Advanced Selenium-Mediated Cyclization for High-Purity Isochroman Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN112794837A introduces a groundbreaking methodology for the synthesis of isochroman compounds, a privileged structure found in numerous bioactive natural products such as the antibiotic citrinin. This patent discloses a novel electrophilic selenium-mediated cyclization strategy that fundamentally shifts the paradigm from traditional transition-metal catalysis to a more sustainable and cost-effective organic selenium reagent system. By utilizing N,N,N-triphenylseleno isocyanurate (TPSCA) in conjunction with a Lewis acid catalyst, the process achieves high regioselectivity and yield under remarkably mild thermal conditions. This technical breakthrough addresses long-standing challenges in impurity control and process scalability, positioning this route as a highly attractive option for the commercial scale-up of complex heterocyclic intermediates. The versatility of the method allows for various substituents on the aromatic ring, making it a universal platform for generating diverse libraries of bioactive molecules.
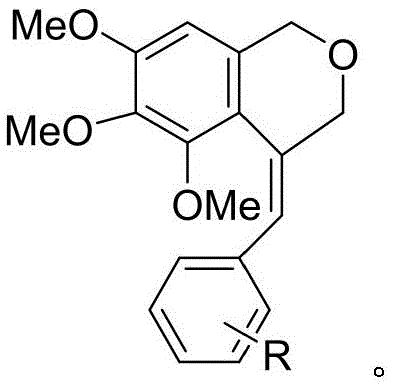
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isochroman core has relied heavily on palladium-catalyzed carbonylation or acid-catalyzed condensation reactions, both of which present significant drawbacks for large-scale manufacturing. Conventional palladium-mediated routes often require stringent anhydrous conditions and expensive ligands, leading to inflated production costs and supply chain vulnerabilities associated with precious metal availability. Furthermore, the removal of trace palladium residues to meet pharmaceutical purity standards necessitates additional purification steps, such as scavenger treatments or extensive chromatography, which drastically reduce overall process efficiency. Alternative acid-catalyzed methods using strong protic acids often suffer from harsh reaction conditions that can degrade sensitive functional groups, resulting in complex impurity profiles and lower isolated yields. These legacy processes frequently generate substantial amounts of hazardous waste, complicating environmental compliance and increasing the burden on waste management infrastructure.
The Novel Approach
In stark contrast, the methodology outlined in CN112794837A employs a unique organoselenium reagent, N,N,N-triphenylseleno isocyanurate (TPSCA), to drive the cyclization through an electrophilic activation mechanism. This approach operates under mild thermal conditions, typically around 60°C, which preserves the integrity of sensitive substituents and minimizes thermal degradation byproducts. The use of TPSCA facilitates a highly specific exo-type cyclization, effectively suppressing the formation of regioisomeric impurities that are common in non-selective acid catalysis. Moreover, the reaction generates diphenyl diselenide as a stoichiometric byproduct, which is not merely waste but a valuable precursor that can be recycled back into the synthesis of the TPSCA reagent. This closed-loop characteristic significantly enhances the atom economy of the process and aligns with green chemistry principles, offering a compelling value proposition for cost reduction in pharmaceutical manufacturing.
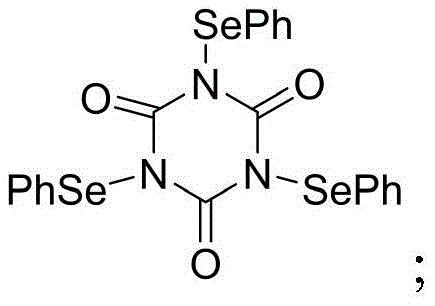
Mechanistic Insights into Electrophilic Selenium-Mediated Cyclization
The core innovation of this synthesis lies in the generation of a reactive selenonium ion intermediate upon the interaction of TPSCA with the alkene moiety of the cinnamyl ether substrate in the presence of boron trifluoride etherate. This Lewis acid activation enhances the electrophilicity of the selenium species, prompting an intramolecular attack by the electron-rich aromatic ring of the trimethoxybenzene segment. The mechanistic pathway is meticulously controlled by steric factors; the bulky phenylseleno group at the 2-position of the intermediate directs the nucleophilic attack to the less hindered beta-position of the aromatic ring. This steric guidance ensures exclusive formation of the exo-cyclized product, avoiding the thermodynamic trap of endo-cyclization that plagues other methods. The subsequent elimination of the selenium functionality occurs spontaneously or under the reaction conditions, releasing the final isochroman scaffold and regenerating the diselenide byproduct.
