Scalable Copper-Catalyzed Synthesis of Bortezomib Intermediates for Commercial API Production
Introduction to Advanced Bortezomib Manufacturing Technology
The pharmaceutical landscape for oncology treatments continues to evolve, with proteasome inhibitors like bortezomib remaining a cornerstone therapy for multiple myeloma. However, the complexity of synthesizing the chiral leucine boronic acid core has historically posed significant challenges for manufacturers seeking reliable supply chains. Patent CN102675415B introduces a transformative methodology that leverages asymmetric copper catalysis to construct the critical carbon-boron bond with exceptional stereocontrol. This technical breakthrough shifts the paradigm from hazardous cryogenic lithiation to a more robust, ambient-temperature catalytic process. By utilizing chiral sulfinamides as induction reagents in conjunction with specialized N-heterocyclic carbene (NHC) copper complexes, this route achieves high diastereoselectivity without the extreme operational constraints of legacy methods.
For R&D directors and procurement specialists, understanding the nuances of this patented pathway is essential for securing a competitive edge in API sourcing. The ability to generate high-purity intermediates through a streamlined sequence not only enhances the impurity profile of the final drug substance but also drastically simplifies the manufacturing workflow. As a leading entity in fine chemical synthesis, we recognize that adopting such advanced catalytic technologies is key to maintaining supply continuity and meeting the stringent quality standards demanded by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for bortezomib and its key leucine boronic acid intermediates have long relied on organolithium chemistry. These conventional methods typically necessitate cryogenic conditions, often requiring temperatures as low as -78°C to maintain the stability of reactive lithiated species and to control stereoselectivity. Such extreme thermal requirements impose a heavy burden on manufacturing infrastructure, demanding specialized refrigeration equipment and substantial energy consumption. Furthermore, organolithium reagents are notoriously sensitive to moisture and oxygen, requiring rigorous anhydrous conditions that complicate scale-up and increase the risk of batch failure. The handling of these hazardous reagents also raises significant safety concerns for plant operators, while the subsequent quenching and workup procedures often generate large volumes of saline waste, complicating environmental compliance and waste management protocols.
The Novel Approach
In stark contrast, the methodology disclosed in CN102675415B utilizes a copper-catalyzed borylation strategy that operates under significantly milder conditions. The core innovation lies in the reaction of a chiral sulfinylimine with a diboron reagent, mediated by a tailored copper catalyst. This transformation can proceed efficiently at room temperature or with mild heating in common solvents like ethanol or dioxane. 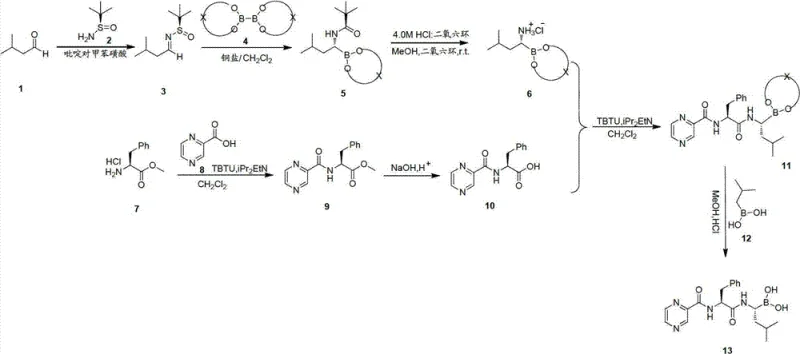
This shift from cryogenic lithiation to ambient catalysis represents a monumental improvement in process safety and scalability. The use of stable diboron esters eliminates the need for pyrophoric reagents, thereby reducing the hazard profile of the manufacturing site. Moreover, the reaction demonstrates excellent functional group tolerance and high yields, minimizing the need for extensive purification steps that often erode overall process efficiency. For a reliable bortezomib intermediate supplier, this novel approach translates directly into a more resilient production capability, capable of delivering consistent quality regardless of seasonal or infrastructural variations.
