Advanced Enzymatic Synthesis of Droxidopa: A Scalable Route for Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in patent CN109811019B, which discloses a novel method for preparing Droxidopa (L-DOPS), a vital therapeutic agent used in the treatment of Parkinson's disease and neurogenic orthostatic hypotension. This patent introduces a biocatalytic approach that utilizes 3,4-dihydroxybenzaldehyde and L-threonine as starting materials, catalyzed by transaldolase. Unlike traditional chemical syntheses that rely on harsh conditions and multiple protection steps, this enzymatic route offers a streamlined, high-yield alternative that addresses long-standing challenges in stereoselectivity and process complexity. For global procurement teams and R&D directors, this technology represents a pivotal shift towards greener, more cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Droxidopa has been plagued by inefficient multi-step chemical syntheses. As illustrated in the prior art, conventional routes typically start with 3,4-dihydroxybenzaldehyde but require extensive benzyl protection and deprotection strategies to manage the reactive hydroxyl groups. These chemical pathways involve chiral resolution steps that inherently limit the maximum theoretical yield to below 50%, and in practice, overall yields often fall below 20% due to cumulative losses across numerous reaction stages. Furthermore, the use of heavy metal catalysts and hazardous organic solvents generates significant waste streams, complicating environmental compliance and driving up disposal costs. The structural complexity necessitates rigorous purification protocols, which further erodes profit margins and extends production lead times, making these legacy methods increasingly untenable for modern, high-volume supply chains.
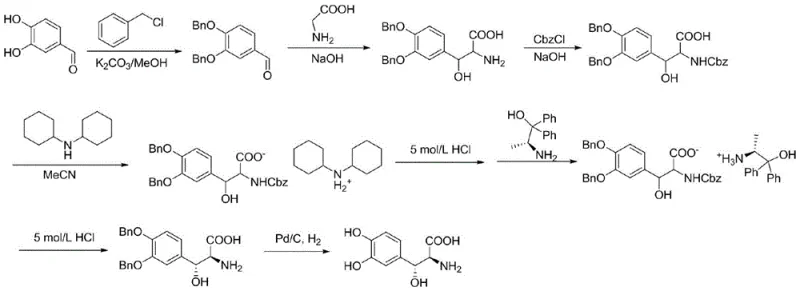
Even earlier biological attempts to synthesize Droxidopa faced substantial hurdles. Previous enzymatic methods utilized glycine as a substrate catalyzed by L-threonine aldolase. While this approach shortened the synthetic route, it suffered from poor stereoselectivity, resulting in a mixture of diastereomers that were difficult and expensive to separate. The low conversion rates and high consumption of glycine rendered these processes economically unviable for large-scale commercialization. The inability to control the stereochemistry at the beta-hydroxy position meant that significant resources were wasted on downstream purification, failing to meet the stringent purity requirements demanded by regulatory bodies for anti-Parkinson medications.
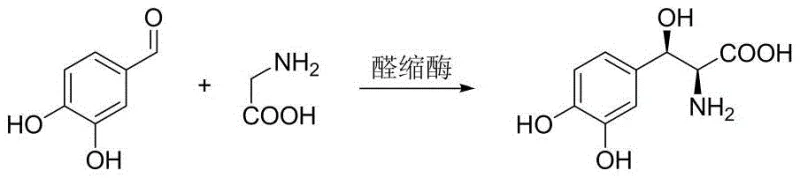
The Novel Approach
The methodology described in CN109811019B fundamentally overcomes these limitations by employing L-threonine as the nucleophilic donor in a transaldolase-catalyzed condensation with 3,4-dihydroxybenzaldehyde. This strategic choice of substrate leverages the existing chirality of L-threonine to induce high stereoselectivity directly during the bond-forming step. The reaction proceeds under mild aqueous conditions, eliminating the need for toxic organic solvents and complex protecting group chemistry. By bypassing the protection-deprotection cycle entirely, the atom economy is drastically improved, and the number of unit operations is significantly reduced. This results in a much shorter process flow that not only enhances the overall yield but also simplifies the isolation of the final product, offering a robust solution for the reliable supply of high-purity pharmaceutical intermediates.

