Advanced Oxidative Bromination for High-Purity 2-Bromo-5-Fluorobenzotrifluoride Commercial Production
Advanced Oxidative Bromination for High-Purity 2-Bromo-5-Fluorobenzotrifluoride Commercial Production
The global demand for fluorinated aromatic intermediates has surged in recent years, driven by their critical role in the development of next-generation pharmaceuticals and agrochemicals. Patent CN114163294A introduces a groundbreaking preparation method for 2-bromo-5-fluorobenzotrifluoride, a key building block characterized by its unique substitution pattern that enables diverse downstream functionalization. This technology represents a significant leap forward in organic synthesis, shifting away from traditional, waste-intensive nitration-reduction sequences towards a more sustainable, one-pot oxidative bromination strategy. By leveraging a novel quaternary ammonium salt composite catalyst system, the process achieves exceptional yields exceeding 92 percent and product purity levels greater than 99 percent, effectively meeting the stringent quality specifications required by top-tier multinational corporations. The innovation lies not only in the chemical transformation but also in the holistic optimization of the reaction engineering, which eliminates the need for specialized, expensive brominating reagents and simplifies the post-reaction workup.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromo-5-fluorobenzotrifluoride has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex fluorinated aromatics. Traditional routes often rely on multi-step sequences involving nitration, reduction, diazotization, and halogen exchange, which inherently suffer from low overall yields due to cumulative losses at each stage. Furthermore, these methods generate substantial quantities of acidic wastewater and heavy metal contaminants, creating severe environmental compliance challenges and escalating three-waste treatment costs. Alternative direct bromination strategies utilizing reagents like 1,3-dibromo-5,5-dimethylhydantoin (DBH) offer better selectivity but are economically prohibitive due to the high cost of the brominating agent and the difficulty in recovering the hydantoin byproduct. Other approaches involving mixed acid systems result in complex reaction mixtures that are notoriously difficult to separate, requiring energy-intensive purification steps that erode profit margins and extend production lead times.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN114163294A utilizes a streamlined, atom-economical approach that directly brominates m-fluorobenzotrifluoride using elemental bromine regenerated in situ by hydrogen peroxide. This strategy circumvents the need for expensive specialty reagents by employing commodity chemicals that are readily available in the global supply chain, thereby drastically simplifying procurement logistics. The reaction proceeds in a single pot under mild thermal conditions, typically between 40-60°C, which eliminates the safety risks associated with exothermic runaway reactions often seen in traditional nitration processes. By integrating a specific quaternary ammonium salt composite catalyst, the system achieves precise control over regioselectivity, favoring the formation of the desired 2-bromo isomer while suppressing the formation of dibrominated side products. This results in a cleaner crude reaction profile that significantly reduces the burden on downstream purification units, allowing for high-purity isolation through simple distillation.
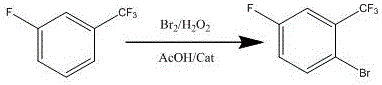
Mechanistic Insights into Quaternary Ammonium Salt Catalyzed Oxidative Bromination
The core of this technological breakthrough lies in the sophisticated interplay between the bromine source, the oxidant, and the phase-transfer-like catalyst. In this system, elemental bromine acts as the primary electrophile, attacking the electron-rich aromatic ring of m-fluorobenzotrifluoride to form the sigma complex. As the reaction progresses, hydrogen bromide (HBr) is generated as a stoichiometric byproduct, which would traditionally terminate the reaction or require neutralization. However, the presence of hydrogen peroxide serves a critical function by oxidizing the accumulated HBr back into active molecular bromine (Br2), effectively recycling the bromine atoms and driving the equilibrium towards completion. This oxidative regeneration loop ensures that the molar ratio of bromine to substrate can be kept close to stoichiometric, minimizing the consumption of raw materials and reducing the load on waste treatment facilities. The mild reaction conditions prevent the decomposition of the sensitive trifluoromethyl group, preserving the integrity of the fluorinated scaffold essential for biological activity in final drug products.
Furthermore, the inclusion of the quaternary ammonium salt composite catalyst, specifically a mixture of N-bromoacetamide and phenyl trimethyl amine tribromide, plays a pivotal role in enhancing reaction kinetics and selectivity. These catalysts likely function by stabilizing the transition state of the electrophilic attack or by facilitating the transport of reactive bromine species within the acetic acid solvent matrix. The polar nature of glacial acetic acid is crucial, as it effectively solvates the hydrogen bromide intermediate, preventing its premature escape as a gas and keeping it available for oxidation by hydrogen peroxide. This synergistic effect between the solvent, the oxidant, and the catalyst creates a highly controlled chemical environment that suppresses radical side reactions and poly-bromination. Consequently, the impurity profile of the final product is remarkably clean, with gas chromatography analysis consistently showing main peak areas above 99 percent, which is vital for reducing lead time for high-purity building blocks in regulated industries.