Mechanistic Insights into Copper-Catalyzed Enantioselective Borylation
The heart of this synthetic advancement is the sophisticated catalytic cycle driven by NHC-copper complexes. The mechanism initiates with the activation of the diboron reagent by the copper center, generating a nucleophilic copper-boryl species. This active complex then undergoes a stereoselective addition to the C=N double bond of the sulfinylimine substrate. The chiral information encoded in the tert-butylsulfinyl group plays a pivotal role here, directing the approach of the copper-boryl species to one specific face of the imine. This cooperative interaction between the chiral auxiliary and the metal catalyst ensures the formation of the desired stereoisomer with high diastereomeric ratio (dr) and enantiomeric excess (ee). 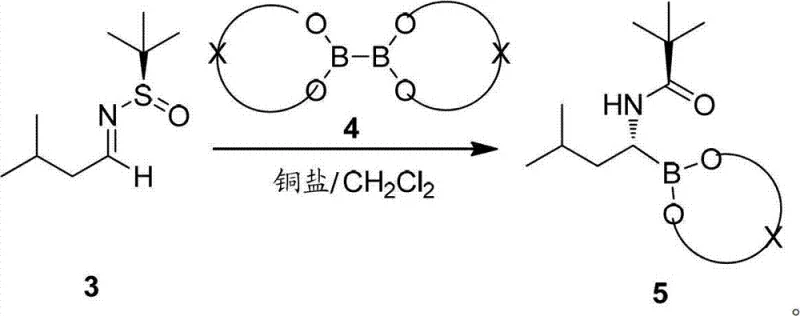
Following the borylation event, the resulting intermediate retains the chiral integrity necessary for downstream coupling. The patent highlights the versatility of various NHC ligands, such as those depicted in Formula I, which can be tuned to optimize reactivity and selectivity. 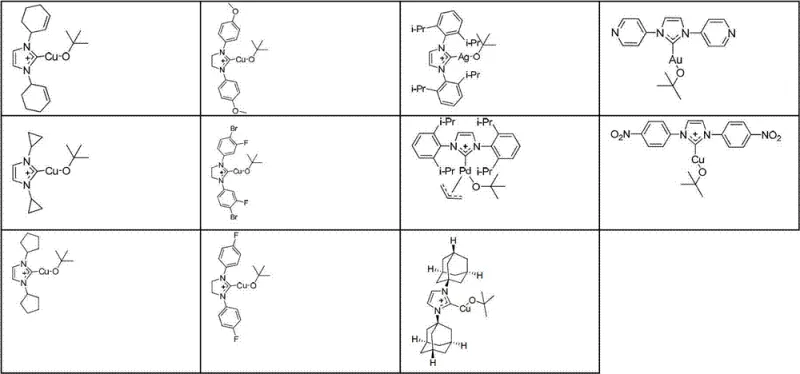
Understanding this mechanistic nuance is critical for impurity control. By precisely managing the catalyst loading and reaction stoichiometry, manufacturers can suppress the formation of undesired diastereomers and homocoupling byproducts. This level of control is paramount for pharmaceutical applications, where the presence of isomeric impurities can compromise the efficacy and safety of the final therapeutic agent. The robustness of this catalytic system allows for reproducible results across different batches, a key metric for quality assurance teams evaluating potential technology transfers.