Mechanistic Insights into Transaldolase-Catalyzed Condensation
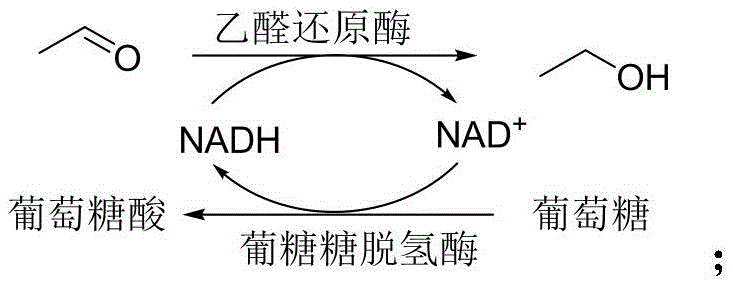
The core of this innovative process lies in the precise mechanistic action of the transaldolase enzyme, which facilitates the carbon-carbon bond formation between the aldehyde and the amino acid. However, a critical discovery detailed in the patent is the management of reaction byproducts to maintain enzymatic efficiency. During the cleavage of L-threonine, the system releases a molecule of acetaldehyde. In standard enzymatic reactions, the accumulation of acetaldehyde can act as a potent inhibitor, leading to the gradual inactivation of the transaldolase enzyme and stalling the reaction before completion. To counteract this, the inventors introduced a coupled enzyme system featuring acetaldehyde reductase. This auxiliary enzyme rapidly reduces the generated acetaldehyde into ethanol, effectively removing the inhibitory species from the reaction equilibrium and driving the primary condensation forward to near-quantitative conversion.
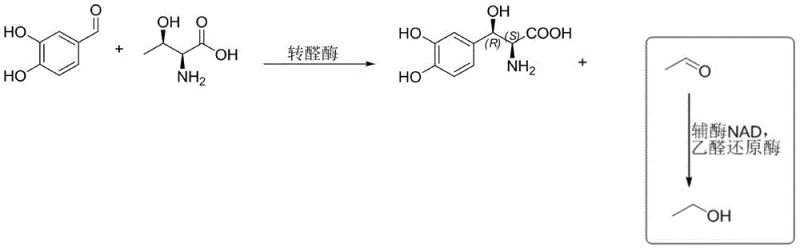
To ensure the economic feasibility of this coupled system, the process incorporates an efficient cofactor regeneration cycle. The reduction of acetaldehyde requires NADH, which would otherwise be a prohibitively expensive stoichiometric reagent. The patent outlines several strategies for regenerating NAD+ back to NADH, such as using glucose dehydrogenase with glucose or alcohol dehydrogenase with isopropanol. This creates a closed catalytic loop where the expensive cofactor is recycled continuously, minimizing raw material costs. This sophisticated engineering of the biocatalytic cascade ensures that the reaction remains vigorous over extended periods, achieving conversion rates exceeding 98% with excellent diastereomeric ratios (dr > 96:4) and enantiomeric excess (ee > 96%), which are critical metrics for R&D directors evaluating process viability.
How to Synthesize Droxidopa Efficiently
The implementation of this transaldolase-mediated pathway requires careful control of reaction parameters to maximize the benefits of the enzymatic cascade. The process begins with the preparation of a buffered aqueous system containing the substrates and essential co-factors like pyridoxal phosphate. Maintaining an inert atmosphere is crucial to prevent the oxidation of the sensitive 3,4-dihydroxybenzaldehyde substrate. The addition of antioxidants, such as sodium bisulfite, further stabilizes the reaction mixture. Following the enzymatic conversion, the workup procedure is remarkably simple compared to chemical synthesis, involving pH adjustment to precipitate proteins and subsequent crystallization. For a detailed, step-by-step technical guide on optimizing these parameters for your specific production scale, please refer to the standardized protocol below.
- Prepare the reaction system by mixing 3,4-dihydroxybenzaldehyde, L-threonine, pyridoxal phosphate, and phosphate buffer under an inert atmosphere.
