Advanced Halogen Exchange Strategy for High-Purity Fluoropyridazinone Manufacturing
The chemical landscape for producing fluorinated heterocycles has evolved significantly with the disclosure of patent CN1583727A, which introduces a robust and scalable method for the synthesis of fluoropyridazinone derivatives. As a critical structural motif found in numerous bioactive molecules, fluoropyridazinones serve as essential building blocks for both pharmaceutical agents, such as anticancer drugs resembling fluorouracil, and advanced agrochemical formulations. The traditional reliance on harsh fluorination techniques often resulted in complex impurity profiles and difficult separations, creating bottlenecks for R&D teams aiming for high-purity standards. This patented approach utilizes a nucleophilic halogen exchange strategy, replacing chlorine, bromine, or iodine atoms on the pyridazinone ring with fluorine using metal fluorides in aprotic polar solvents. By shifting away from extreme thermal conditions and non-selective reagents, this technology provides a reliable pathway for generating high-value intermediates with exceptional control over regioselectivity and product quality.
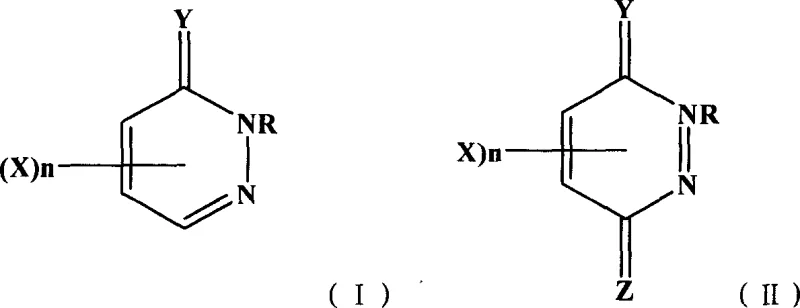
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of this halogen exchange methodology, the industry relied heavily on three primary techniques, each fraught with significant operational and chemical drawbacks that hindered efficient commercial scale-up. The melting method, while useful for generating polyfluorinated species, typically suffered from poor selectivity, often yielding mixtures of perfluorinated byproducts that were extremely difficult to separate into single, pure fluorinated isomers required for drug synthesis. Furthermore, the diazotization method, although capable of introducing fluorine with good specificity, was limited by a narrow scope of application and the instability of diazonium intermediates, making it unsuitable for large-scale industrial production where safety and consistency are paramount. Additionally, condensation methods using hydrocarbon fluorides were constrained by the limited availability and high cost of specialized fluorinated starting materials, creating supply chain vulnerabilities. These legacy processes often demanded excessive energy inputs and generated substantial waste streams, complicating environmental compliance and driving up the overall cost of goods for procurement managers seeking economical sourcing solutions.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally transforms the synthesis landscape by employing a direct nucleophilic substitution of halogens (Cl, Br, I) with fluoride ions under mild, controlled conditions. By utilizing readily available halogenated pyridazinone precursors and reacting them with inexpensive alkali metal fluorides in solvents like dimethyl sulfoxide or acetone, the process achieves high conversion rates without the need for exotic catalysts or extreme temperatures. This method allows for precise tuning of reaction parameters, such as temperature ranges between 30°C and 100°C, ensuring that the delicate pyridazinone ring structure remains intact while selectively installing the fluorine atom. The versatility of this approach is evident in its ability to accommodate various substituents on the ring, including phenyl and alkyl groups, thereby enabling the production of a diverse library of fluorinated derivatives. For supply chain leaders, this translates to a more resilient manufacturing process that relies on commodity chemicals rather than scarce, high-cost reagents, effectively de-risking the procurement of these critical intermediates.
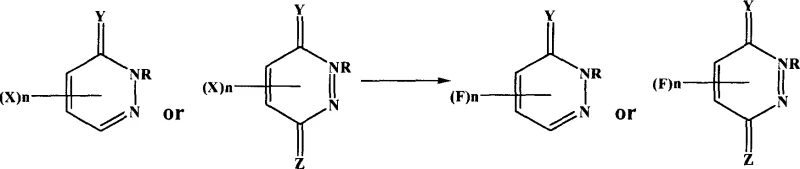
Mechanistic Insights into Metal Fluoride-Mediated Halogen Exchange
The core of this synthetic breakthrough lies in the mechanism of nucleophilic aromatic substitution, facilitated by the unique properties of aprotic polar solvents. In this system, the metal fluoride, particularly potassium fluoride, acts as the source of nucleophilic fluoride ions. The choice of solvent is critical; aprotic polar solvents such as DMF, DMSO, or acetonitrile effectively solvate the metal cation (e.g., K+) while leaving the fluoride anion relatively "naked" and highly reactive. This enhanced nucleophilicity allows the fluoride ion to attack the electron-deficient carbon atom on the pyridazinone ring bearing the leaving group (chlorine, bromine, or iodine). The reaction proceeds through a transition state where the C-X bond breaks concurrently with the formation of the C-F bond, driven by the high lattice energy of the resulting metal halide salt and the strength of the carbon-fluorine bond. Understanding this mechanistic nuance is vital for R&D directors, as it explains why the process avoids the formation of polyfluorinated side products common in radical-based or high-temperature thermal methods, ensuring a cleaner reaction profile.
