Revolutionizing Bortezomib Production: A Scalable Sulfinamide-Based Synthetic Strategy
The global demand for proteasome inhibitors in oncology therapy continues to surge, driving the need for robust and economically viable manufacturing processes for key active pharmaceutical ingredients (APIs). Patent CN102659919A introduces a transformative synthetic methodology for bortezomib, a dipeptidyl boronic acid compound widely used in the treatment of multiple myeloma and mantle cell lymphoma. This innovation addresses critical bottlenecks in existing supply chains by replacing costly chiral auxiliaries with readily available (R)-tert-butylsulfinamide. By leveraging a copper-catalyzed borylation strategy, this route not only enhances stereochemical control but also simplifies the purification landscape, positioning it as a preferred pathway for reliable API intermediate suppliers seeking to optimize their portfolio.
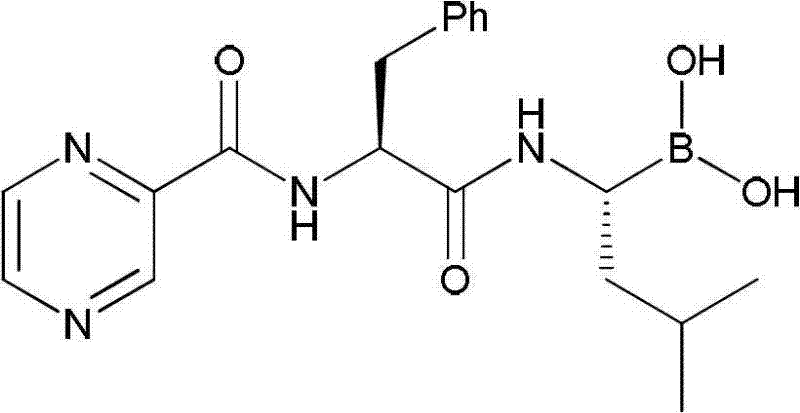
Traditional manufacturing pathways for bortezomib have long been plagued by prohibitive costs and operational complexities that hinder efficient commercial scale-up of complex peptide boronates. One prominent legacy method, disclosed in WO2009004350, relies heavily on (1S,2S,3R,5S)-(+)-2,3-pinanediol as a chiral ligand. While effective in establishing stereochemistry, this reagent is exceptionally expensive, drastically inflating the bill of materials. Furthermore, this route necessitates cryogenic reaction conditions, often requiring temperatures as low as -70°C for chloromethylene insertion and amination steps. Such extreme thermal requirements demand specialized refrigeration infrastructure and result in significant energy consumption, rendering the process economically unfeasible for high-volume production.
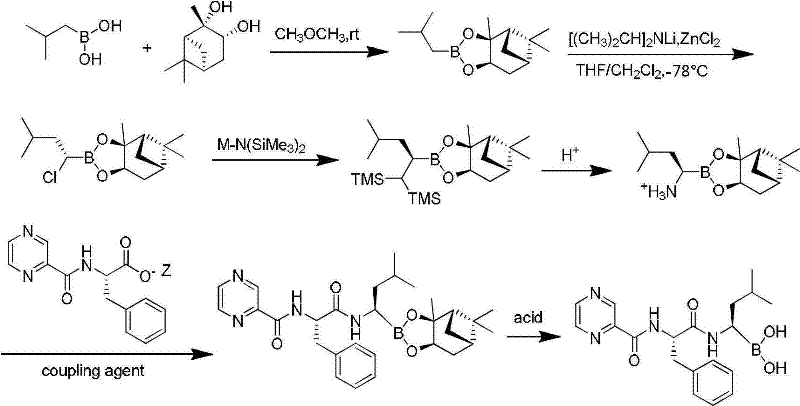
Another alternative approach, described in CN101812026, attempts to mitigate raw material costs by utilizing 3-methylbutyraldehyde and (R)-1-phenylethylamine. However, this modification introduces new liabilities regarding product quality and safety. The use of (R)-1-phenylethylamine often results in lower enantiomeric excess (ee) values, compromising the optical purity of the final drug substance and necessitating additional, yield-losing purification steps. Moreover, this route depends on catalytic hydrogenation using palladium on carbon (Pd/C) under elevated pressure and temperature. The reliance on precious metal catalysts not only increases direct material costs but also introduces stringent regulatory hurdles regarding residual metal limits in the final API, complicating the validation process for pharmaceutical manufacturers.