- Add transaldolase enzyme and optionally acetaldehyde reductase with a cofactor regeneration system (e.g., glucose dehydrogenase) to drive the reaction.
- After reaction completion, adjust pH to precipitate proteins, concentrate the filtrate, and purify the product via crystallization to obtain high-purity Droxidopa.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic methodology offers profound strategic advantages beyond mere technical elegance. The elimination of benzyl protection groups and the associated harsh chemical reagents translates directly into a simplified supply chain with fewer raw material dependencies. By removing the need for complex chiral resolution steps, the process inherently reduces the risk of batch failures and variability, ensuring a more consistent and reliable supply of the intermediate. The mild reaction conditions (30-35°C) also imply lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and reduced utility costs, which aligns with the growing corporate mandates for sustainable manufacturing practices.
- Cost Reduction in Manufacturing: The most significant economic driver is the drastic reduction in processing steps. Traditional chemical routes involve multiple isolation and purification stages, each adding labor, time, and material costs. By consolidating the synthesis into a single enzymatic pot with in-situ byproduct removal, the operational expenditure is substantially lowered. Furthermore, the use of inexpensive, commodity-grade substrates like L-threonine and glucose, combined with the catalytic recycling of cofactors, minimizes the bill of materials. The avoidance of expensive chiral resolving agents and heavy metal catalysts removes entire cost centers from the P&L statement, allowing for a more competitive pricing structure in the final API market.
- Enhanced Supply Chain Reliability: Reliance on specialized, hazardous reagents often creates bottlenecks in the supply chain, subject to regulatory scrutiny and logistical delays. This biocatalytic route utilizes stable, non-hazardous enzymes and common buffer salts, which are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency. Additionally, the high stereoselectivity of the enzyme means that the crude product quality is superior, reducing the burden on downstream purification facilities and shortening the overall manufacturing cycle time. This agility allows for faster response to market demand fluctuations and tighter delivery schedules.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is generally more straightforward than scaling complex organic syntheses involving exothermic hazards. The aqueous nature of the reaction eliminates the risks associated with large volumes of flammable organic solvents, simplifying safety protocols and facility requirements. From an environmental perspective, the process generates significantly less hazardous waste, primarily consisting of biodegradable protein residues and ethanol. This ease of waste treatment reduces disposal costs and simplifies the permitting process for new manufacturing lines, facilitating rapid capacity expansion to meet commercial volume requirements without extensive environmental remediation investments.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new technology is essential for stakeholders evaluating its adoption. The following questions address common concerns regarding the technical robustness and commercial applicability of the transaldolase-based synthesis. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making regarding the integration of this route into existing production portfolios.
Q: What are the advantages of using L-threonine over glycine in Droxidopa synthesis?
A: Using L-threonine as a substrate with transaldolase provides superior stereoselectivity (high ee and dr values) compared to glycine-based routes, significantly reducing the difficulty of downstream purification and improving overall yield.
Q: How does the new method prevent enzyme inactivation during the reaction?
A: The process incorporates an acetaldehyde reductase system that converts the inhibitory byproduct acetaldehyde into ethanol, thereby maintaining high transaldolase activity and ensuring high conversion rates throughout the batch.
Q: Is this enzymatic route suitable for large-scale industrial production?
A: Yes, the method operates under mild conditions (30-35°C, aqueous buffer) without hazardous organic solvents or complex protection/deprotection steps, making it highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Droxidopa Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalytic technologies like the transaldolase-mediated synthesis of Droxidopa. As a premier CDMO partner, we possess the technical expertise to translate such innovative patent methodologies into robust, commercial-scale processes. Our facilities are equipped to handle complex enzymatic cascades, ensuring that the delicate balance of cofactor regeneration and substrate stability is maintained from pilot trials to full-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee product consistency.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and sustainable route for Droxidopa manufacturing. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this enzymatic approach can optimize your supply chain. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and to discuss how we can support your long-term sourcing strategies for high-quality pharmaceutical intermediates.
