Advanced Isochroman Synthesis via Organoselenium Catalysis for Commercial Scale-Up
Advanced Isochroman Synthesis via Organoselenium Catalysis for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes for heterocyclic scaffolds that balance efficiency with regulatory compliance. Patent CN112794837B introduces a groundbreaking methodology for the synthesis of isochroman compounds, a privileged structure found in numerous bioactive natural products and drug candidates. This technology leverages a novel organoselenium reagent, N,N,N-triphenylselenoisocyanurate (TPSCA), to facilitate an exo-type cyclization under mild Lewis acid catalysis. Unlike traditional methods that rely on harsh conditions or expensive transition metals, this approach utilizes a recyclable selenium cycle to construct the isochroman core with high regioselectivity. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for complex pharmaceutical intermediates while adhering to stringent purity specifications.
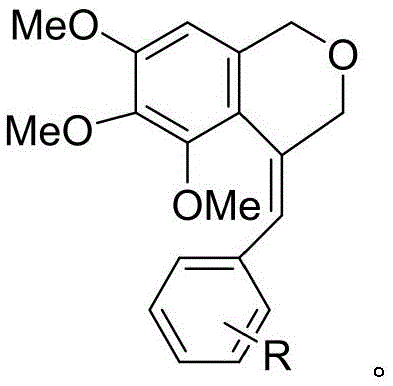
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isochroman ring system has relied heavily on palladium-catalyzed carbon-iodination or acid-catalyzed cyclizations using strong protic acids like hydrogen chloride. These conventional pathways often suffer from significant drawbacks, including the requirement for stoichiometric amounts of expensive noble metals, which complicates downstream purification and raises concerns about residual heavy metals in the final API. Furthermore, methods utilizing strong mineral acids frequently necessitate high temperatures, leading to potential decomposition of sensitive functional groups and the formation of complex impurity profiles that are difficult to separate. The environmental footprint of these legacy processes is also substantial, generating large volumes of acidic waste and requiring specialized equipment to handle corrosive reagents safely.
The Novel Approach
The methodology disclosed in CN112794837B circumvents these issues by employing a unique electrophilic selenium species generated in situ. By utilizing TPSCA in conjunction with boron trifluoride etherate, the reaction proceeds through a highly selective selenonium ion intermediate that directs the cyclization exclusively towards the exo-product. This mechanistic pathway operates at moderate temperatures, typically between 55°C and 70°C, preserving the integrity of delicate substituents such as methoxy groups. The process eliminates the need for palladium entirely, thereby removing the costly and time-consuming step of heavy metal scavenging. Additionally, the selenium byproduct, diphenyl diselenide, is not waste but a valuable precursor that can be recycled back into the reagent synthesis loop, creating a closed-loop system that enhances overall sustainability.
Mechanistic Insights into TPSCA-Mediated Cyclization
The core innovation lies in the generation and reactivity of the N,N,N-triphenylselenoisocyanurate species. Upon activation by the Lewis acid catalyst BF3·Et2O, the selenium atom becomes highly electrophilic, attacking the electron-rich double bond of the cinnamyl ether substrate to form a cyclic selenonium ion. This intermediate is pivotal because it locks the conformation of the molecule, forcing the nucleophilic attack by the electron-rich 3,4,5-trimethoxybenzene ring to occur at the specific beta-position. This steric control ensures that only the exo-type cyclization product is formed, effectively suppressing the formation of endo-isomers or other regioisomeric byproducts that typically plague Friedel-Crafts type alkylations.
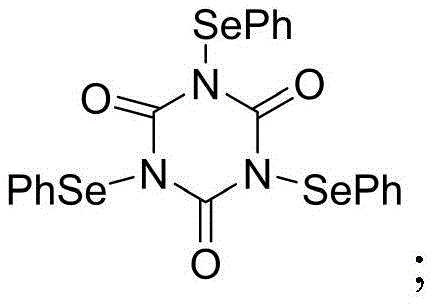
Furthermore, the reaction mechanism includes an automatic elimination step where the selenium functionality is expelled as diphenyl diselenide, restoring the aromaticity of the system and yielding the final isochroman product without retaining any selenium atoms in the final structure. This "traceless" aspect is crucial for pharmaceutical applications, as it ensures the final intermediate is free from organoselenium impurities. The ability to tune the electronic properties of the starting cinnamyl alcohol allows for the synthesis of a diverse library of derivatives, including those with halogen or alkyl substitutions, all while maintaining high yields ranging from 60% to 95% as demonstrated in the patent examples.
