Advanced Synthesis of Valsartan Intermediate via Salt-Effect Mediated Acylation for Commercial Scale
Advanced Synthesis of Valsartan Intermediate via Salt-Effect Mediated Acylation for Commercial Scale
The pharmaceutical industry continuously seeks robust and cost-effective pathways for the production of critical antihypertensive agents, with Valsartan standing as a cornerstone in cardiovascular therapy. A significant technological breakthrough in this domain is documented in patent CN110078640B, which details an innovative synthesis method for the key Valsartan intermediate, N-[(2'-cyanobiphenyl-4-yl)methyl]-N-(1-oxo-pentyl)-L-valine methyl ester. This patent introduces a novel approach that leverages the common ion effect to stabilize the reaction environment, specifically addressing the chronic issue of acid chloride hydrolysis in biphasic systems. By integrating specific inorganic salts into the reaction matrix, the process achieves exceptional control over impurity profiles and reagent consumption. For R&D directors and procurement specialists seeking a reliable valsartan intermediate supplier, understanding the mechanistic advantages of this patented route is essential for securing a stable and high-quality supply chain. The following analysis dissects the technical merits and commercial implications of this synthesis strategy.
![Chemical structure of N-[(2'-cyanobiphenyl-4-yl)methyl]-N-(1-oxo-pentyl)-L-valine methyl ester (Formula I)](/insights/img/valsartan-intermediate-synthesis-supplier-acylation-20260314195005-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
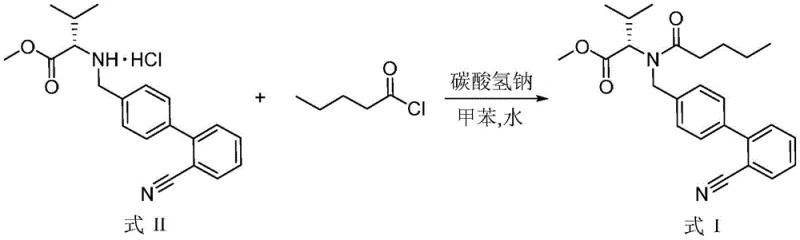
Traditional synthetic routes for preparing this specific valsartan intermediate typically involve the acylation of N-[(2'-cyanobiphenyl-4-yl)methyl]-(L)-valine methyl ester hydrochloride with n-valeryl chloride in the presence of an inorganic base and water. However, the inherent presence of water in these biphasic reaction systems poses a significant chemical challenge, as n-valeryl chloride is highly susceptible to hydrolysis. In prior art methodologies, this unavoidable hydrolysis necessitates the use of a substantial excess of the acid chloride reagent to drive the reaction to completion, which drastically inflates raw material costs. Furthermore, the hydrolysis byproduct, n-valeric acid, accumulates in the reaction mixture, complicating the purification process and requiring multiple, rigorous ammonia water washing steps to remove. These excessive washing cycles not only increase the volume of nitrogen-containing wastewater but also pose risks to the final product's purity and yield, creating bottlenecks for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The method disclosed in patent CN110078640B fundamentally alters the reaction dynamics by introducing a calculated amount of sodium chloride or potassium chloride into the system. This strategic addition utilizes the common ion effect to suppress the dissociation and subsequent hydrolysis of n-valeryl chloride, thereby preserving the reagent for the intended acylation reaction. As a result, the molar ratio of n-valeryl chloride to the starting material can be tightly controlled within a narrow range of 1:1.05 to 1:1.2, a significant improvement over the wasteful excesses of conventional methods. This optimization not only enhances the atomic economy of the process but also simplifies the downstream workup, as the reduced formation of valeric acid impurities minimizes the need for aggressive washing protocols. The reaction is conducted at mild temperatures between 15°C and 35°C, ensuring operational safety and energy efficiency while delivering high-purity products suitable for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Salt-Effect Mediated Acylation
The core innovation of this synthesis lies in the physicochemical manipulation of the biphasic reaction interface through the introduction of inorganic salts. In a standard aqueous-organic system, water molecules actively compete with the amine nucleophile to attack the carbonyl carbon of the acid chloride, leading to hydrolysis. By saturating the aqueous phase with chloride ions from sodium chloride or potassium chloride, the chemical potential for the hydrolysis reaction is thermodynamically disfavored. This phenomenon effectively shields the n-valeryl chloride from water attack, directing the reactivity almost exclusively towards the nucleophilic substitution with the valine ester hydrochloride. The acid binding agent, typically sodium carbonate or potassium carbonate, serves to neutralize the hydrochloric acid generated during the acylation, driving the equilibrium forward without promoting the side reactions associated with strong bases. This delicate balance ensures that the reaction proceeds with high selectivity, maintaining the integrity of the chiral center and preventing racemization, which is critical for the biological activity of the final API.
Impurity control is another critical aspect where this mechanism excels, particularly concerning the removal of unreacted starting materials and acid byproducts. The reduced generation of n-valeric acid means that the organic phase remains cleaner throughout the reaction, facilitating a more efficient separation during the quenching and washing stages. The protocol specifies a sequential washing procedure involving dilute ammonia, hydrochloric acid, and sodium bicarbonate, which is now more effective due to the lower burden of acidic impurities. This streamlined purification process ensures that the final product meets stringent purity specifications, often exceeding 99.7% as demonstrated in the patent examples. For quality assurance teams, this implies a more robust process with lower batch-to-batch variability, reducing the risk of failed quality control tests and ensuring a consistent supply of high-purity valsartan intermediates for downstream coupling reactions.
