Advanced Manufacturing of Dasatinib Intermediates via Green Catalytic Cyclization
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of critical oncology therapeutics, particularly tyrosine kinase inhibitors like Dasatinib. Patent CN109503511B introduces a transformative preparation method for the key intermediate, 2-amino-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide, addressing long-standing inefficiencies in existing synthetic routes. This innovation leverages a concise two-step process that bypasses the formation of hazardous volatile intermediates, thereby enhancing operational safety and environmental compliance. By utilizing ethyl 3-oxopropionate as a stable starting material and employing polyethylene glycol (PEG 400) as a green reaction medium, the technology offers a compelling alternative for high-purity API intermediate manufacturing. The structural integrity and purity of the final compound are paramount for downstream drug efficacy, and this patented approach ensures consistent quality through simplified purification protocols. As a reliable dasatinib intermediate supplier, understanding these mechanistic advancements is crucial for securing a competitive edge in the global supply chain.
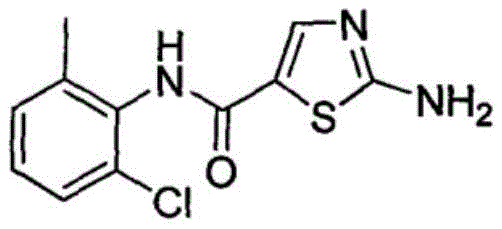
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazole-based kinase inhibitor intermediates has been plagued by complex multi-step sequences that demand rigorous control over reaction parameters. Traditional methodologies, such as those depicted in prior art involving oxalyl chloride and vinyl ethyl ether, necessitate the handling of highly volatile and unstable intermediates like 3-ethoxyacryloyl chloride. These legacy processes often require cryogenic conditions, anhydrous environments, and the use of expensive brominating agents like N-bromosuccinimide (NBS), which significantly inflate production costs and safety risks. Furthermore, the reliance on chlorinated solvents and the generation of substantial chemical waste pose severe challenges for environmental compliance and waste management in large-scale facilities. The inherent instability of key intermediates in these older routes frequently leads to polymerization side reactions, resulting in lower overall yields and difficult purification burdens that hinder commercial viability.
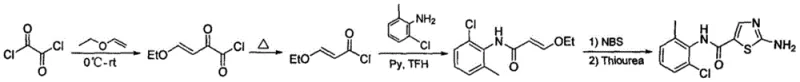
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel methodology disclosed in CN109503511B streamlines the synthesis into a highly efficient two-step sequence that prioritizes stability and safety. This innovative route initiates with the condensation of ethyl 3-oxopropionate and 2-chloro-6-methylaniline, followed by a controlled bromination using copper bromide to generate a stable alpha-bromo ketone intermediate. The subsequent cyclization with thiourea is conducted in PEG 400, a non-toxic and recyclable solvent that facilitates the reaction at ambient temperatures without the need for extreme thermal or pressure conditions. By eliminating the formation of volatile acyl chlorides and replacing hazardous NBS with cost-effective copper salts, this approach drastically simplifies the operational workflow and reduces the dependency on specialized equipment. The result is a robust process capable of delivering high-purity products with superior selectivity, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
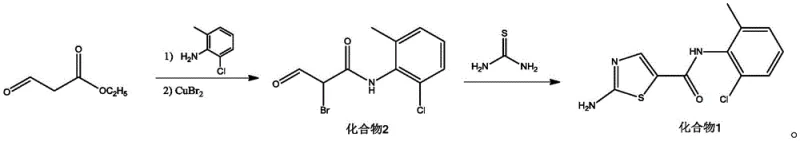
Mechanistic Insights into Copper-Mediated Bromination and Thiourea Cyclization
The core of this technological breakthrough lies in the strategic substitution of traditional brominating agents with copper bromide within an alkaline medium, which fundamentally alters the reaction kinetics and impurity profile. In the first step, the enolizable beta-keto ester undergoes a base-catalyzed condensation with the aniline derivative to form an enaminone species, which is then selectively brominated at the alpha-position by the copper salt. This copper-mediated process avoids the radical mechanisms often associated with NBS, thereby minimizing the formation of poly-brominated byproducts and ensuring a cleaner reaction mixture. The use of tetrahydrofuran as a co-solvent in this stage further enhances the solubility of the inorganic copper salts, promoting efficient mass transfer and consistent reaction rates across large batches. This precise control over the bromination step is critical for maintaining the structural fidelity of the intermediate prior to the final ring closure.
Following the isolation of the alpha-bromo ketone, the second step involves a nucleophilic substitution and cyclization cascade driven by thiourea in a PEG 400 matrix. The polyethylene glycol solvent acts not merely as a medium but as a phase-transfer catalyst that stabilizes the transition states involved in the thiazole ring formation. The sulfur atom of the thiourea attacks the electrophilic carbon of the bromo-ketone, followed by intramolecular cyclization and elimination of hydrogen bromide to yield the target thiazole carboxamide. This mechanism proceeds efficiently at room temperature, avoiding the thermal degradation issues common in high-temperature reflux conditions used in older methods. The ability of PEG 400 to dissolve both organic substrates and inorganic byproducts facilitates easy workup procedures, where the product can be precipitated and the solvent recovered, significantly enhancing the atom economy of the overall process.