How to Synthesize 2-Bromo-5-Fluorobenzotrifluoride Efficiently
The operational protocol for this synthesis is designed for robustness and ease of execution in a standard multipurpose chemical plant. The process begins with the preparation of the reaction mixture by combining glacial acetic acid and the starting material, m-fluorotrifluorotoluene, at ambient temperature. A precise amount of the composite catalyst is introduced before the system is gently heated to the target reaction window. The addition of bromine is conducted in a controlled, dropwise manner to manage the exotherm, followed by the sequential addition of hydrogen peroxide to sustain the bromination cycle. Detailed standardized synthetic steps see the guide below.
- Mix acetic acid and m-fluorotrifluorotoluene, add quaternary ammonium salt composite catalyst, heat to 40-60°C, and dropwise add bromine.
- After bromine addition, maintain temperature and dropwise add hydrogen peroxide to oxidize generated hydrogen bromide for continued reaction.
- Maintain reaction until raw material residue is <0.5%, quench excess oxidant with sodium sulfite, remove solvent, and distill to collect fractions at 158-162°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers transformative benefits that extend far beyond simple yield improvements. The shift from specialized, high-cost brominating agents to commodity-grade bromine and hydrogen peroxide fundamentally alters the cost structure of the manufacturing process. Since hydrogen peroxide and acetic acid are produced on a massive global scale, their prices are stable and predictable, shielding the production budget from the volatility often associated with niche fine chemical reagents. Moreover, the elimination of heavy metal catalysts and complex nitration steps removes the need for expensive wastewater treatment protocols and hazardous waste disposal services, resulting in substantial cost savings in environmental compliance. The simplified one-pot nature of the reaction also reduces the requirement for multiple reactor vessels and intermediate storage tanks, optimizing capital expenditure and facility utilization rates.
- Cost Reduction in Manufacturing: The economic viability of this route is underpinned by the drastic reduction in raw material costs and processing complexity. By avoiding the use of expensive reagents like dibromohydantoin or specialized diazotization salts, the direct material cost per kilogram of product is significantly lowered. Additionally, the ability to recycle the acetic acid solvent through simple distillation further enhances the process economics, as solvent recovery rates are high and the solvent quality remains sufficient for reuse. The high selectivity of the reaction minimizes the loss of valuable fluorinated starting material to side products, ensuring that the maximum amount of input mass is converted into saleable product. This efficiency translates directly into improved gross margins for manufacturers operating in competitive markets where price pressure is intense.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on widely available commodity chemicals mitigates the risk of production stoppages due to raw material shortages. Unlike proprietary reagents that may have single-source suppliers and long lead times, bromine and hydrogen peroxide are accessible from a broad network of global chemical distributors. This diversification of the supply base ensures business continuity and allows for more flexible inventory management strategies. Furthermore, the mild reaction conditions reduce the wear and tear on production equipment, leading to lower maintenance downtime and higher overall equipment effectiveness (OEE). The robustness of the process also means that it can be easily transferred between different manufacturing sites without extensive re-validation, providing strategic flexibility in global sourcing.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly smaller than that of conventional routes, aligning with the increasing regulatory pressures and corporate sustainability goals of modern chemical enterprises. The absence of heavy metals and the reduction in acidic waste streams simplify the permitting process for new production lines and reduce the liability associated with long-term environmental monitoring. The process is inherently safer, operating at atmospheric pressure and moderate temperatures, which lowers the insurance premiums and safety infrastructure costs required for operation. Scalability is straightforward, as the reaction kinetics are not limited by mass transfer issues common in heterogeneous systems, allowing for seamless translation from pilot plant batches to multi-ton commercial production campaigns without loss of yield or purity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative bromination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their specific supply chains.
Q: What are the advantages of using hydrogen peroxide in this bromination process?
A: Hydrogen peroxide acts as a green oxidant that regenerates bromine from the hydrogen bromide byproduct, significantly improving atom economy and reducing hazardous waste compared to stoichiometric brominating agents.
Q: How does the quaternary ammonium salt catalyst improve the reaction?
A: The composite catalyst enhances the electrophilicity of the brominating species and improves regioselectivity, ensuring high conversion to the 2-bromo isomer while minimizing poly-brominated impurities.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild temperatures (40-60°C) without requiring cryogenic conditions or high-pressure equipment, making it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-5-Fluorobenzotrifluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a laboratory recipe; it demands a partner with deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We understand the critical nature of stringent purity specifications in the pharmaceutical and agrochemical sectors, which is why our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. By leveraging the innovative oxidative bromination technology described in patent CN114163294A, we can offer a supply solution that balances cost-effectiveness with uncompromising quality.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Whether you are looking to secure a long-term supply agreement or need assistance with custom process development, we are prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical capabilities can drive value and reliability in your supply chain.
