Advanced Synthesis of 5-Chlorothiophene-2-Formic Acid for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for critical intermediates, particularly those serving as building blocks for novel anticoagulants like razaxaban. Patent CN102993165A introduces a transformative synthesis method for 5-chlorothiophene-2-formic acid, a pivotal precursor in cardiovascular drug development. This technology replaces hazardous cryogenic lithiation processes with a温和 (mild) two-step sequence involving Vilsmeier-Haack formylation followed by nitric acid oxidation. By shifting away from pyrophoric reagents and extreme low-temperature requirements, this innovation addresses fundamental safety and scalability challenges inherent in legacy manufacturing protocols. The methodology demonstrates exceptional regioselectivity and yield efficiency, positioning it as a superior alternative for industrial-scale production of high-purity thiophene derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-chlorothiophene-2-formic acid has relied heavily on organolithium chemistry or haloform reactions, both of which present severe operational bottlenecks for commercial manufacturing. The traditional lithiation route utilizing n-Butyl Lithium necessitates deep cooling conditions, often below -70°C, to maintain stability and prevent side reactions, imposing immense energy costs and requiring specialized cryogenic infrastructure. Furthermore, n-Butyl Lithium is highly pyrophoric and moisture-sensitive, creating significant safety hazards that complicate logistics and increase insurance liabilities for production facilities. Alternatively, the haloform reaction pathway starting from 5-chloro-2-acetyl thiophene suffers from poor atom economy, generating substantial volumes of chlorinated wastewater and hazardous byproducts that strain environmental treatment systems. These legacy methods collectively result in lower overall yields, higher raw material costs, and complex purification workflows that hinder the reliable supply of this critical pharmaceutical intermediate.
The Novel Approach
The patented methodology outlined in CN102993165A circumvents these historical deficiencies by employing a direct formylation-oxidation strategy that operates under significantly milder conditions. The process initiates with the Vilsmeier-Haack reaction, utilizing readily available 2-chlorothiophene, phosphorus oxychloride, and DMF to construct the aldehyde functionality with high precision. Subsequent oxidation is achieved using dilute nitric acid catalyzed by sodium nitrite, eliminating the need for heavy metal oxidants or dangerous peracids. This approach not only simplifies the reaction setup to standard glass-lined reactors but also dramatically reduces the thermal load on the facility, as the exotherms are manageable within a 30-50°C range. The transition from hazardous lithiation to this solution-phase chemistry represents a paradigm shift in process safety, enabling manufacturers to achieve consistent quality without compromising operator safety or environmental standards.
Mechanistic Insights into Vilsmeier-Haack Formylation and Nitrite-Catalyzed Oxidation
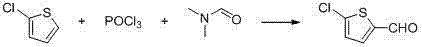
The first stage of this synthesis leverages the electrophilic aromatic substitution mechanism characteristic of the Vilsmeier-Haack reaction. In this process, phosphorus oxychloride reacts with N,N-dimethylformamide (DMF) to generate the highly reactive chloroiminium ion species, which acts as the active electrophile. This electrophile attacks the electron-rich 2-position of the 2-chlorothiophene ring, driven by the resonance stabilization of the thiophene heterocycle and the directing effects of the chlorine substituent. The reaction proceeds through a stable intermediate complex which, upon hydrolysis during the workup phase, releases the desired 5-chlorothiophene-2-aldehyde. The stoichiometry is carefully balanced, typically employing a molar ratio of 2-chlorothiophene to POCl3 to DMF of approximately 1:1.5:1.5, ensuring complete conversion while minimizing the formation of poly-formylated byproducts or tar.
Following the isolation of the aldehyde, the second stage involves a selective oxidation mechanism catalyzed by sodium nitrite in the presence of dilute nitric acid. The nitrite ion serves as a catalyst to generate reactive nitrogen oxide species in situ, which facilitate the transfer of oxygen to the aldehyde carbonyl group, converting it efficiently into the carboxylic acid moiety. This oxidation is highly chemoselective, preserving the sensitive thiophene ring and the chloro-substituent from oxidative degradation or dehalogenation, which are common failure modes in harsher oxidation protocols. The reaction temperature is strictly maintained between 30°C and 40°C to control the rate of gas evolution and prevent runaway exotherms, ensuring a smooth conversion profile. This mechanistic elegance allows for the direct crystallization of the crude acid, which can be further purified to pharmaceutical grade specifications through simple ethanol recrystallization.