How to Synthesize Isochroman Compounds Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and scalability, making it ideal for transfer from laboratory to pilot plant. The process begins with the preparation of the key electrophile, 1-bromomethyl-3,4,5-trimethoxybenzene, followed by its coupling with various substituted cinnamyl alcohols to form the ether precursor. The critical cyclization step involves mixing this precursor with the pre-synthesized TPSCA reagent in anhydrous dichloromethane. Detailed standard operating procedures regarding stoichiometry, temperature gradients, and workup protocols are essential for reproducibility. For a comprehensive breakdown of the exact molar ratios and quenching procedures required to achieve optimal purity, please refer to the standardized synthesis guide below.
- Preparation of 1-bromomethyl-3,4,5-trimethoxybenzene from 3,4,5-trimethoxybenzyl alcohol using PBr3.
- Etherification of substituted cinnamyl alcohol with the bromide intermediate using NaH in THF.
- Synthesis of N,N,N-triphenylselenoisocyanurate (TPSCA) from silver isocyanurate and phenylselenobromide.
- Cyclization reaction using TPSCA and BF3·Et2O in dichloromethane under reflux to form the isochroman ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling advantages for supply chain stability and cost management. The reliance on readily available starting materials such as 3,4,5-trimethoxybenzaldehyde and substituted cinnamic acids ensures a robust supply base that is not subject to the geopolitical volatility often associated with rare earth or precious metal catalysts. The elimination of palladium not only reduces the direct cost of goods sold but also significantly lowers the capital expenditure required for wastewater treatment and metal recovery systems. This streamlined process translates to faster batch turnover times and reduced inventory holding costs for manufacturers.
- Cost Reduction in Manufacturing: The ability to recycle the selenium byproduct diphenyl diselenide back into phenylselenobromide creates a circular economy within the production facility, drastically reducing the consumption of fresh selenium reagents. This internal recycling loop minimizes raw material procurement costs and mitigates the price volatility of selenium commodities. Furthermore, the mild reaction conditions reduce energy consumption compared to high-temperature acid catalysis methods, contributing to lower utility overheads per kilogram of product produced.
- Enhanced Supply Chain Reliability: By avoiding the use of specialized palladium catalysts which often have long lead times and single-source suppliers, manufacturers can diversify their vendor base for common organic chemicals. The robustness of the reaction against moisture and oxygen variations, provided standard inert atmosphere protocols are followed, ensures consistent batch-to-batch quality. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream API manufacturers.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of recoverable organic solvents and recyclable selenium salts, simplifying compliance with increasingly strict environmental regulations. The absence of heavy metal residues in the crude product simplifies the purification workflow, allowing for larger batch sizes without proportional increases in purification complexity. This scalability ensures that the technology can seamlessly transition from kilogram-scale development to multi-ton commercial production without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium-mediated synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on feasibility and performance metrics for potential adopters.
Q: What are the advantages of using TPSCA over traditional Palladium catalysts for isochroman synthesis?
A: TPSCA offers a metal-free alternative to expensive Pd(0) catalysts, reducing heavy metal contamination risks and simplifying purification processes for pharmaceutical intermediates.
Q: Is the organoselenium reagent recyclable in this process?
A: Yes, the byproduct diphenyl diselenide can be recovered and converted back into phenylselenobromide, significantly improving atom economy and reducing raw material costs.
Q: What is the typical yield range for this selenium-mediated cyclization?
A: According to patent data, the process achieves yields ranging from 60% to 95%, depending on the substituents on the cinnamyl alcohol moiety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochroman Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic methodologies like the TPSCA-mediated cyclization for producing high-value pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of isochroman derivative meets the exacting standards required for global pharmaceutical supply chains.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your project's timeline and budget efficiency.