How to Synthesize N-[(2'-cyanobiphenyl-4-yl)methyl]-N-(1-oxo-pentyl)-L-valine methyl ester Efficiently
Implementing this synthesis route requires precise adherence to the specified reagent ratios and addition sequences to maximize the salt-effect benefit. The process begins with the dissolution of the starting material in a suitable organic solvent such as toluene, followed by the simultaneous addition of the base, salt, and water mixture to establish the protective ionic environment. The n-valeryl chloride is then added dropwise under strict temperature control to manage the exotherm and maintain reaction selectivity. Detailed standardized synthesis steps, including specific stirring rates, addition times, and analytical checkpoints, are provided in the technical guide below to ensure reproducibility at scale.
- Dissolve the valine ester hydrochloride (Formula II) in an organic solvent like toluene, then add an acid binding agent, sodium chloride or potassium chloride, and water to the mixture.
- Dropwise add n-valeryl chloride to the reaction system while maintaining a temperature between 15-35°C to ensure controlled acylation and minimize side reactions.
- Upon completion, quench with water and perform sequential alkali washing, acid washing, and a final alkali wash before reduced pressure distillation to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this salt-mediated synthesis method offers substantial advantages in terms of cost structure and supply chain resilience. The primary economic driver is the drastic reduction in the consumption of n-valeryl chloride, a reagent that represents a significant portion of the raw material cost. By minimizing hydrolysis, the process eliminates the need for purchasing and handling large excesses of this corrosive and moisture-sensitive chemical, leading to direct material cost savings. Additionally, the simplified workup procedure reduces the consumption of washing solvents and neutralizing agents, further lowering the variable costs associated with production. These efficiencies translate into a more competitive pricing model for the intermediate, allowing pharmaceutical manufacturers to optimize their overall cost of goods sold without compromising on quality standards.
- Cost Reduction in Manufacturing: The inhibition of acid chloride hydrolysis directly correlates to a significant decrease in raw material waste, as the reagent is utilized primarily for product formation rather than side reactions. This efficiency eliminates the need for expensive over-charging of reagents, resulting in substantial cost savings per kilogram of produced intermediate. Furthermore, the reduced impurity load decreases the demand for extensive purification steps, lowering energy consumption and solvent usage during the isolation phase. These cumulative effects create a leaner manufacturing process that is highly resistant to fluctuations in raw material market prices.
- Enhanced Supply Chain Reliability: The use of readily available inorganic salts like sodium chloride and potassium chloride ensures that the process is not dependent on scarce or specialized catalysts that could pose supply risks. The robustness of the reaction conditions, operating at mild temperatures and ambient pressure, reduces the likelihood of equipment failure or process upsets that could interrupt production schedules. This stability allows for consistent batch turnover and reliable delivery timelines, which is crucial for maintaining the continuity of the downstream API synthesis supply chain. Suppliers utilizing this method can offer greater assurance of on-time delivery and volume flexibility.
- Scalability and Environmental Compliance: The reduction in nitrogen-containing wastewater discharge, achieved by minimizing the need for multiple ammonia washes, significantly lowers the environmental burden of the manufacturing process. This aligns with increasingly stringent global environmental regulations, reducing the risk of compliance-related shutdowns or fines. The process is inherently scalable, as the salt-effect mechanism functions effectively regardless of batch size, facilitating a smooth transition from pilot plant to multi-ton commercial production. This scalability ensures that the supply can grow in tandem with market demand for Valsartan without requiring complex process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, based on the data provided in patent CN110078640B. These insights are designed to clarify the operational benefits and quality implications for stakeholders evaluating this technology for their supply chain. Understanding these details is key to making informed decisions about sourcing strategies and process adoption.
Q: How does the addition of sodium chloride improve the synthesis of valsartan intermediates?
A: The addition of sodium chloride or potassium chloride utilizes the common ion effect to significantly inhibit the hydrolysis of n-valeryl chloride in the aqueous-organic biphasic system. This suppression of hydrolysis reduces the consumption of the expensive acid chloride reagent and minimizes the formation of n-valeric acid impurities, leading to higher product purity and yield.
Q: What are the optimal reaction conditions for this acylation process?
A: The process operates effectively at mild temperatures ranging from 15°C to 35°C using solvents such as toluene, xylene, or dichloromethane. The molar ratio of the acid binding agent to the starting material is maintained between 2.2:1 and 4:1, while the mass ratio of the salt additive to the starting material is optimized between 0.3:1 and 1:1 for maximum efficiency.
Q: Why is this method considered superior for industrial scale-up compared to prior art?
A: Unlike conventional methods that require excessive amounts of acid chloride to compensate for hydrolysis, this method allows for a near-stoichiometric ratio (1:1.05-1.2). This reduction in reagent excess simplifies the workup procedure, reduces nitrogen-containing wastewater discharge, and lowers overall production costs, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and high-quality intermediate synthesis in the global pharmaceutical supply chain. Our technical team has extensively analyzed the patented salt-effect mediated acylation route and possesses the expertise to implement this advanced methodology at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this process are fully realized in terms of yield and purity. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Valsartan intermediate meets the exacting standards required for API manufacturing.
We invite procurement leaders and technical directors to collaborate with us to optimize their supply chain for Valsartan production. By leveraging our manufacturing capabilities and this advanced synthesis technology, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate how our optimized process can enhance your production efficiency and reduce overall costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
