Optimizing Blonanserin Intermediate Production: A Technical Breakthrough for Commercial Scale-Up
Optimizing Blonanserin Intermediate Production: A Technical Breakthrough for Commercial Scale-Up
The pharmaceutical landscape for atypical antipsychotics continues to evolve, driven by the demand for more efficient and scalable synthetic routes for key active ingredients. A pivotal development in this domain is detailed in patent CN101747273B, which discloses a superior preparation method for a critical Blonanserin intermediate, specifically 4-(4-fluorophenyl)-5,6,7,8,9,10-hexahydrocyclooctano[b]pyridin-2(1H)-one. This compound serves as the foundational scaffold for Blonanserin, a potent dopamine-serotonin antagonist used in the treatment of schizophrenia. The structural integrity and purity of this intermediate are paramount, as they directly influence the efficacy and safety profile of the final API. As depicted in the chemical structure below, the molecule features a fused cyclooctane-pyridone system with a para-fluorophenyl substituent, presenting unique challenges in ring closure and functional group tolerance during synthesis.
![Chemical structure of 4-(4-fluorophenyl)-5,6,7,8,9,10-hexahydrocyclooctano[b]pyridin-2(1H)-one](/insights/img/blonanserin-intermediate-synthesis-pharma-supplier-20260308181935-01.png)
For R&D directors and process chemists, the significance of this patent lies in its ability to overcome the severe yield limitations associated with traditional methodologies. Historically, the construction of this fused heterocyclic system has been plagued by low conversion rates and difficult purification protocols. The innovation presented in CN101747273B shifts the paradigm from a direct, harsh one-pot cyclization to a controlled, two-step sequence. This strategic modification not only enhances the overall throughput but also simplifies the downstream processing, making it an attractive candidate for reliable pharmaceutical intermediates supplier networks aiming to secure stable supply chains for neurology drug portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, such as those described in JP4099768A and EP0385237, typically rely on a direct condensation reaction between 3-(4-fluorophenyl)-3-oxopropionitrile and cyclooctanone in the presence of polyphosphoric acid (PPA). While theoretically straightforward, this one-step approach suffers from significant practical drawbacks when implemented on an industrial scale. The reaction conditions are notoriously harsh, often requiring temperatures around 120°C, which can promote thermal degradation and uncontrolled polymerization of the reactive nitrile species. Furthermore, the use of large excesses of PPA creates a highly viscous reaction medium that complicates mixing and heat transfer, leading to inconsistent batch quality.
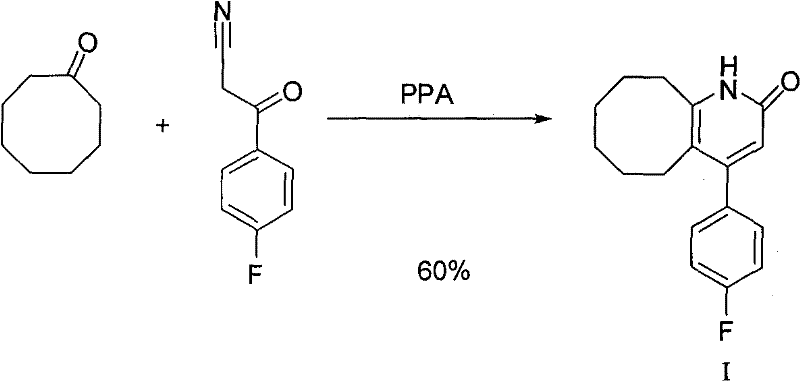
Most critically, the yield reported in literature for this direct method is often optimistic, with practical implementations yielding merely about 17% of the desired product. This discrepancy highlights the presence of substantial side reactions, likely involving the hydrolysis of the nitrile group to carboxylic acids or the formation of tarry byproducts that are difficult to separate. The generation of acidic impurities necessitates rigorous purification steps, often involving multiple recrystallizations or column chromatography, which drastically erodes the final mass balance and increases the cost of goods sold. For procurement managers, such inefficiencies translate into higher raw material consumption and unpredictable lead times, rendering the conventional route economically unviable for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN101747273B introduces a decoupled synthetic strategy that isolates the formation of the amide bond from the cyclization event. By first converting 3-(4-fluorophenyl)-3-oxopropionitrile into 3-(4-fluorophenyl)-3-oxopropionamide (Intermediate II) as a discrete step, the process gains a crucial handle on reaction selectivity. This intermediate is then subjected to cyclization with cyclooctanone under milder, optimized conditions. The visual representation of this improved pathway clearly demonstrates the sequential nature of the transformation, avoiding the chaotic environment of the one-pot PPA method.
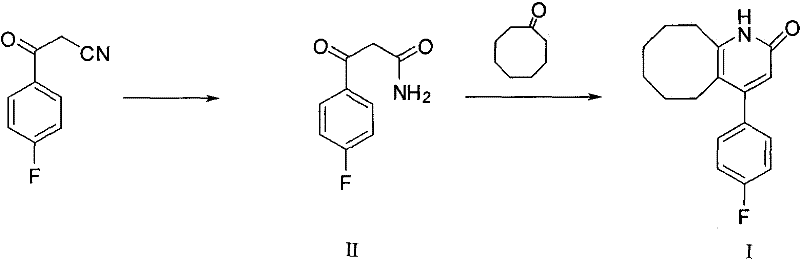
The impact of this procedural change is profound, with yields soaring from the dismal 17% baseline to approximately 80% in optimized embodiments. This five-fold increase in efficiency is achieved by utilizing specific dehydrating agents like p-toluenesulfonic acid in aromatic solvents such as toluene, rather than relying solely on the brute force of polyphosphoric acid. The separation of steps allows for the removal of impurities after the first stage, ensuring that the cyclization proceeds with a cleaner substrate. Consequently, the final product requires only primary crystallization to achieve high purity standards, eliminating the need for resource-intensive recrystallization cycles and positioning this method as a robust solution for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Stepwise Cyclization and Amide Formation
The success of this novel route hinges on the precise control of nucleophilic attack and dehydration kinetics during the second step. In the initial phase, the nitrile group undergoes partial hydrolysis to form the primary amide. This transformation is facilitated by polyphosphoric acid or boron trifluoride acetic acid complexes at moderate temperatures ranging from 60°C to 130°C. The formation of the amide is critical because it acts as a stabilized nucleophile in the subsequent condensation. Unlike the nitrile, which is prone to over-hydrolysis to the carboxylic acid under strong acidic conditions, the amide retains sufficient reactivity to attack the ketone carbonyl of cyclooctanone while resisting further degradation. This selectivity is the cornerstone of the yield improvement, as it prevents the loss of material to acid byproducts that plague the direct nitrile route.