How to Synthesize Bortezomib Intermediates Efficiently
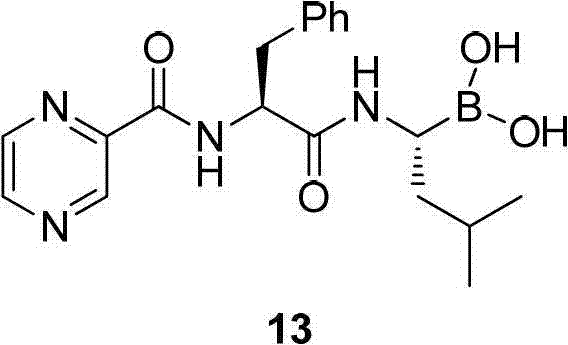
The practical execution of this synthesis involves a convergent strategy that assembles the molecule from two primary fragments: the chiral leucine boronic acid derivative and the pyrazine-phenylalanine acid component. The process begins with the condensation of isobutyraldehyde and R-tert-butylsulfinamide to form the imine, followed by the crucial copper-catalyzed borylation step. Subsequent acidic deprotection yields the free amino boronic acid salt, which is then coupled with the protected phenylalanine derivative using standard peptide coupling reagents like TBTU. The final step involves hydrolysis to reveal the active boronic acid pharmacophore. For detailed operational parameters, solvent choices, and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Condense isobutyraldehyde with R-tert-butylsulfinamide under acid catalysis to form the chiral sulfinylimine intermediate.
- Perform enantioselective borylation using a diboron reagent and an NHC-Copper catalyst to generate the chiral amino boronic acid derivative.
- Deprotect the sulfinyl group and condense the resulting amine with a pyrazine-phenylalanine derivative, followed by final hydrolysis to yield bortezomib.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the catalytic route described in CN102675415B offers profound strategic benefits for procurement managers and supply chain heads focused on cost reduction in API manufacturing. The transition away from cryogenic processes eliminates a major bottleneck in production scheduling, allowing for more flexible batch planning and reduced utility costs associated with extreme cooling. Furthermore, the reliance on commodity chemicals like isobutyraldehyde and stable diboron reagents mitigates the risk of raw material shortages that often plague supply chains dependent on specialized, hazardous reagents. This stability ensures a continuous flow of materials, safeguarding against market volatility and delivery delays.
- Cost Reduction in Manufacturing: The elimination of energy-intensive cryogenic cooling systems results in substantial operational expenditure savings. Additionally, the high selectivity of the copper catalyst reduces the burden on downstream purification, lowering solvent consumption and waste disposal costs. By avoiding expensive and hazardous organolithium reagents, the overall material cost per kilogram of the intermediate is significantly optimized, enhancing the margin profile for the final API.
- Enhanced Supply Chain Reliability: The use of robust, air-stable catalysts and reagents simplifies logistics and storage requirements. Unlike moisture-sensitive lithiation reagents that require special handling and transport, the materials in this process can be managed with standard warehouse protocols. This ease of handling reduces the risk of supply disruptions caused by transportation incidents or storage failures, ensuring a dependable supply of high-purity pharmaceutical intermediates for downstream formulation.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced hazard profile make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The simplified workup procedures generate less hazardous waste, aligning with increasingly stringent environmental regulations. This green chemistry advantage not only facilitates regulatory approval but also enhances the corporate sustainability profile of the manufacturing partner, a growing priority for global pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What are the advantages of the copper-catalyzed method over traditional lithiation for bortezomib?
A: The copper-catalyzed method described in CN102675415B operates under mild conditions (room temperature to moderate heat) compared to the cryogenic conditions (-78°C) required for traditional organolithium routes, significantly reducing energy costs and improving operational safety.
Q: What is the role of the sulfinamide in this synthesis?
A: The chiral tert-butylsulfinamide acts as a chiral auxiliary that directs the stereoselective addition of the boron species, ensuring high diastereoselectivity and optical purity in the final amino boronic acid intermediate.
Q: Can this process be scaled for industrial API production?
A: Yes, the process utilizes commercially available raw materials like isobutyraldehyde and avoids hazardous cryogenic steps, making it highly suitable for commercial scale-up and consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bortezomib Intermediate Supplier
The technological advancements detailed in patent CN102675415B represent a significant leap forward in the efficient production of proteasome inhibitor intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such cutting-edge methodologies for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of bortezomib intermediate meets the exacting standards required for clinical and commercial use.
We invite you to collaborate with us to optimize your supply chain for this critical oncology ingredient. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced catalytic capabilities can drive value and security for your pharmaceutical projects.