Impurity control in this process is inherently managed through the selection of reaction conditions and the subsequent workup procedure. Since the reaction does not involve unstable diazonium salts or aggressive fluorinating gases, the generation of hazardous or hard-to-remove byproducts is minimized. The patent describes a straightforward purification protocol where the reaction solvent is recovered via distillation, and the crude product is isolated by filtration after dissolution in warm water. This phase separation exploits the differential solubility of the organic fluoropyridazinone product versus inorganic salts and polar impurities. Further purification via column chromatography or recrystallization from ethanol-water mixtures yields a white crystalline solid of high purity. This robustness in impurity management ensures that the final API intermediate meets stringent quality specifications, reducing the burden on analytical teams and minimizing the risk of batch failures during commercial production runs.
How to Synthesize Fluoropyridazinone Efficiently
The synthesis of fluoropyridazinone via this patented route involves a streamlined sequence of mixing, heating, and isolation steps that are amenable to standard chemical processing equipment. The process begins with the suspension of the halogenated precursor and the metal fluoride salt in the chosen aprotic solvent, followed by heating to initiate the substitution reaction. Detailed operational parameters, including specific molar ratios and temperature ramps, are critical for maximizing yield and minimizing side reactions. For laboratory and pilot plant chemists looking to implement this technology, adherence to the standardized protocol ensures reproducibility and safety. The detailed standardized synthesis steps are provided in the guide below.
- Mix halogenated pyridazinone compounds with alkali metal fluorides (such as potassium fluoride) in an aprotic polar solvent like DMSO or acetone.
- Heat the reaction mixture to temperatures between 30°C and 100°C and stir for 3 to 10 hours to facilitate the nucleophilic substitution.
- Recover the solvent via distillation, dissolve the residue in warm water, filter the insoluble organic solid, and purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers profound advantages that directly address the pain points of cost, reliability, and scalability in the fine chemical sector. By replacing expensive and hazardous fluorinating agents with commodity metal fluorides like potassium fluoride, the raw material costs are drastically simplified and reduced. The ability to recover and recycle the aprotic polar solvents further enhances the economic viability of the process, as solvent purchase and disposal represent significant portions of operating expenses in batch manufacturing. For procurement managers, this means a more stable pricing structure for fluoropyridazinone intermediates, insulated from the volatility of specialized reagent markets. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, contributing to a lower overall cost of manufacturing without compromising on the quality or purity of the final product.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of inexpensive, widely available starting materials drive significant cost efficiencies. Unlike methods requiring custom-synthesized fluorinated building blocks, this route utilizes generic halogenated precursors that are easily sourced from the global supply chain. The solvent recovery loop creates a closed-loop system that minimizes waste disposal fees and reduces the volume of fresh solvent required per kilogram of product. These factors combine to create a leaner manufacturing process that delivers substantial cost savings, allowing buyers to negotiate more competitive terms for their active pharmaceutical ingredient supply chains.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents such as alkali metal fluorides ensures a consistent and uninterrupted supply of raw materials. Traditional methods involving diazotization often face bottlenecks due to the short shelf-life of intermediates and safety restrictions on scaling up hazardous reactions. In contrast, this halogen exchange process is robust and forgiving, capable of being scaled from kilogram to multi-ton quantities without fundamental changes to the chemistry. This scalability assures supply chain heads of long-term continuity, mitigating the risk of production stoppages caused by reagent shortages or regulatory hurdles associated with more dangerous chemical processes.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing waste generation and avoiding the use of toxic heavy metal catalysts. The simplicity of the workup procedure, involving filtration and recrystallization, reduces the need for complex extraction protocols that generate large volumes of aqueous waste. This ease of handling facilitates smoother regulatory approvals and environmental audits, which are critical for maintaining operational licenses in strict jurisdictions. The ability to run the reaction at moderate temperatures also lowers the facility's energy footprint, aligning with corporate sustainability goals and reducing the carbon intensity of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluoropyridazinone synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope, reagent selection, and process optimization. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific project needs.
Q: What are the advantages of this halogen exchange method over traditional melting methods?
A: Unlike traditional melting methods which often result in perfluorinated products with poor selectivity and require high temperatures, this patented halogen exchange method operates under milder conditions (30-100°C) and offers superior selectivity for mono-fluorinated products, significantly simplifying downstream purification.
Q: Which metal fluorides are most effective for this synthesis?
A: The patent specifies that alkali metal fluorides are particularly effective, with potassium fluoride (KF) being the most preferred reagent due to its balance of reactivity and cost-effectiveness, although sodium fluoride and cesium fluoride are also viable options depending on the specific substrate.
Q: Can the solvents used in this process be recycled?
A: Yes, a key economic advantage of this process is the use of aprotic polar solvents like dimethyl sulfoxide or acetone, which can be recovered through reduced pressure distillation after the reaction, allowing for循环利用 (recycling) and substantially reducing raw material costs and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoropyridazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality fluorinated intermediates in the development of next-generation therapeutics and crop protection agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering fluoropyridazinone derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the advanced halogen exchange methodology described in patent CN1583727A, we can offer our partners a superior product profile with improved cost-efficiency and supply security.
We invite global pharmaceutical and agrochemical companies to collaborate with us to optimize their supply chains for fluorinated building blocks. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate our commitment to excellence in fine chemical manufacturing.
