Scalable Synthesis of 2-Bromo-5-Fluorobenzotrifluoride for High-Purity Pharmaceutical Intermediates
The chemical landscape for fluorinated aromatic intermediates is constantly evolving, driven by the need for more robust and scalable manufacturing processes. Patent CN102951996A introduces a significant advancement in the synthesis of 2-bromo-5-fluorobenzotrifluoride, a critical building block for various pharmaceutical applications. This technology departs from conventional reliance on exotic reagents, establishing a streamlined three-step pathway that begins with the nitration of m-fluorobenzotrifluoride. By leveraging a classic nitration-reduction-diazotization sequence, the method achieves exceptional regioselectivity and product integrity. For R&D directors and process chemists, this represents a viable alternative that balances high yield with operational simplicity, ensuring that the supply of this key intermediate remains stable and economically feasible for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct bromination of fluorinated benzotrifluorides has posed significant logistical and economic challenges for industrial manufacturers. Traditional protocols often depend on specialized brominating agents, such as 1,3-dibromo-2,4-imidazoledione, which are not only difficult to source in bulk quantities but also command premium pricing due to their niche status. These reagents introduce substantial supply chain vulnerabilities, as their production is limited to a small number of global suppliers, creating bottlenecks for large-scale API manufacturing. Furthermore, the handling of such specialized oxidants often requires stringent safety measures and specific waste treatment protocols, adding layers of complexity and cost to the overall production workflow. The scarcity of these reagents makes long-term planning difficult for procurement teams aiming to secure stable raw material streams for multi-ton campaigns.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a strategic sequence of nitration, catalytic hydrogenation, and Sandmeyer bromination to bypass the need for rare reagents entirely. This approach relies exclusively on commodity chemicals like nitric acid, sulfuric acid, and cuprous bromide, which are readily available in the global chemical market at competitive prices. The process demonstrates remarkable efficiency, converting m-fluorobenzotrifluoride into the target molecule through well-understood unit operations that are easily adaptable to existing reactor infrastructure. By shifting the synthetic logic to a stepwise functional group transformation, the method ensures precise control over the substitution pattern, avoiding the formation of difficult-to-separate isomers that often plague direct halogenation routes. This transition to common reagents fundamentally de-risks the manufacturing process, offering a sustainable path forward for commercial production.
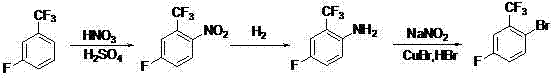
Mechanistic Insights into Nitration-Reduction-Bromination Sequence
The core of this synthetic strategy lies in the precise control of electrophilic aromatic substitution and subsequent functional group interconversions. The initial nitration step utilizes a mixed acid system where the interplay between the electron-withdrawing trifluoromethyl group and the fluorine atom dictates the regiochemistry. Despite the deactivating nature of the CF3 group, the reaction conditions—specifically maintaining temperatures below 25°C during acid addition—favor the formation of the 2-nitro-5-fluoro isomer with high specificity. This thermal control is critical for suppressing poly-nitration and minimizing oxidative side reactions, ensuring that the crude intermediate is of sufficient quality for subsequent processing without extensive chromatographic purification. The subsequent isolation via recrystallization further refines the material, setting a high baseline for the final product purity.
Following nitration, the reduction of the nitro group to an amine is achieved through catalytic hydrogenation, a cornerstone of green chemistry in fine chemical synthesis. Using Raney Nickel or Palladium on Carbon under moderate hydrogen pressure (0.5 MPa) allows for the clean conversion of the nitro functionality while preserving the sensitive carbon-fluorine and carbon-trifluoromethyl bonds. The choice of solvent, typically ethanol or similar alcohols, facilitates efficient mass transfer and heat dissipation during this exothermic reaction. The final transformation involves a classic diazotization followed by a Sandmeyer reaction, where the in situ generated diazonium salt is displaced by a bromide ion in the presence of cuprous bromide. This step is particularly robust, tolerating the harsh acidic conditions required for diazotization while delivering the bromine atom precisely at the ortho position relative to the trifluoromethyl group.