From an impurity control perspective, this mechanism offers superior predictability compared to radical-based or high-temperature acid catalysis. The specificity of the selenonium ion formation means that side reactions such as polymerization of the alkene or over-alkylation of the aromatic ring are kinetically disfavored. The mild reaction window prevents the decomposition of the methoxy groups, which are prone to demethylation under harsh acidic conditions. Consequently, the crude reaction profile is significantly cleaner, reducing the load on downstream purification units. For R&D teams, understanding this mechanism allows for precise tuning of reaction parameters, such as the molar ratio of TPSCA to substrate and the choice of solvent, to maximize throughput while maintaining high-purity isochroman compounds suitable for direct use in subsequent API synthesis steps.
How to Synthesize Isochroman Compounds Efficiently
The synthesis protocol described in the patent provides a clear, three-stage workflow that transitions from simple starting materials to the complex heterocyclic target. The process begins with the preparation of the key ether linkage between the trimethoxybenzyl fragment and the cinnamyl alcohol derivative, followed by the critical selenium-mediated cyclization step. Detailed operational parameters, including specific temperature ramps, quenching procedures, and purification techniques, are essential for reproducing the high yields reported in the examples. The following guide summarizes the standardized synthesis steps derived from the patent data to assist process chemists in implementing this route.
- Preparation of 1-bromomethyl-3,4,5-trimethoxybenzene from 3,4,5-trimethoxybenzyl alcohol using phosphorus tribromide.
- Etherification with substituted cinnamyl alcohol using sodium hydride to form the cinnamyloxy methyl precursor.
- Cyclization using N,N,N-triphenylseleno isocyanurate (TPSCA) and boron trifluoride etherate under reflux to yield the target isochroman.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this selenium-mediated route offers tangible strategic benefits beyond mere technical feasibility. The elimination of palladium and other precious metals from the catalyst system immediately decouples production costs from the volatile pricing of noble metals, providing greater financial predictability for long-term contracts. Additionally, the ability to recycle the selenium byproduct back into the active reagent creates a self-sustaining material loop that drastically reduces the consumption of raw selenium sources. This efficiency translates directly into a lower cost of goods sold (COGS) and a reduced environmental footprint, which is increasingly critical for meeting corporate sustainability goals. The mild reaction conditions also imply lower energy consumption for heating and cooling, further contributing to overall operational expenditure savings.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with an organic selenium reagent system removes the necessity for costly metal scavenging resins and extensive analytical testing for residual metals. This simplification of the downstream processing train reduces both material costs and labor hours associated with purification. Furthermore, the high regioselectivity of the reaction minimizes the loss of valuable intermediates to isomeric byproducts, thereby improving the overall mass balance and yield of the process. These factors combine to deliver substantial cost savings without compromising the quality of the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted cinnamyl alcohols and trimethoxybenzyl derivatives, are commodity chemicals with robust global supply chains. Unlike specialized ligands or rare earth catalysts that may face geopolitical supply risks, these precursors are readily available from multiple vendors. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in utility quality or environmental conditions, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production schedules and reducing lead time for high-purity API precursors.
- Scalability and Environmental Compliance: The process operates at near-atmospheric pressure and moderate temperatures, making it inherently safer and easier to scale from kilogram to multi-ton quantities. The absence of highly corrosive strong acids or toxic heavy metals simplifies waste treatment protocols and reduces the regulatory burden associated with hazardous waste disposal. The potential for recycling the diphenyl diselenide byproduct further aligns the process with circular economy principles, enhancing the company's environmental, social, and governance (ESG) profile. This scalability ensures that the method can support commercial demands from clinical trial phases through to full-scale market launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium-mediated synthesis route. These answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is vital for project managers evaluating the feasibility of integrating this technology into existing manufacturing pipelines.
Q: What is the primary advantage of using TPSCA over traditional palladium catalysts?
A: TPSCA eliminates the need for expensive noble metals like palladium, significantly reducing raw material costs and removing the complex purification steps required to lower residual metal content in pharmaceutical grades.
Q: Can the selenium byproducts be recycled in this process?
A: Yes, the process generates diphenyl diselenide as a byproduct, which serves as the direct starting material for regenerating the TPSCA reagent, creating a closed-loop system that minimizes waste.
Q: What represents the critical control point for regioselectivity in this synthesis?
A: The reaction temperature and the specific steric environment created by the TPSCA intermediate ensure exclusive exo-type cyclization, preventing the formation of unwanted endo-type isomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochroman Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the selenium-mediated cyclization technology described in CN112794837A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into a robust industrial process. Our facilities are equipped to handle complex organic synthesis involving sensitive reagents, ensuring that the strict stoichiometric controls and inert atmosphere requirements of the TPSCA reaction are meticulously maintained. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with unwavering consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for isochroman-based therapeutics. Our team can provide a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this novel selenium route compared to your current legacy processes. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your specific target molecule. Let us help you secure a competitive advantage through superior chemistry and reliable manufacturing execution.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
