms. Detailed standard operating procedures for the acylation, phosphate precipitation, and gradient crystallization steps are essential for reproducibility. For a comprehensive breakdown of the specific reagent quantities, stirring speeds, and drying conditions required to achieve the reported 99% purity, please refer to the standardized protocol below.
- React 7-AVCA with the active benzothiazole ester in dichloromethane using tri-n-butylamine as a base at 20-30°C.
- Remove solvent to obtain crude ester, then dissolve in acetonitrile and precipitate as a phosphate salt using phosphoric acid.
- Hydrolyze the phosphate salt in weak base, decolorize, and perform acid gradient crystallization to isolate pure Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this patented methodology offers profound advantages that extend beyond mere technical feasibility. The elimination of hazardous anti-solvents like diethyl ether drastically reduces the regulatory burden associated with storage and handling of flammable materials, thereby lowering insurance premiums and facility compliance costs. Furthermore, the ability to recycle dichloromethane and the reduced need for complex solvent exchange operations translate into significant reductions in raw material consumption and waste disposal fees. The robustness of the process, characterized by its tolerance to mild temperature fluctuations and its high selectivity, ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply lines to downstream API manufacturers. By mitigating the risks of equipment corrosion and thermal degradation, this method enhances the longevity of capital assets and reduces unplanned downtime, securing a more reliable flow of high-purity intermediates to the market.
- Cost Reduction in Manufacturing: The process achieves substantial cost optimization by eliminating the need for expensive and hazardous anti-solvents such as ether, which also removes the associated costs of specialized explosion-proof infrastructure. The high molar yield of over 80% significantly improves material efficiency, reducing the cost per kilogram of the final active pharmaceutical ingredient. Additionally, the simplified workup procedure, which avoids complex distillation steps for acid removal, lowers energy consumption and labor hours required for production.
- Enhanced Supply Chain Reliability: By utilizing readily available solvents like dichloromethane and tri-n-butylamine, the method reduces dependency on specialty chemicals that may face supply volatility. The mild reaction conditions minimize the risk of batch failures due to thermal runaway or equipment corrosion, ensuring a steady and predictable output of Cefdinir intermediates. This stability allows procurement managers to forecast inventory needs with greater accuracy and maintain leaner safety stocks without compromising production schedules.
- Scalability and Environmental Compliance: The absence of toxic aromatic solvents and the reduction in hazardous waste generation align perfectly with increasingly stringent global environmental regulations. The process is inherently scalable, as the removal of anti-solvent crystallization steps prevents the formation of difficult-to-filter agglomerates that often plague kilogram-to-ton scale-ups. This facilitates a smoother technology transfer from pilot plant to commercial manufacturing, accelerating time-to-market for generic versions of the antibiotic.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Cefdinir synthesis route. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on process robustness and quality control measures. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: Why is the tri-n-butylamine/DCM system superior to traditional tosic acid methods?
A: The tri-n-butylamine system operates under milder conditions (20-30°C) and eliminates the need for hazardous anti-solvents like ether, significantly improving process safety and environmental compliance compared to corrosive tosic acid routes.
Q: How does the phosphate salt purification step enhance product quality?
A: Converting the crude ester into a phosphate salt allows for highly selective precipitation, effectively separating unreacted starting materials and by-products before the final deprotection step, ensuring purity levels exceed 99%.
Q: What are the scalability benefits of avoiding anti-solvents in this synthesis?
A: Eliminating anti-solvents removes the risk of uncontrolled coprecipitation and agglomeration, leading to consistent crystal morphology and simplified downstream processing, which is critical for reliable commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103319503A are fully realized in practical, industrial settings. We are committed to delivering high-purity Cefdinir intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to optimize reaction parameters and manage complex purification sequences allows us to offer a product profile that consistently exceeds industry standards for impurity control and physical properties.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this superior manufacturing technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this active ester-based route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a secure and cost-effective supply of this vital antibiotic intermediate.
