Revolutionizing Droxidopa Production: A Deep Dive into Enzymatic Asymmetric Condensation
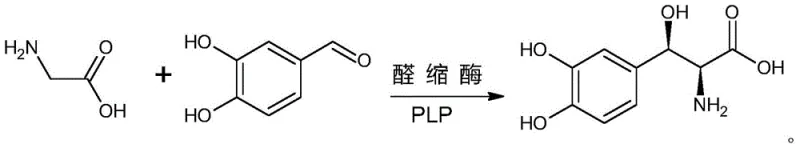
The global demand for high-purity anti-Parkinsonian agents continues to drive innovation in synthetic methodology, particularly for complex amino acid derivatives like Droxidopa. Patent CN112011580B introduces a groundbreaking biocatalytic approach that fundamentally shifts the paradigm from multi-step chemical synthesis to a streamlined enzymatic process. This technology leverages a specific aldolase enzyme to catalyze the asymmetric condensation of 3,4-dihydroxybenzaldehyde and glycine, offering a direct route to the target molecule with exceptional stereocontrol. For R&D directors and procurement strategists, this represents a significant opportunity to optimize supply chains for reliable pharmaceutical intermediates supplier networks. The method not only simplifies the operational workflow but also aligns with modern green chemistry principles by eliminating hazardous reagents and reducing waste generation. By adopting this enzymatic pathway, manufacturers can achieve superior cost reduction in pharmaceutical intermediates manufacturing while ensuring consistent quality standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis routes for Droxidopa have long been plagued by inherent inefficiencies and environmental burdens that hinder large-scale production viability. Conventional processes typically initiate with 3,4-dihydroxybenzaldehyde, which must first undergo benzyl protection to form 3,4-dibenzyloxybenzaldehyde before condensation with glycine can occur. This protection-deprotection strategy inevitably introduces multiple additional reaction steps, including chiral resolution and catalytic hydrogenation, which collectively cap the theoretical yield at merely 50%. Furthermore, the reliance on heavy metal catalysts for hydrogenation and the use of hazardous protecting group reagents create substantial disposal challenges and safety risks. The cumulative effect of these繁琐 steps results in poor atom economy and high production costs, making the traditional chemical route increasingly unsustainable in a regulatory environment that demands stricter environmental compliance and lower carbon footprints.
The Novel Approach
In stark contrast, the novel biocatalytic method disclosed in the patent utilizes a highly specific aldolase to facilitate a direct, one-step asymmetric condensation reaction. By employing 3,4-dihydroxybenzaldehyde and glycine as direct substrates without the need for hydroxyl protection, the process bypasses the cumbersome protection and deprotection stages entirely. This streamlined approach theoretically achieves 100% atom utilization, as every atom in the starting materials is incorporated into the final product structure without generating stoichiometric byproducts. The enzymatic catalyst exhibits remarkable chiral selectivity, directly yielding the desired (2S,3R) stereoisomer with high purity, thereby obviating the need for energy-intensive chiral resolution procedures. This transition to biocatalysis not only simplifies the process flow but also significantly enhances the overall economic feasibility and environmental sustainability of producing high-purity Droxidopa for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Aldolase-Catalyzed Asymmetric Condensation
The core of this technological breakthrough lies in the precise mechanistic action of the engineered aldolase enzyme, which functions as a highly efficient biocatalyst for carbon-carbon bond formation. The enzyme operates via a Pyridoxal 5'-phosphate (PLP) dependent mechanism, where the cofactor forms a Schiff base intermediate with the glycine substrate to generate a reactive quinonoid species. This activated nucleophile then attacks the carbonyl carbon of the 3,4-dihydroxybenzaldehyde electrophile with exquisite stereochemical control. The active site of the aldolase is structurally tuned to stabilize the transition state leading specifically to the (2S,3R) configuration, ensuring that the resulting beta-hydroxy-alpha-amino acid possesses the correct pharmacological activity. This level of enzymatic precision is difficult to replicate with small-molecule organocatalysts or transition metal complexes, highlighting the unique value of protein engineering in modern synthetic biology.
Beyond mere bond formation, the enzymatic system provides a robust mechanism for impurity control that is critical for pharmaceutical grade materials. The high diastereomeric excess (de > 99.5%) observed in experimental data indicates that the enzyme effectively suppresses the formation of unwanted stereoisomers such as the threo-form or enantiomers. In traditional chemical synthesis, minor variations in temperature or reagent purity can lead to significant racemization, necessitating costly purification steps like recrystallization or chromatography. However, the biological catalyst maintains its fidelity across a broad range of operating conditions, with optimal performance noted at 30°C and pH 7. This inherent selectivity reduces the burden on downstream processing units, allowing for simpler isolation protocols that preserve the integrity of the sensitive catechol moiety against oxidation. Consequently, the final product profile is cleaner, with a reduced risk of genotoxic impurities often associated with harsh chemical reagents.
