Revolutionizing Asymmetric Synthesis with Recyclable Polyionic Liquid Chiral Copper Catalysts
Revolutionizing Asymmetric Synthesis with Recyclable Polyionic Liquid Chiral Copper Catalysts
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes in the pharmaceutical industry. Patent CN111285768B, published in September 2022, introduces a groundbreaking class of polyionic liquid chiral amino acid copper catalysts, designated as P(IL-A)n-Cu. This technology addresses long-standing challenges in aqueous-phase catalysis, specifically targeting the asymmetric Henry reaction, a pivotal transformation for generating chiral beta-nitroalcohols which are essential precursors for various bioactive molecules. Unlike traditional small-molecule catalysts that struggle with solubility and recovery in water, this novel polymeric architecture leverages the unique properties of ionic liquids to create a robust, water-soluble, and highly recyclable catalytic system. For R&D directors and process chemists, this represents a paradigm shift towards sustainable manufacturing without compromising on enantioselectivity or yield.
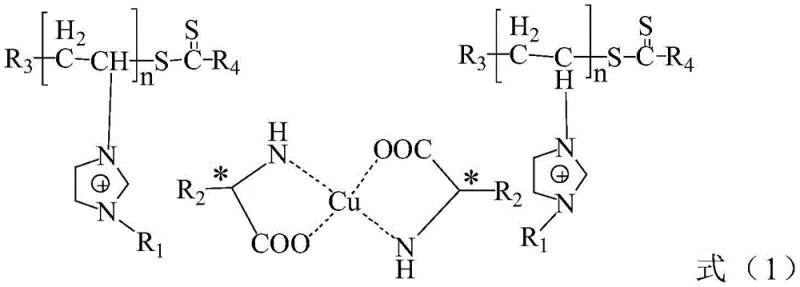
The core innovation lies in the structural design where chiral amino acids are covalently tethered to a polyionic liquid backbone. This design not only preserves the chiral environment necessary for high enantioselectivity but also imparts amphiphilic characteristics that enhance interaction with substrates in aqueous media. As a reliable pharmaceutical intermediate supplier, understanding such technological advancements is crucial for maintaining a competitive edge in the synthesis of high-purity chiral building blocks. The catalyst's ability to operate efficiently in pure water eliminates the environmental burden associated with volatile organic compounds (VOCs), aligning perfectly with modern green chemistry principles and regulatory pressures facing the fine chemical sector today.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric Henry reaction has been catalyzed by small-molecule metal-amino acid complexes. While effective in organic solvents, these traditional catalysts face severe limitations when applied to aqueous systems, which are preferred for their safety and environmental profile. The primary bottleneck is the poor solubility of hydrophobic amino acid ligands and their metal complexes in water, leading to heterogeneous reaction conditions that severely hinder mass transfer between the catalyst and the organic substrates. This often results in prolonged reaction times, inconsistent yields, and difficulty in controlling the stereochemical outcome. Furthermore, the recovery of these homogeneous catalysts from the reaction mixture is notoriously difficult, often requiring complex extraction procedures that generate significant chemical waste and lead to the loss of expensive chiral ligands and metals.
The Novel Approach
The methodology described in CN111285768B overcomes these hurdles by integrating the chiral ligand into a polyionic liquid matrix. By polymerizing ionic liquid monomers functionalized with chiral amino acids, the resulting catalyst exhibits exceptional solubility in water while maintaining a defined chiral pocket around the copper center. This homogeneity in the aqueous phase ensures intimate contact between the catalyst and substrates, dramatically accelerating reaction kinetics. Moreover, the polymeric nature of the catalyst introduces a unique recovery mechanism; post-reaction, the addition of a non-solvent or specific organic solvent induces phase separation, allowing the catalyst to precipitate or partition into a distinct layer for easy retrieval. This approach effectively combines the high activity of homogeneous catalysis with the ease of separation typical of heterogeneous systems, offering a superior solution for cost reduction in chiral catalyst manufacturing.
Mechanistic Insights into Polyionic Liquid Copper Coordination
The efficacy of the P(IL-A)n-Cu catalyst stems from a sophisticated interplay between polymer chemistry and coordination geometry. The synthesis begins with the quaternization of vinyl imidazole to form an ionic liquid monomer, which is subsequently neutralized with a chiral amino acid. This creates a zwitterionic or salt-like monomer that retains the polymerizable vinyl group. Through Reversible Addition-Fragmentation Chain Transfer (RAFT) polymerization, these monomers are assembled into a well-defined polymer chain with controlled molecular weight and narrow dispersity. This control is vital as it prevents the formation of insoluble cross-linked networks that would negate the solubility benefits. The resulting polyionic liquid acts as a multidentate ligand scaffold, presenting multiple amino acid coordination sites along the chain.