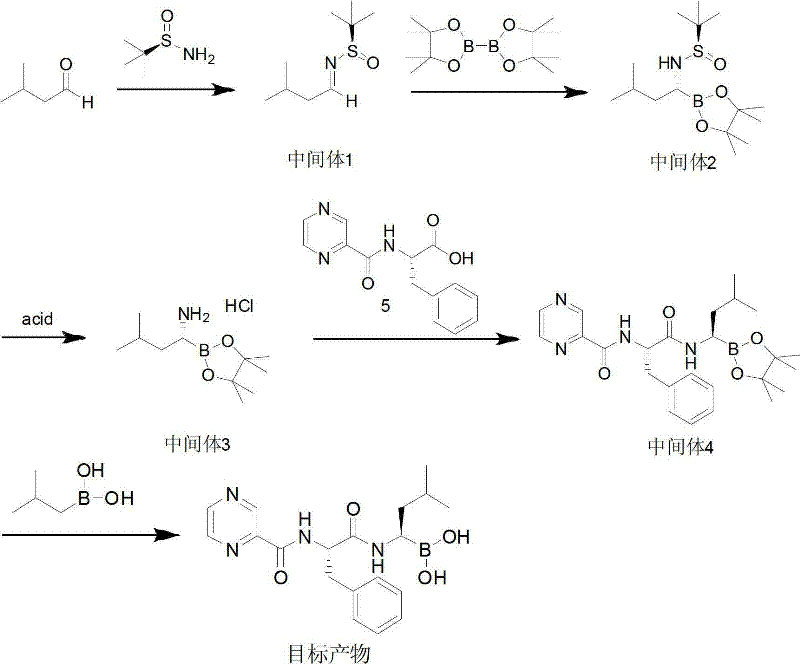
In stark contrast, the novel approach detailed in CN102659919A offers a streamlined, cost-reduction in pharmaceutical manufacturing by fundamentally redesigning the chiral induction strategy. This method initiates with the condensation of inexpensive isovaleraldehyde and (R)-tert-butylsulfinamide, forming a chiral sulfinamide intermediate with exceptional stereocontrol. The subsequent key transformation involves a copper-catalyzed nucleophilic addition of pinacol diboron, which proceeds efficiently at mild temperatures ranging from 20°C to 80°C. This elimination of cryogenic and high-pressure steps represents a paradigm shift in process safety and energy efficiency. The final deprotection and coupling stages utilize standard reagents like isobutylboronic acid for hydrolysis, ensuring that the overall process remains accessible and scalable for industrial facilities without requiring exotic equipment.
Mechanistic Insights into Copper-Catalyzed Asymmetric Borylation
The core technological breakthrough of this synthesis lies in the stereoselective construction of the C-B bond using a copper/N-heterocyclic carbene (NHC) catalytic system. In this mechanism, the copper salt, typically cuprous chloride or bromide, coordinates with the imidazolium salt to generate an active copper-NHC species in situ. This complex facilitates the transmetallation with pinacol diboron, creating a nucleophilic copper-boryl species. This species then attacks the electrophilic imine carbon of the sulfinamide intermediate with high facial selectivity, dictated by the bulky tert-butyl group on the sulfur atom. The steric hindrance provided by the sulfinamide moiety ensures that the boron adds to the desired face of the imine, locking in the (R)-configuration with high fidelity. This mechanistic precision is crucial for achieving the high enantiomeric purity required for oncology drugs, minimizing the formation of diastereomers that are difficult to separate downstream.
