Advanced Recovery of Avibactam Intermediates: Transforming Waste into High-Value API Precursors
Advanced Recovery of Avibactam Intermediates: Transforming Waste into High-Value API Precursors
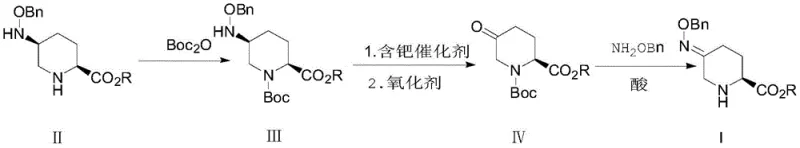
The pharmaceutical industry continuously seeks methods to enhance the sustainability and economic viability of complex API synthesis, particularly for critical antibiotics like Avibactam. Patent CN108373442B introduces a groundbreaking recovery method specifically designed to reclaim the (2S,5S)-5-((benzyloxy)amino)piperidine-2-carboxylate isomer, a significant by-product often discarded in traditional manufacturing processes. This technology addresses the critical inefficiency where valuable chiral intermediates are lost in mother liquors during salt formation, representing a substantial financial and environmental burden. By implementing this novel recovery route, manufacturers can transform what was previously considered waste into a high-purity feedstock, thereby optimizing the entire production lifecycle. The method leverages a sophisticated sequence of protection, catalytic transformation, and condensation to restore the stereochemical integrity required for downstream synthesis. For global procurement teams and R&D directors, this patent represents a pivotal shift towards circular chemistry principles within the pharmaceutical intermediates sector, offering a tangible pathway to reduce dependency on fresh raw materials while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for Avibactam intermediates, such as those disclosed in international patent applications like WO2012172368, typically involve the generation of diastereomeric mixtures that require rigorous separation. In these established processes, the desired (2S,5R) isomer is isolated via salification, but the resulting mother liquor retains a high concentration of the unwanted (2S,5S) isomer. Historically, this mother liquor has been treated as waste, leading to a significant loss of expensive chiral starting materials and increasing the overall cost of goods sold. Alternative attempts to recover these isomers, such as the direct oxidation methods described in CN107540600A, have proven inefficient due to low reaction yields and the generation of numerous impurities caused by harsh oxidizing conditions. These conventional approaches often fail to protect sensitive functional groups, resulting in the degradation of the piperidine ring or hydrolysis of ester moieties. Consequently, the purity of the recovered material is often insufficient for direct reuse in GMP manufacturing, necessitating additional purification steps that further erode profit margins. The accumulation of such chemical waste also poses severe challenges for environmental compliance, as the disposal of complex organic residues requires specialized treatment facilities.
The Novel Approach
The methodology outlined in CN108373442B offers a superior alternative by introducing a strategic protection-deprotection strategy that preserves the molecular scaffold while altering the oxidation state of the nitrogen center. Instead of attempting direct conversion which risks structural damage, this novel approach first masks the secondary amine with a Boc protecting group under mild basic conditions. This crucial step stabilizes the molecule against unwanted side reactions during the subsequent catalytic hydrogenation and oxidation phases. The process then utilizes a palladium-catalyzed hydrogenation followed by a controlled oxidation to convert the amino group into a ketone functionality, effectively resetting the stereocenter. Finally, condensation with benzyloxyamine hydrochloride regenerates the desired oxime intermediate with high fidelity. This multi-step recovery loop ensures that the (2S,5S) isomer is not merely recycled but chemically upgraded back into the main synthetic stream. The result is a robust, scalable process that maximizes atom economy and significantly lowers the environmental footprint of API intermediate production.
