Scalable Synthesis of 5-Chlorothiophene-2-Formic Acid: A Breakthrough for Rivaroxaban Production
The pharmaceutical industry's relentless pursuit of efficient anticoagulant therapies has placed 5-chlorothiophene-2-formic acid in the spotlight as a critical building block for Rivaroxaban, a next-generation Factor Xa inhibitor. Patent CN110317189B introduces a transformative synthetic methodology that shifts the paradigm from complex, hazardous multi-step sequences to a streamlined, direct chlorination process. By utilizing thiophene-2-formic acid as the starting substrate and trichloroisocyanuric acid (TCCA) as the chlorinating agent, this invention addresses the longstanding bottlenecks of yield, safety, and environmental impact that have plagued previous manufacturing protocols. For R&D directors and supply chain strategists, this patent represents not merely a chemical variation, but a robust platform for securing the supply chain of high-value cardiovascular medications through a route that is inherently safer and more economically viable.
This technical insight report dissects the mechanistic advantages of the TCCA-mediated electrophilic substitution, contrasting it sharply with legacy methods that rely on unstable organometallics or wasteful formylation chemistries. The ability to operate under mild acidic conditions with recyclable solvents offers a compelling value proposition for commercial scale-up, promising significant reductions in both capital expenditure and operational overhead. As we delve into the specifics of this innovation, it becomes clear that adopting this technology aligns perfectly with the goals of modern green chemistry while delivering the high-purity standards required for GMP-grade API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-chlorothiophene-2-formic acid has been fraught with chemical inefficiencies and safety hazards that complicate large-scale manufacturing. One prevalent legacy route, described in Chinese Patent CN 109422720A, initiates with 2-chlorothiophene, subjecting it to Vilsmeier-Haack formylation to generate the aldehyde, followed by a subsequent oxidation step to reach the carboxylic acid. 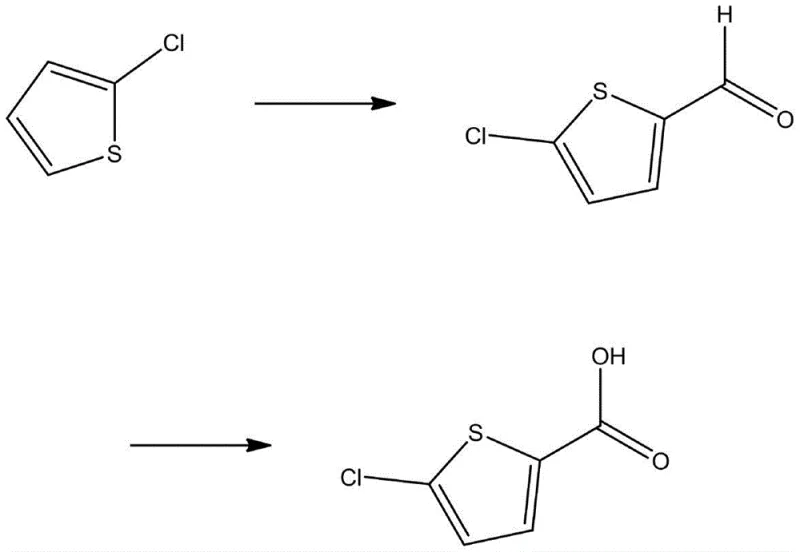 This pathway is fundamentally flawed for industrial application due to the hydrolytic instability of the formylation reagents and the generation of massive quantities of phosphoric acid wastewater, creating a severe environmental burden and escalating waste treatment costs. Furthermore, the multi-step nature of this sequence inevitably leads to cumulative yield losses, making the final product economically uncompetitive.
This pathway is fundamentally flawed for industrial application due to the hydrolytic instability of the formylation reagents and the generation of massive quantities of phosphoric acid wastewater, creating a severe environmental burden and escalating waste treatment costs. Furthermore, the multi-step nature of this sequence inevitably leads to cumulative yield losses, making the final product economically uncompetitive.
