Advanced Tin-Free Synthesis of Valsartan Intermediates for Commercial Scale-Up
Advanced Tin-Free Synthesis of Valsartan Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with environmental sustainability, particularly for blockbuster antihypertensive agents like Valsartan. Patent CN101270096A introduces a groundbreaking improvement in the synthesis of Valsartan by fundamentally eliminating the use of toxic organotin compounds, which have long plagued traditional production methods. This innovation addresses critical regulatory hurdles regarding heavy metal residues while simultaneously enhancing overall process yield and operational simplicity. By utilizing a condensate of N-[(2′-cyano-1,1′-biphenyl-4-yl)alkyl]-L-valine ester as the starting material, the process achieves a streamlined two-step reaction sequence involving valerylation and subsequent tetrazole ring formation. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, ensuring compliance with stringent ICH guidelines without compromising on production efficiency or cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Valsartan has relied heavily on the use of halogenated alkyl tin compounds as catalysts for the formation of the tetrazole ring, a critical structural motif in the active pharmaceutical ingredient. While effective in driving the reaction, these organotin reagents introduce severe downstream processing challenges, primarily due to their high toxicity and the extreme difficulty in removing residual tin from the final product to meet the rigorous ICH limit of less than 1 ppm. Conventional routes often require complex purification steps, specialized scavengers, or extensive chromatography to mitigate heavy metal contamination, which drastically increases manufacturing costs and extends production lead times. Furthermore, the disposal of tin-containing waste streams poses significant environmental liabilities, complicating waste management protocols for large-scale facilities. These factors collectively create a bottleneck for reliable API intermediate suppliers aiming to deliver cost-effective and environmentally compliant solutions to the global market.
The Novel Approach
In stark contrast to legacy technologies, the method disclosed in CN101270096A circumvents the need for any tin-based catalysts by employing a combination of metal azides and specific amine salts or Lewis acids to facilitate the cyclization reaction. This novel approach utilizes readily available raw materials such as sodium azide and triethylamine hydrochloride, which are not only more economical but also significantly safer to handle on a commercial scale. The process allows for the use of common aromatic solvents like toluene or p-chlorotoluene, which can be easily recovered and recycled, thereby reducing solvent consumption and waste generation. By simplifying the reaction workflow into two distinct yet efficient stages—valerylation followed by tetrazole synthesis—the new method achieves a total yield exceeding 75%, demonstrating superior atom economy. This shift represents a paradigm change in cost reduction in API manufacturing, offering a cleaner, faster, and more scalable alternative for producing complex antihypertensive agents.
Mechanistic Insights into Tin-Free Tetrazole Cyclization
The core chemical innovation lies in the replacement of the tin-catalyzed [2+3] cycloaddition with a thermally driven reaction mediated by metal azides and proton sources or Lewis acids. In the first stage, the starting condensate, N-[(2′-cyano-1,1′-biphenyl-4-yl)alkyl]-L-valine ester, undergoes N-acylation with valeryl chloride in the presence of a carbonate base at controlled temperatures between 0°C and 40°C. This step ensures the selective formation of the pentanoyl intermediate without racemization of the chiral center. Subsequently, the nitrile group on the biphenyl moiety reacts with the metal azide (such as sodium azide, potassium azide, or lithium azide) under reflux conditions ranging from 70°C to 150°C. The presence of an amine salt, such as triethylamine hydrochloride, or other Lewis acids like zinc chloride, acts as a crucial promoter, facilitating the nucleophilic attack of the azide on the nitrile carbon and stabilizing the transition state for tetrazole ring closure.
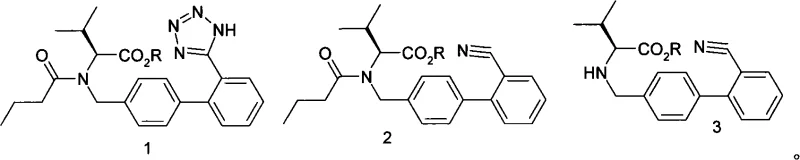
This mechanistic pathway effectively bypasses the formation of stable organotin byproducts, thereby inherently preventing heavy metal contamination in the crude reaction mixture. The absence of tin simplifies the workup procedure significantly; after the cyclization is complete, typically within 10 to 50 hours depending on the solvent system, the reaction mixture can be directly treated with an alkaline solution to hydrolyze the ester protecting group. Following phase separation and acidification to pH 1-2, the final Valsartan product precipitates with high purity. This robust mechanism ensures that impurity profiles are dominated by organic byproducts rather than inorganic toxins, making the purification process far more predictable and manageable for quality control laboratories. For technical teams, understanding this mechanism is vital for optimizing reaction parameters such as temperature and stoichiometry to maximize yield while maintaining the integrity of the chiral valine moiety.