Mechanistic Insights into Palladium-Catalyzed Oxidative Recovery
The core of this recovery technology lies in the precise manipulation of the nitrogen oxidation state within the piperidine ring, facilitated by a tandem catalytic hydrogenation and oxidation sequence. In the second step of the process, the Boc-protected intermediate undergoes catalytic hydrogenation in the presence of a palladium-containing catalyst, such as palladium carbon or palladium hydroxide. This step selectively reduces the benzyloxyamino group to a primary amine without affecting the ester functionality or the carbamate protecting group. Immediately following this reduction, an oxidant such as 3,5-di-tert-butyl-1,2-benzoquinone is introduced to the reaction mixture. This specific oxidant is chosen for its ability to efficiently convert the newly formed amine into a ketone (Formula IV) while minimizing over-oxidation or ring cleavage. The mechanistic pathway likely involves the formation of an imine intermediate which is subsequently hydrolyzed or oxidized to the ketone. The use of alcohol solvents like methanol or ethanol in this stage is critical, as they facilitate the solubility of both the organic substrate and the inorganic catalyst while allowing for easy removal post-reaction. This seamless transition from reduction to oxidation in a single pot or sequential addition minimizes handling losses and exposure to air, which is vital for maintaining the high optical purity required for beta-lactamase inhibitor synthesis.
Impurity control is rigorously managed through the initial protection step and the selection of mild reaction conditions throughout the sequence. By employing weak bases such as triethylamine or sodium bicarbonate during the Boc protection phase, the process avoids the strong alkaline conditions that typically trigger ester hydrolysis, a common failure mode in piperidine chemistry. Furthermore, the specific choice of oxidants prevents the formation of chlorinated by-products that often arise when using hypochlorites or strong mineral acids. The condensation step in the final stage utilizes acid-binding agents like sodium acetate or pyridine to neutralize the hydrochloric acid released from the benzyloxyamine hydrochloride salt. This ensures that the reaction equilibrium shifts towards the formation of the oxime product rather than reverting to the ketone. The final deprotection using gaseous hydrogen chloride or trifluoroacetic acid is conducted under controlled temperatures to prevent racemization of the chiral centers. This comprehensive approach to impurity management ensures that the recovered material meets the high-purity pharmaceutical intermediates specifications necessary for regulatory approval, with HPLC purities consistently exceeding 98% in experimental trials.
How to Synthesize Avibactam Intermediate Efficiently
The synthesis of the recovered Avibactam intermediate involves a streamlined three-stage protocol that begins with the isolation of the isomer from mother liquor and concludes with the regeneration of the oxime precursor. The process is designed to be operationally simple, utilizing standard equipment found in most fine chemical manufacturing facilities, which facilitates rapid technology transfer and scale-up. Detailed procedural parameters, including specific solvent ratios, temperature gradients, and catalyst loading rates, are essential for achieving the optimal balance between reaction rate and selectivity. Operators must strictly adhere to the specified molar ratios of reagents, particularly the oxidant and the protecting group source, to prevent the accumulation of unreacted starting material or over-oxidized by-products. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process engineers aiming to implement this recovery loop.
- Protect the amine group of the waste isomer (Formula II) using Boc2O under weakly basic conditions to form the protected intermediate (Formula III).
- Perform catalytic hydrogenation using a palladium catalyst followed by oxidation with agents like 3,5-di-tert-butyl-1,2-benzoquinone to generate the ketone intermediate (Formula IV).