An alternative approach documented in RSC Advances (2014) attempts to bypass formylation by employing Lithium Diisopropylamide (LDA) to metalate 2-chlorothiophene, followed by carboxylation with carbon dioxide. 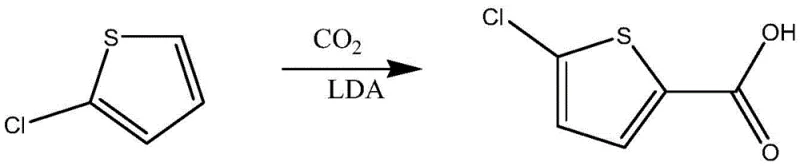 While chemically elegant on a laboratory scale, this method is a logistical nightmare for production facilities. LDA is a pyrophoric, highly corrosive base that demands stringent cryogenic conditions (typically below -70°C) to prevent decomposition and side reactions. The infrastructure required to maintain such low temperatures safely on a metric-ton scale is prohibitively expensive, and the handling risks associated with bulk LDA storage and transfer pose unacceptable safety liabilities for modern chemical plants.
While chemically elegant on a laboratory scale, this method is a logistical nightmare for production facilities. LDA is a pyrophoric, highly corrosive base that demands stringent cryogenic conditions (typically below -70°C) to prevent decomposition and side reactions. The infrastructure required to maintain such low temperatures safely on a metric-ton scale is prohibitively expensive, and the handling risks associated with bulk LDA storage and transfer pose unacceptable safety liabilities for modern chemical plants.
The Novel Approach
In stark contrast to these cumbersome legacy strategies, the method disclosed in CN110317189B leverages a direct electrophilic aromatic substitution on a pre-functionalized thiophene ring. By starting with thiophene-2-formic acid, the synthesis bypasses the need for introducing the carboxyl group entirely, focusing solely on the regioselective installation of the chlorine atom at the 5-position. The use of trichloroisocyanuric acid (TCCA) as the chlorinating source is a masterstroke of process chemistry; TCCA is a stable, solid, non-hygroscopic reagent that is inexpensive and easy to handle compared to gaseous chlorine or corrosive sulfuryl chloride. This shift allows the reaction to proceed in a homogeneous acidic medium, typically sulfuric acid, which serves dual roles as both solvent and catalyst, thereby simplifying the reaction matrix and facilitating easier downstream processing.
Mechanistic Insights into TCCA-Mediated Electrophilic Chlorination
The core of this innovation lies in the precise control of electrophilic aromatic substitution (EAS) on the thiophene heterocycle. Thiophene-2-carboxylic acid possesses an electron-withdrawing carboxyl group at the C2 position, which deactivates the ring towards electrophilic attack compared to unsubstituted thiophene. However, the sulfur atom in the thiophene ring exerts a strong activating influence, particularly at the alpha positions (C2 and C5). Since the C2 position is blocked by the carboxyl group, the C5 position remains the most nucleophilic site available for substitution. In the presence of a strong acid like sulfuric acid, TCCA generates a potent chloronium ion equivalent (Cl+) or a protonated TCCA species that acts as the active electrophile. The reaction mechanism involves the formation of a sigma-complex (Wheland intermediate) at the C5 position, followed by rearomatization via the loss of a proton, driven by the restoration of the aromatic sextet.
From an impurity control perspective, this mechanism offers distinct advantages over radical chlorination pathways which often lead to polychlorinated byproducts or ring degradation. The acidic conditions suppress radical formation, ensuring that the substitution remains strictly electrophilic and regioselective. The patent data indicates that by controlling the stoichiometry of TCCA (molar ratio of 1:0.3 to 1:3.5 relative to substrate) and maintaining low temperatures (-5 to 30°C), the formation of dichloro-impurities is minimized. Furthermore, the byproduct of the chlorination, cyanuric acid, is insoluble in the acidic reaction medium and can be easily removed during the aqueous workup, preventing it from contaminating the final organic phase. This inherent cleanliness of the reaction profile reduces the burden on purification steps, directly contributing to the high purity (>98%) reported in the experimental examples.
How to Synthesize 5-Chlorothiophene-2-Formic Acid Efficiently
The operational simplicity of this patented route makes it highly attractive for technology transfer and scale-up. The process begins with the preparation of the acidic reaction medium, where concentrated sulfuric acid is cooled to near-freezing temperatures to manage the exotherm of the subsequent additions. Thiophene-2-formic acid is dissolved into this medium, creating a homogeneous solution ready for chlorination. The critical step involves the controlled, batched addition of solid TCCA, which must be dosed slowly to maintain the reaction temperature between -5°C and 0°C, preventing thermal runaway and ensuring optimal selectivity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction vessel by adding concentrated sulfuric acid and cooling the mixture to 0°C to ensure thermal control before substrate addition.