How to Synthesize Valsartan Efficiently
Implementing this synthesis route requires precise control over reaction conditions to ensure optimal conversion and minimal byproduct formation. The process begins with the preparation of the pentanoyl intermediate by mixing the condensate hydrochloride with an aromatic solvent and an aqueous carbonate solution, followed by the dropwise addition of valeryl chloride while maintaining the temperature below 30°C. Once the valerylation is complete, the organic layer containing the intermediate can be carried forward directly or isolated as a solid. The subsequent cyclization step involves dissolving this intermediate in a suitable solvent, adding the metal azide and the chosen amine salt or Lewis acid, and heating the mixture to reflux. Detailed operational parameters, including specific molar ratios, stirring speeds, and crystallization protocols, are essential for reproducibility and are outlined in the standardized synthesis guide below.
- Perform valerylation of N-[(2′-cyano-1,1′-biphenyl-4-yl)alkyl]-L-valine ester using valeryl chloride and carbonate base in aromatic solvent at 0-40°C.
- React the resulting pentanoyl intermediate with metal azide (e.g., sodium azide) and a Lewis acid or amine salt (e.g., triethylamine hydrochloride).
- Heat the mixture to 70-150°C for reflux cyclization (10-50 hours), followed by alkaline hydrolysis and acidification to isolate high-purity Valsartan.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this tin-free methodology offers substantial advantages that extend beyond mere chemical elegance, directly impacting the bottom line and supply chain resilience. By eliminating the dependency on expensive and highly corrosive organotin reagents, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing through both raw material savings and reduced waste disposal fees. The ability to use common solvents like toluene, which are easily recyclable and widely available in the global chemical market, further enhances supply chain reliability by mitigating the risk of shortages associated with specialty reagents. Moreover, the simplified purification process reduces the number of unit operations required, leading to shorter batch cycles and increased throughput capacity for existing production facilities. These factors collectively contribute to a more agile and cost-efficient supply chain capable of meeting the demanding volume requirements of the generic pharmaceutical market.
- Cost Reduction in Manufacturing: The removal of organotin catalysts eliminates the need for costly heavy metal scavenging resins and complex purification steps, which traditionally account for a significant portion of production expenses. Additionally, the use of inexpensive inorganic salts like sodium azide and ammonium chloride instead of proprietary tin reagents drastically lowers the bill of materials. The high yield reported in the patent, exceeding 75%, ensures that raw material utilization is maximized, further driving down the cost per kilogram of the final API. This economic efficiency makes the process highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: Sourcing organotin compounds can be challenging due to their classification as hazardous substances and varying regulatory restrictions across different jurisdictions. By switching to universally available chemicals like carbonates, azides, and simple amine salts, the supply chain becomes far more robust and less susceptible to disruptions. The flexibility to use various aromatic solvents, including p-chlorotoluene which can shorten reaction times, provides procurement managers with multiple options to secure raw materials based on regional availability and pricing. This diversification of the supply base ensures continuous production capability and reduces lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reaction conditions and equipment that are common in fine chemical plants. The absence of toxic tin waste simplifies environmental compliance, reducing the burden on wastewater treatment facilities and lowering the risk of regulatory penalties. The ability to potentially run the reaction in a one-pot manner or with minimal solvent switching further enhances the green chemistry profile of the synthesis. This alignment with sustainable manufacturing practices not only meets current regulatory standards but also future-proofs the production facility against increasingly stringent environmental laws, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing portfolios or sourcing strategies.
Q: Why is the elimination of organotin catalysts critical in Valsartan production?
A: Traditional methods use toxic halogenated alkyl tin catalysts which leave residual heavy metals difficult to remove below ICH limits (1 ppm). This new method uses amine salts/Lewis acids, ensuring safer, compliant products.
Q: What yields can be expected from this improved synthesis route?
A: The patent data indicates a total yield greater than 75% calculated from the condensate, with specific examples demonstrating yields up to 78%, significantly improving process efficiency.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the method utilizes common aromatic solvents like toluene or p-chlorotoluene which are easy to recycle, and the reaction conditions (0-150°C) are compatible with standard industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN101270096A to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity Valsartan and its intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to verify the absence of heavy metal contaminants and ensure consistent quality batch after batch. Our infrastructure is specifically designed to handle complex organic syntheses involving hazardous reagents like azides with the utmost safety and precision.
We invite potential partners to engage with our technical procurement team to discuss how this innovative tin-free technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your antihypertensive drug production programs.