Furthermore, the impurity profile of this route is significantly cleaner compared to transition-metal hydrogenation methods. By avoiding palladium catalysts, the process eliminates the risk of Pd contamination, a common failure point in regulatory filings. The hydrolysis steps are carefully controlled using acidic conditions (pH 5-6) with isobutylboronic acid acting as a trans-esterification agent to liberate the free boronic acid. This specific choice of hydrolysis agent prevents the decomposition of the sensitive boronic acid moiety, which can otherwise degrade into inactive boroxines or undergo protodeboronation. The result is a high-purity bortezomib intermediate that meets stringent pharmacopeial standards with minimal need for extensive chromatographic purification, thereby enhancing the overall mass balance and yield of the operation.
How to Synthesize Bortezomib Efficiently
The synthesis of this critical oncology agent requires precise control over reaction parameters to maintain stereochemical integrity and maximize yield. The process begins with the formation of the chiral imine, followed by the pivotal copper-catalyzed borylation which sets the stereocenter. Subsequent hydrolysis and peptide coupling steps must be managed to prevent racemization. For process chemists looking to implement this technology, the detailed standardized synthesis steps are outlined below to ensure reproducibility and compliance with GMP standards.
- Condense isovaleraldehyde with (R)-tert-butylsulfinamide using a titanium or cesium catalyst to form the chiral sulfinamide intermediate.
- Perform copper-catalyzed nucleophilic addition with pinacol diboron to generate the chiral boronate ester.
- Hydrolyze the boronate ester under acidic conditions, couple with the pyrazine-phenylalanine fragment, and finalize with isobutylboronic acid hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfinamide-based route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material supply chain. By substituting rare and expensive chiral ligands like pinanediol with commodity chemicals like isovaleraldehyde and tert-butylsulfinamide, manufacturers can insulate themselves from volatile pricing fluctuations associated with specialty fine chemicals. This shift towards abundant feedstocks ensures a more stable and predictable cost structure, allowing for long-term supply agreements with reduced risk of interruption due to raw material scarcity.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the elimination of cryogenic operations and precious metal catalysts. Operating reactions at ambient or slightly elevated temperatures (20-80°C) removes the need for energy-intensive cooling systems, leading to significant utility savings. Additionally, the avoidance of palladium catalysts eradicates the costs associated with metal scavenging and validation testing for residual metals. These cumulative efficiencies translate into a leaner cost of goods sold (COGS), enabling competitive pricing strategies in the generic API market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on easily sourced starting materials enhances the resilience of the supply chain. Isovaleraldehyde and basic copper salts are produced globally in large volumes, reducing the dependency on single-source suppliers for niche chiral reagents. This diversification of the supply base mitigates the risk of production stoppages caused by upstream shortages. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility availability, ensuring consistent output even in challenging operational environments.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is inherently greener and safer to scale. The absence of high-pressure hydrogenation removes a major safety hazard from the plant floor, lowering insurance premiums and safety compliance costs. The milder reaction conditions also reduce the generation of hazardous waste associated with extreme pH adjustments or solvent exchanges required in cryogenic workups. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, making the manufacturing process more attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding the scalability, purity, and regulatory implications of the sulfinamide route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of the technology's maturity and readiness for commercial deployment.
Q: Why is the sulfinamide route superior to pinanediol-based synthesis?
A: The sulfinamide route eliminates the need for expensive chiral ligands like pinanediol and avoids cryogenic conditions (-70°C), significantly reducing operational costs and equipment requirements.
Q: How does this method ensure high enantiomeric purity?
A: By utilizing (R)-tert-butylsulfinamide as a chiral inducing reagent, the process achieves high ee values (>95%) without requiring complex chiral chromatography steps.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (20-80°C), utilize inexpensive starting materials like isovaleraldehyde, and avoid hazardous high-pressure hydrogenation, making it ideal for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bortezomib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this patent can be realized in a GMP-compliant environment. We are equipped with state-of-the-art reactors capable of handling sensitive organometallic chemistry and rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of bortezomib intermediate meets the highest global standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthetic route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security into your supply chain.