- Add thiophene-2-formic acid to the cooled acid solution, followed by the batched addition of trichloroisocyanuric acid (TCCA) while maintaining the temperature between -5°C and 0°C.
- Monitor the reaction progress via TLC until completion, then quench into ice water to crystallize the crude product, followed by recrystallization from ethanol to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this TCCA-based synthesis route offers profound strategic benefits that extend far beyond simple reagent substitution. The elimination of cryogenic requirements and pyrophoric reagents fundamentally alters the risk profile of the manufacturing process, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized low-temperature infrastructure. This reduction in capital intensity translates directly into lower fixed costs per kilogram of product. Moreover, the stability of TCCA allows for flexible inventory management; unlike LDA which must be generated in situ or stored under inert atmosphere with short shelf-life, TCCA can be stocked in bulk, ensuring supply continuity even during market fluctuations of specialty reagents.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the replacement of expensive, hazardous reagents with commodity chemicals. LDA is a high-cost reagent that requires careful quenching and disposal, whereas TCCA is a mass-produced industrial chemical with a fraction of the cost. Additionally, the ability to recycle the sulfuric acid solvent significantly reduces raw material consumption and waste disposal fees. By collapsing a multi-step sequence (formylation + oxidation) into a single substitution step, the process also saves on labor, energy, and reactor occupancy time, leading to substantial overall cost savings in API intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Thiophene-2-formic acid is a commercially accessible building block, and TCCA is produced globally in large volumes for water treatment and sanitation, ensuring a robust supply base that is not subject to the bottlenecks of niche fine chemical suppliers. The simplified workflow also reduces the lead time for production batches; without the need for complex distillation or chromatographic purification often required to remove LDA byproducts or phosphoric waste, the turnaround time from raw material intake to finished goods is drastically shortened, enabling faster response to market demand.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden flaws in laboratory routes, but this method is designed with scale-up in mind. The heterogeneous nature of the workup (crystallization from ice water) is easily scalable using standard filtration equipment, avoiding the emulsions and extraction difficulties common in other organic syntheses. Environmentally, the process is superior as it avoids the generation of phosphorus-containing wastewater, which is difficult and costly to treat to meet regulatory discharge limits. The primary byproduct, cyanuric acid, is non-toxic and can potentially be recovered or disposed of with minimal environmental impact, aligning the manufacturing process with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical disclosures within Patent CN110317189B, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: Why is the TCCA method superior to LDA carboxylation for producing 5-chlorothiophene-2-formic acid?
A: The TCCA method avoids the use of LDA (Lithium Diisopropylamide), which is pyrophoric, highly corrosive, and requires cryogenic conditions (-78°C). The new method operates at mild temperatures (-5 to 30°C) using stable, solid reagents, drastically improving operational safety and reducing equipment costs.
Q: What is the expected purity and yield of 5-chlorothiophene-2-formic acid using this patent route?
A: According to Patent CN110317189B, the optimized process yields products with purity exceeding 98% and isolated yields around 82%, which is significantly higher than many traditional multi-step routes that suffer from cumulative yield losses.
Q: How does this synthesis route impact environmental compliance?
A: Unlike the Vilsmeier-Haack formylation route which generates large volumes of phosphoric acid wastewater, the TCCA method utilizes recyclable sulfuric acid and produces cyanuric acid as a benign byproduct, simplifying waste treatment and lowering environmental compliance costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chlorothiophene-2-Formic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving anticoagulants depends on the reliability and quality of the underlying supply chain. Our technical team has thoroughly analyzed the TCCA-mediated synthesis route and validated its potential for high-efficiency production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-chlorothiophene-2-formic acid meets the exacting standards required for downstream API synthesis.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this advanced patent technology, we can offer a competitive pricing structure without compromising on quality or delivery timelines. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can strengthen your supply chain for Rivaroxaban and related therapeutic programs.