Following the isolation of the amide, the cyclization mechanism involves an acid-catalyzed condensation where the amide nitrogen attacks the electrophilic carbon of the cyclooctanone. The use of p-toluenesulfonic acid (p-TsOH) in toluene creates an ideal environment for this dehydration reaction. Toluene serves a dual purpose: it acts as a solvent to dissolve the organic reactants and facilitates the azeotropic removal of water generated during the ring closure. The removal of water drives the equilibrium towards the product side according to Le Chatelier's principle, ensuring high conversion rates. Furthermore, the milder acidity of p-TsOH compared to concentrated PPA minimizes the risk of ring-opening side reactions or polymerization of the cyclooctanone ring, thereby preserving the structural integrity of the eight-membered ring fusion.
How to Synthesize 4-(4-fluorophenyl)-5,6,7,8,9,10-hexahydrocyclooctano[b]pyridin-2(1H)-one Efficiently
Implementing this high-yield protocol requires careful attention to reaction parameters and workup procedures to maximize the benefits of the two-step design. The process begins with the controlled hydrolysis of the nitrile starting material, followed by a distinct cyclization phase that leverages azeotropic distillation. Operators must ensure strict temperature control during the amide formation to prevent over-hydrolysis, and subsequently maintain reflux conditions in toluene to drive the cyclization to completion. The detailed standardized synthesis steps, including specific molar ratios and workup instructions derived from the patent examples, are provided in the guide below to assist technical teams in replicating these results.
- Hydrolyze 3-(4-fluorophenyl)-3-oxopropionitrile using polyphosphoric acid at 60-130°C to form the amide intermediate.
- React the resulting amide with cyclooctanone in toluene using p-toluenesulfonic acid as a dehydrating agent at 90-140°C.
- Purify the final product via simple crystallization to achieve high purity without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers substantial strategic advantages for organizations managing the supply chain of psychiatric medications. The primary benefit stems from the dramatic improvement in process mass intensity; by boosting yields from roughly 17% to nearly 80%, the consumption of key raw materials such as 3-(4-fluorophenyl)-3-oxopropionitrile and cyclooctanone is significantly reduced. This efficiency gain directly translates to lower variable costs per kilogram of the intermediate, providing a buffer against fluctuations in raw material pricing. Additionally, the elimination of complex purification steps reduces the demand for solvents and energy, further contributing to cost reduction in API manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The shift to a two-step process eliminates the need for expensive and hazardous reagents in excessive quantities. By avoiding the heavy reliance on polyphosphoric acid for the entire sequence, the process reduces the burden on waste treatment facilities and lowers the cost associated with neutralizing large volumes of acidic waste. The use of toluene, a commodity solvent with a well-established recovery infrastructure, allows for efficient recycling, minimizing solvent purchase costs. Furthermore, the high yield means that less starting material is required to produce the same amount of final product, effectively stretching the purchasing power of the procurement budget and enhancing overall margin potential.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding production delays. The simplified purification protocol, which relies on simple crystallization rather than multiple recrystallizations or chromatography, shortens the cycle time for each batch. This reduction in processing time allows manufacturers to respond more agilely to market demand, reducing lead time for high-purity pharmaceutical intermediates. Moreover, the reagents used, such as p-toluenesulfonic acid and toluene, are widely available globally, mitigating the risk of supply disruptions that might occur with more specialized or niche catalysts.
- Scalability and Environmental Compliance: Scaling this process from laboratory to pilot and eventually to commercial production is facilitated by the use of standard unit operations such as reflux and crystallization. The reaction conditions, particularly the temperature range of 90°C to 140°C, are well within the operating limits of standard glass-lined or stainless steel reactors, removing the need for specialized high-pressure or cryogenic equipment. From an environmental standpoint, the reduction in acidic waste and the ability to recycle toluene align with green chemistry principles, helping companies meet increasingly stringent environmental regulations. This sustainability profile enhances the corporate image and ensures long-term operational viability in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in patent CN101747273B, offering clarity on yield expectations, solvent selection, and purification strategies. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for large-scale production.
Q: How does the new two-step method improve yield compared to prior art?
A: The novel method increases yield from approximately 17% in conventional one-step processes to about 80% by isolating the amide intermediate, thereby reducing side reactions and impurity formation.
Q: What solvents are recommended for the cyclization step?
A: Toluene is the preferred solvent due to its high boiling point which facilitates the reaction temperature of 110°C, low toxicity profile, and cost-effectiveness compared to benzene or xylene.
Q: Is recrystallization necessary for purification?
A: No, the process generates fewer acidic impurities, allowing for purification via simple primary crystallization, which streamlines the workflow and reduces processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Blonanserin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to implement advanced synthetic routes, such as the high-yield cyclization method discussed, underscores our dedication to providing value-driven solutions for our global partners.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical foundation available.