How to Synthesize 2-Bromo-5-Fluorobenzotrifluoride Efficiently
Executing this synthesis on an industrial scale requires careful attention to thermal management and stoichiometry, particularly during the exothermic nitration and diazotization phases. The patent outlines a reproducible protocol that balances reaction kinetics with safety, ensuring that the process can be scaled from kilogram to multi-ton batches without loss of fidelity. Operators must strictly adhere to the temperature profiles specified, especially the cooling requirements during acid addition and diazonium formation, to prevent runaway reactions and ensure operator safety. The detailed standardized synthesis steps provided below offer a comprehensive guide for process engineers looking to implement this route in their facilities, highlighting critical control points for quality assurance.
- Nitration of m-fluorobenzotrifluoride using a mixed acid system (HNO3/H2SO4) at controlled low temperatures to form 5-fluoro-2-nitrobenzotrifluoride.
- Catalytic hydrogenation of the nitro intermediate using Raney Nickel or Pd/C under mild pressure (0.5 MPa) to yield the corresponding aniline derivative.
- Diazotization of the amine with sodium nitrite and hydrobromic acid, followed by Sandmeyer bromination using cuprous bromide to finalize the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the shift towards this synthetic route offers profound strategic benefits that extend beyond simple unit cost calculations. By eliminating the dependency on proprietary or scarce brominating agents, manufacturers can insulate their supply chains from market volatility and geopolitical disruptions that often affect specialty chemical sectors. The use of ubiquitous raw materials means that sourcing can be diversified across multiple vendors, fostering competition and driving down input costs significantly. Furthermore, the ability to recycle the hydrogenation catalyst adds another layer of economic efficiency, reducing the consumption of precious metals and lowering the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only reduces waste disposal costs but also enhances the sustainability profile of the final pharmaceutical product.
- Cost Reduction in Manufacturing: The replacement of expensive, specialized brominating reagents with commodity acids and salts results in a drastic reduction in raw material expenditure. Since the process avoids complex purification steps associated with removing exotic by-products, the overall operational expenditure is lowered, allowing for more competitive pricing in the global market. The elimination of transition metal scavengers, often required when using palladium-catalyzed direct coupling, further streamlines the downstream processing costs.
- Enhanced Supply Chain Reliability: Sourcing risk is minimized because every reagent in this three-step sequence is a high-volume industrial chemical with a stable global supply network. This reliability ensures that production schedules are not held hostage by the lead times of niche reagent suppliers, guaranteeing consistent delivery of the intermediate to API manufacturers. The robustness of the chemistry also means that batch-to-batch variability is low, reducing the incidence of failed batches that can disrupt supply continuity.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-atmospheric or low pressures, making the process inherently safer and easier to scale in standard glass-lined or stainless steel reactors. The waste streams generated are primarily inorganic salts and aqueous acids, which are easier to treat and neutralize compared to the complex organic waste from specialized reagent synthesis. This simplifies regulatory compliance and reduces the burden on wastewater treatment facilities, supporting long-term environmental sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and quality standards associated with this manufacturing route. Understanding these details is crucial for technical teams evaluating the integration of this intermediate into their broader drug substance supply chains.
Q: What are the key advantages of this synthesis route over traditional bromination methods?
A: Unlike traditional methods relying on specialized and scarce brominating agents like 1,3-dibromo-2,4-imidazoledione, this process utilizes common industrial chemicals such as nitric acid, sulfuric acid, and cuprous bromide. This significantly enhances raw material availability and reduces procurement risks for large-scale manufacturing.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The process incorporates rigorous purification steps, including recrystallization of the nitro intermediate and vacuum distillation of the final product. The specific reaction conditions, particularly the controlled temperature during nitration and the selective catalytic reduction, minimize by-product formation, consistently achieving purity levels greater than 98%.
Q: Is the catalyst system suitable for continuous industrial production?
A: Yes, the hydrogenation step employs Raney Nickel or Palladium on Carbon, both of which are robust catalysts suitable for high-pressure autoclave operations. The patent highlights that the catalyst can be recovered and recycled, which supports sustainable and cost-effective continuous processing capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-5-Fluorobenzotrifluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new pharmaceutical therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of 2-bromo-5-fluorobenzotrifluoride meets the exacting standards required for GMP manufacturing. Our facility is equipped to handle the specific thermal and pressure conditions of this nitration-hydrogenation sequence safely and efficiently.
We invite you to collaborate with us to optimize your supply chain for this essential building block. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and commercial production goals effectively.