How to Synthesize 2-amino-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide Efficiently
The practical implementation of this synthesis requires careful attention to stoichiometry and solvent management to maximize the benefits of the green chemistry principles embedded in the patent. The process begins with the preparation of the brominated intermediate under reflux, followed by a straightforward filtration and washing protocol to remove inorganic copper residues. The subsequent cyclization step is remarkably forgiving, allowing for a wide range of thiourea ratios while maintaining high conversion rates, which provides flexibility for process optimization during scale-up. Detailed standardized operating procedures regarding temperature ramping, addition rates, and crystallization parameters are essential to replicate the high purity levels reported in the patent examples. For a comprehensive guide on the specific molar ratios, reaction times, and purification techniques required to achieve optimal results, please refer to the technical instructions below.
- React ethyl 3-oxopropionate with 2-chloro-6-methylaniline under alkaline conditions, followed by bromination with copper bromide to obtain the alpha-bromo ketone intermediate.
- Cyclize the intermediate with thiourea in PEG 400 solvent at room temperature to form the thiazole ring.
- Purify the final product via pH adjustment, extraction, and crystallization to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. By shifting away from volatile and hazardous reagents, companies can significantly reduce the costs associated with specialized storage, transportation, and safety compliance measures. The elimination of expensive brominating agents like NBS in favor of readily available copper salts translates into substantial raw material cost savings, while the use of recyclable PEG 400 minimizes solvent procurement and waste disposal expenses. Furthermore, the stability of the intermediates generated in this process ensures a more reliable supply chain, as there is no risk of degradation during storage or transit that often plagues unstable acyl chloride derivatives. This enhanced stability allows for greater inventory flexibility and reduces the risk of production stoppages due to raw material spoilage.
- Cost Reduction in Manufacturing: The replacement of high-cost reagents such as N-bromosuccinimide with economical copper bromide and thiourea creates a direct pathway for lowering the cost of goods sold. Additionally, the simplified two-step nature of the synthesis reduces labor hours, energy consumption for heating and cooling, and the need for complex distillation equipment. The ability to recover and reuse the PEG 400 solvent further amplifies these savings by decreasing the volume of fresh solvent required per batch. Consequently, the overall manufacturing footprint becomes leaner and more cost-efficient, enabling competitive pricing strategies in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Sourcing stable starting materials like ethyl 3-oxopropionate and 2-chloro-6-methylaniline mitigates the risks associated with supply chain disruptions common for specialty reagents. Unlike the volatile intermediates required in conventional routes, the compounds generated in this process are solid and stable, allowing for safer warehousing and longer shelf lives. This stability ensures continuous production capabilities even during periods of logistical delay, providing a buffer against market volatility. Moreover, the reduced dependency on hazardous chemicals simplifies regulatory compliance and shipping logistics, facilitating smoother cross-border trade and faster delivery times to downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic chlorinated solvents make this process inherently scalable from pilot plant to commercial production volumes. The use of PEG 400 aligns with modern green chemistry initiatives, reducing the environmental burden of volatile organic compound (VOC) emissions and hazardous waste generation. This eco-friendly profile not only meets stringent international environmental regulations but also enhances the corporate sustainability image of the manufacturing entity. The simplified workup procedures, involving basic filtration and crystallization rather than complex chromatography or distillation, further support seamless scale-up without the need for prohibitive capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for dasatinib intermediates. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation to assist decision-makers in evaluating the technology. Understanding these nuances is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios. The answers highlight the specific advantages in terms of yield, purity, and operational safety that distinguish this method from prior art.
Q: What are the primary advantages of the PEG 400 solvent system in this synthesis?
A: The use of PEG 400 eliminates the need for volatile organic solvents and allows for solvent recycling, significantly reducing environmental impact and waste disposal costs compared to traditional acetonitrile or chloroform methods.
Q: How does this method improve upon the use of NBS in traditional routes?
A: By utilizing copper bromide instead of N-bromosuccinimide (NBS), the process avoids the high costs and harsh low-temperature conditions associated with NBS, while simultaneously improving reaction yield and selectivity.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method operates under mild conditions without requiring anhydrous or oxygen-free environments, and avoids unstable volatile intermediates, making it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-amino-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain leadership in the competitive pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN109503511B are fully realized in practical manufacturing settings. We are committed to delivering high-purity intermediates that meet stringent purity specifications through our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in green chemistry and process optimization, we can help you navigate the complexities of scaling this novel route while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to explore how this innovative synthesis can transform your supply chain dynamics and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible value of partnering with a leader in fine chemical innovation. Let us work together to build a more efficient, sustainable, and profitable future for your oncology drug portfolio.