- Condense the ketone intermediate with benzyloxyamine hydrochloride and perform acid-mediated deprotection to regenerate the target Avibactam precursor (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the implementation of this recovery method translates directly into enhanced operational resilience and cost efficiency. By reclaiming the (2S,5S) isomer from waste streams, manufacturers can significantly reduce the volume of fresh raw materials required per kilogram of final API produced. This reduction in material consumption acts as a natural hedge against price volatility in the global market for chiral building blocks and specialty chemicals. Moreover, the ability to internally recycle intermediates reduces the dependency on external suppliers for every batch, thereby mitigating risks associated with logistics delays or geopolitical supply disruptions. The simplified workflow, which avoids complex chromatographic separations or extreme reaction conditions, also lowers the barrier for commercial scale-up of complex pharmaceutical intermediates. This efficiency allows production facilities to increase throughput without proportional increases in capital expenditure or utility consumption, driving down the unit cost of production.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the conversion of a waste product into a valuable asset, effectively lowering the net cost of raw materials. By recovering the isomer that would otherwise be discarded, the overall yield of the synthesis pathway is improved, meaning less starting material is needed to produce the same amount of final drug substance. The elimination of harsh reagents and the use of recyclable solvents like alcohols further contribute to cost savings by reducing waste disposal fees and solvent procurement expenses. Additionally, the high selectivity of the palladium catalyst minimizes the formation of difficult-to-remove impurities, reducing the need for expensive downstream purification processes such as preparative HPLC. These cumulative efficiencies result in a leaner manufacturing process with a substantially improved margin profile.
- Enhanced Supply Chain Reliability: Integrating this recovery loop creates a more self-sufficient supply chain by maximizing the utility of every batch of input material. In scenarios where global supply chains for specific chiral precursors are constrained, the ability to recycle internal waste streams provides a critical buffer against shortages. The robustness of the reaction conditions, which tolerate a range of solvents and mild temperatures, ensures consistent production output even if specific reagent grades vary slightly. This reliability is paramount for maintaining continuous manufacturing schedules and meeting the strict delivery timelines demanded by large-scale pharmaceutical clients. Furthermore, the reduced environmental burden simplifies regulatory reporting and compliance, ensuring uninterrupted operations in regions with stringent environmental laws.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as filtration, distillation, and crystallization that are standard in industrial chemical plants. The avoidance of toxic heavy metals beyond the contained palladium catalyst, and the use of benign oxidants, aligns with modern green chemistry principles. This alignment facilitates easier permitting and reduces the long-term liability associated with hazardous waste storage and treatment. The ability to operate at near-ambient temperatures and pressures also enhances safety profiles, reducing the risk of thermal runaways and allowing for larger batch sizes. Consequently, this technology supports the commercial scale-up of Avibactam production while adhering to increasingly strict global environmental, social, and governance (ESG) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isomer recovery technology. These answers are derived directly from the experimental data and claims presented in patent CN108373442B, providing a factual basis for decision-making. Understanding the nuances of catalyst selection, solvent compatibility, and purification requirements is essential for R&D teams evaluating this process for adoption. The responses below clarify the operational boundaries and expected performance metrics based on the disclosed embodiments.
Q: Why is the recovery of the (2S,5S) isomer critical in Avibactam synthesis?
A: In traditional synthesis routes, the formation of diastereomeric mixtures leads to significant material loss in the mother liquor during salification. Recovering the (2S,5S) isomer converts this waste stream back into usable starting material, drastically improving overall atom economy and reducing raw material costs.
Q: What are the advantages of using weak bases in the protection step?
A: The use of weak bases such as triethylamine or sodium carbonate prevents the hydrolysis of the sensitive ester bonds present in the molecule while still providing sufficient alkalinity to facilitate the introduction of the Boc protecting group on the nitrogen atom.
Q: How does this method improve environmental compliance?
A: By recycling the isomer waste instead of discarding it, the process significantly reduces the volume of chemical waste requiring treatment. Furthermore, the mild reaction conditions and selective catalysts minimize the generation of hazardous by-products compared to harsh direct oxidation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avibactam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate recovery in the competitive landscape of beta-lactamase inhibitor production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN108373442B can be seamlessly integrated into your supply chain. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of recovered intermediate meets the exacting standards required for API synthesis. Our infrastructure is designed to handle complex chiral molecules with precision, utilizing state-of-the-art analytical equipment to monitor stereochemical integrity throughout the manufacturing process.
We invite you to collaborate with us to optimize your Avibactam production costs and enhance your supply chain sustainability. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced recovery capabilities can drive value for your organization. Let us be your partner in achieving both economic and environmental excellence in pharmaceutical manufacturing.