How to Synthesize Droxidopa Efficiently
Implementing this biocatalytic route requires careful optimization of reaction parameters to maximize space-time yield and enzyme stability. The patent details a standardized protocol where recombinant E. coli cells expressing the aldolase gene are utilized as the biocatalyst source, either as wet cells or lyophilized powder. The reaction is conducted in a buffered aqueous medium, typically phosphate buffer at pH 7, which mimics physiological conditions favorable for enzyme activity. Substrate concentrations are balanced to prevent inhibition; for instance, maintaining 3,4-dihydroxybenzaldehyde at around 50g/L ensures a product titer of 14.4g/L within one hour without overwhelming the catalytic capacity. Detailed standardized synthesis steps follow below to guide process engineers in replicating these results.
- Prepare the reaction system by mixing 3,4-dihydroxybenzaldehyde and glycine substrates in a phosphate buffer solution adjusted to pH 7.
- Add the cofactor Pyridoxal 5'-phosphate (PLP) and the recombinant aldolase catalyst (expressed in E. coli) to the mixture.
- Maintain the reaction at 30°C with magnetic stirring for 1 hour, then separate the product to obtain Droxidopa with high diastereomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of protection groups and heavy metal catalysts fundamentally alters the cost structure of the manufacturing process, removing the need for expensive reagents like benzyl chloride and palladium on carbon. This simplification leads to substantial cost savings by reducing the number of unit operations, thereby lowering both capital expenditure on equipment and operational expenditure on labor and utilities. Furthermore, the aqueous nature of the reaction medium reduces the consumption of organic solvents, aligning with corporate sustainability goals and reducing the costs associated with solvent recovery and waste disposal. These factors collectively enhance the competitiveness of the supply chain for reliable Droxidopa supplier partnerships.
- Cost Reduction in Manufacturing: The transition to a one-step enzymatic process drastically simplifies the production workflow, removing the need for multiple isolation and purification stages associated with traditional chemical synthesis. By avoiding the use of precious metal catalysts and hazardous protecting groups, the raw material costs are significantly lowered, and the safety infrastructure requirements are reduced. This lean manufacturing approach allows for a more efficient allocation of resources, driving down the overall cost of goods sold (COGS) while maintaining high product quality standards required for regulatory approval.
- Enhanced Supply Chain Reliability: Relying on fermentable biocatalysts produced in E. coli ensures a stable and scalable supply of the key processing agent, independent of fluctuating markets for rare earth metals or specialized chemical reagents. The robustness of the enzyme under mild reaction conditions minimizes the risk of batch failures due to thermal runaway or exothermic events, ensuring consistent delivery schedules. This reliability is crucial for maintaining continuous production lines and meeting the just-in-time delivery expectations of downstream pharmaceutical clients who depend on uninterrupted access to critical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard fermentation and bioreaction equipment that can be easily expanded from pilot to commercial scales without significant process redesign. The green chemistry profile, characterized by high atom economy and aqueous processing, facilitates easier compliance with increasingly stringent environmental regulations regarding wastewater discharge and volatile organic compound (VOC) emissions. This future-proofs the manufacturing asset against regulatory changes and enhances the brand reputation of the manufacturer as a leader in sustainable pharmaceutical production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aldolase-based synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements.
Q: What are the stereoselectivity advantages of this aldolase method?
A: The patented aldolase catalyst demonstrates exceptional chiral control, achieving a diastereomeric excess (de) of greater than 99.5%, which eliminates the need for complex downstream chiral resolution steps required in traditional chemical synthesis.
Q: How does this biocatalytic route improve atom economy?
A: Unlike conventional methods that require protection and deprotection groups leading to significant waste, this one-step condensation theoretically achieves 100% atom utilization, drastically reducing chemical waste and environmental impact.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes robust E. coli expression systems and operates under mild conditions (30°C, pH 7), making it highly suitable for commercial scale-up from kilogram to multi-ton production without requiring extreme pressure or temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Droxidopa Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this biocatalytic technology in reshaping the landscape of amino acid derivative production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise measurement of diastereomeric excess and residual solvent levels. We are committed to delivering high-purity Droxidopa that meets the exacting standards of the global pharmaceutical industry, leveraging our expertise in enzyme engineering and process optimization to guarantee supply continuity.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and value in your drug development programs. Together, we can accelerate the availability of life-saving medications through superior manufacturing excellence.