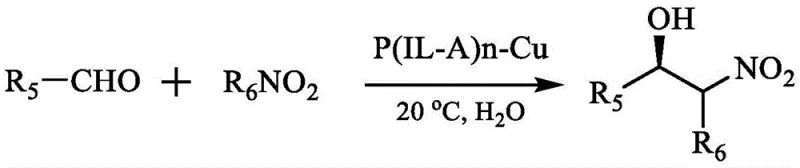
Upon introduction of a copper salt, such as copper acetate, the carboxylate and amine groups of the pendant amino acids coordinate with the copper ions to form the active catalytic species. In the context of the Henry reaction, this copper center activates the nitroalkane by deprotonation to form a copper-nitronate intermediate, while simultaneously coordinating the aldehyde substrate. The chiral environment provided by the amino acid side chains dictates the facial selectivity of the nucleophilic attack, ensuring high enantiomeric excess (ee). The ionic liquid moieties surrounding the active site create a localized microenvironment that stabilizes the transition state and enhances the solubility of organic substrates within the aqueous phase, effectively acting as a nanoreactor. This mechanistic advantage translates directly to the observed high yields and ee values exceeding 90% for a wide range of aromatic aldehydes, demonstrating the robustness of this catalytic platform for producing high-purity pharmaceutical intermediates.
How to Synthesize P(IL-A)n-Cu Efficiently
The preparation of this advanced catalyst follows a modular four-step sequence that is amenable to scale-up. It starts with the synthesis of the double-bonded ionic liquid precursor, followed by neutralization with the chosen chiral amino acid to install the stereocenter. The third step involves the RAFT polymerization to build the polymeric backbone, and finally, coordination with copper salt yields the active catalyst. This structured approach ensures reproducibility and quality control at every stage. For detailed operational parameters and specific stoichiometric ratios required for laboratory or pilot-scale preparation, please refer to the standardized guide below.
- React vinyl imidazole with alkyl halides followed by anion exchange resin treatment to synthesize double-bonded ionic liquid monomers.
- Neutralize the ionic liquid monomers with chiral amino acids to form double-bonded ionic liquid chiral amino acid salts.
- Perform controlled radical polymerization (RAFT) using AIBN and chain transfer agents to generate the polyionic liquid amino acid polymer.
- Coordinate the polymer with copper salts (e.g., copper acetate) in organic solvents to finalize the P(IL-A)n-Cu catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the P(IL-A)n-Cu catalyst technology offers compelling strategic advantages beyond mere technical performance. The shift towards aqueous-phase catalysis fundamentally alters the cost structure of chiral intermediate production by removing the dependency on large volumes of hazardous organic solvents. This transition not only reduces raw material costs but also significantly lowers the expenses associated with solvent recovery, waste treatment, and regulatory compliance. The ability to recycle the catalyst multiple times without substantial loss of activity means that the effective cost of the chiral ligand and metal per kilogram of product is drastically reduced, enhancing the overall economic viability of the process.
- Cost Reduction in Manufacturing: The implementation of this water-based catalytic system eliminates the need for expensive anhydrous organic solvents and the energy-intensive distillation processes required for their recovery. By utilizing water as the primary medium, facilities can achieve substantial cost savings in utility consumption and waste disposal fees. Furthermore, the high turnover number and reusability of the polyionic liquid catalyst mean that the loading of expensive chiral amino acids and copper can be minimized over multiple batches, driving down the variable cost of goods sold for high-value chiral intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for synthesizing this catalyst, including imidazoles, alkyl halides, and natural amino acids, are commodity chemicals with stable and abundant global supply chains. This reduces the risk of supply disruptions compared to specialized, proprietary ligands that may have single-source dependencies. Additionally, the simplified work-up procedure involving phase separation streamlines the manufacturing timeline, potentially reducing lead times for high-purity pharmaceutical intermediates and allowing for more responsive production scheduling to meet market demand fluctuations.
- Scalability and Environmental Compliance: The RAFT polymerization technique used to create the catalyst backbone is a well-established industrial process that scales linearly from gram to ton quantities without losing control over polymer properties. This ensures that the catalyst quality remains consistent during commercial scale-up of complex chiral catalysts. From an environmental perspective, the near-zero discharge of organic solvents and the minimization of heavy metal waste align with stringent global environmental regulations, future-proofing the manufacturing process against tightening ecological standards and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of polyionic liquid catalysts in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making.
Q: How does this polyionic liquid catalyst improve upon traditional copper-amino acid complexes?
A: Traditional copper-amino acid complexes often suffer from poor solubility in aqueous media and difficult recovery. This polyionic liquid variant introduces hydrophilic ionic groups that ensure excellent water solubility, facilitating mass transfer and allowing for simple phase-separation recovery after the reaction.
Q: What represents the primary cost advantage of this catalytic system?
A: The primary cost advantage lies in the use of water as the sole reaction solvent, eliminating the need for expensive and toxic organic solvents. Additionally, the catalyst's ability to be recovered and reused multiple times without significant loss of activity drastically reduces the cost per batch for high-value chiral intermediates.
Q: Is this catalyst suitable for large-scale industrial production?
A: Yes, the synthesis utilizes mild conditions and commercially available raw materials like amino acids and imidazoles. The RAFT polymerization technique allows for controlled molecular weight distribution, ensuring consistent batch-to-bquality which is critical for scaling up complex chiral catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable P(IL-A)n-Cu Catalyst Supplier
The development of polyionic liquid chiral copper catalysts marks a significant milestone in the evolution of green asymmetric synthesis, offering a viable path towards sustainable manufacturing of complex chiral molecules. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of such technologies and have invested heavily in mastering the intricacies of polymer-supported catalysis and coordination chemistry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst or intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific synthesis needs. Whether you require custom synthesis of the P(IL-A)n-Cu catalyst itself or the application of this methodology to produce specific chiral beta-nitroalcohol intermediates, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current process. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this green catalytic solution can optimize your supply chain and enhance your product portfolio.