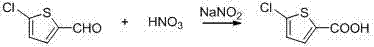
How to Synthesize 5-Chlorothiophene-2-Formic Acid Efficiently
Implementing this synthesis route requires precise control over addition rates and temperature gradients to maximize yield and safety. The process begins with the controlled addition of phosphorus oxychloride to a mixture of 2-chlorothiophene and DMF, maintaining the internal temperature between 40°C and 50°C to drive the formylation to completion over a 4-hour period. After quenching with water and extracting with dichloromethane, the organic layer is washed and distilled to isolate the aldehyde intermediate with purity exceeding 99%. The subsequent oxidation step involves dissolving the aldehyde in water with sodium nitrite, heating to 40°C, and slowly dripping in 60-65% nitric acid while keeping the temperature below 40°C. Detailed standardized operating procedures regarding stirring speeds, specific addition times, and crystallization parameters are essential for reproducibility.
- Perform Vilsmeier-Haack formylation by reacting 2-chlorothiophene with DMF and POCl3 at 40-50°C to obtain 5-chlorothiophene-2-aldehyde.
- Oxidize the resulting aldehyde using dilute nitric acid (60-65%) catalyzed by sodium nitrite at 30-40°C.
- Isolate the crude product via suction filtration and purify through ethanol recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages beyond mere technical feasibility. By eliminating the dependency on n-Butyl Lithium, organizations can remove a high-risk, high-cost reagent from their supply chain, thereby stabilizing raw material sourcing and reducing inventory holding costs associated with hazardous material storage. The shift to commodity chemicals like phosphorus oxychloride and nitric acid ensures a robust supply base with multiple global vendors, mitigating the risk of single-source bottlenecks that frequently plague specialty reagent markets. Furthermore, the simplified workup procedure, which avoids complex chromatographic separations in favor of crystallization and distillation, significantly reduces solvent consumption and processing time, leading to substantial cost reductions in API manufacturing overheads.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling systems and expensive organometallic reagents directly translates to lower utility and material expenses. The process utilizes inexpensive, bulk-available starting materials, and the high selectivity minimizes the loss of valuable intermediates to side reactions, optimizing the overall mass balance and improving the cost-per-kilogram metric significantly.
- Enhanced Supply Chain Reliability: Operating under mild thermal conditions reduces equipment stress and maintenance downtime, ensuring consistent production schedules. The use of stable reagents simplifies logistics and transportation compliance, allowing for faster replenishment cycles and reducing the lead time for high-purity pharmaceutical intermediates required for just-in-time manufacturing models.
- Scalability and Environmental Compliance: The reduction in hazardous waste generation, particularly the avoidance of chlorinated byproducts from haloform reactions, streamlines wastewater treatment processes. This environmental efficiency facilitates easier regulatory approval for scale-up activities, enabling seamless transition from pilot plant to multi-ton commercial production without extensive retrofitting of effluent treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams evaluating process transfer and for quality assurance personnel establishing specification limits. The answers provided are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: Why is this synthesis method safer than traditional lithiation routes?
A: Traditional methods often utilize n-Butyl Lithium, which requires cryogenic conditions (-78°C) and poses severe pyrophoric hazards. This patented route operates at mild temperatures (30-50°C) using stable reagents like POCl3 and dilute nitric acid, drastically reducing operational risk.
Q: What is the expected purity and yield of the final product?
A: According to the embodiment data in patent CN102993165A, the process achieves a final purity of up to 99.6% after ethanol recrystallization, with an overall yield exceeding 85% for the oxidation step and over 90% for the formylation step.
Q: How does this method impact environmental compliance?
A: Unlike the haloform reaction which generates significant chlorinated waste and wastewater, this oxidative method utilizes dilute nitric acid and produces fewer three wastes, simplifying downstream effluent treatment and lowering environmental compliance costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chlorothiophene-2-Formic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel anticoagulants depends on the uninterrupted supply of high-quality intermediates like 5-chlorothiophene-2-formic acid. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for GMP pharmaceutical synthesis. Our commitment to process excellence means we can deliver this critical building block with the consistency and reliability necessary to support your global drug development timelines.
We invite you to collaborate with us to optimize your supply chain for cardiovascular therapeutics. By leveraging our expertise in this specific synthetic route, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven manufacturing capabilities. Let us help you secure a competitive advantage through superior chemical supply solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
