Optimizing Dasatinib Production: A Green Catalytic Route for Commercial Scale-Up
Optimizing Dasatinib Production: A Green Catalytic Route for Commercial Scale-Up
The pharmaceutical landscape for oncology treatments continues to evolve, with tyrosine kinase inhibitors like Dasatinib (marketed as Sprycel) remaining critical assets for treating chronic myelogenous leukemia. However, the manufacturing of such complex small molecules often faces significant hurdles regarding yield, environmental impact, and operational safety. Patent CN109369638B introduces a transformative preparation process that addresses these industrial pain points through a novel catalytic strategy. This technical insight report analyzes the patented methodology, which leverages heteropolyacid salts and copper-mediated transformations to streamline the synthesis of the key thiazole intermediate and the final API. By shifting away from cryogenic conditions and expensive palladium catalysts, this route offers a compelling value proposition for supply chain stability and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dasatinib and its precursors has relied on chemically aggressive and operationally complex pathways. As detailed in prior art literature such as J. Med. Chem. (2004, 2006), conventional routes frequently necessitate the use of n-butyllithium at cryogenic temperatures of -78°C to facilitate lithiation steps. These conditions impose severe constraints on industrial equipment, requiring specialized low-temperature reactors and significant energy expenditure for cooling. Furthermore, alternative pathways disclosed in patents like CN200580011916.6 often employ volatile and hazardous intermediates such as (E)-3-ethoxyacryloyl chloride, which poses storage and handling risks due to its high volatility and instability. The reliance on stoichiometric amounts of N-bromosuccinimide (NBS) for bromination in other routes further complicates the process, generating substantial organic waste and requiring rigorous purification to remove succinimide by-products.
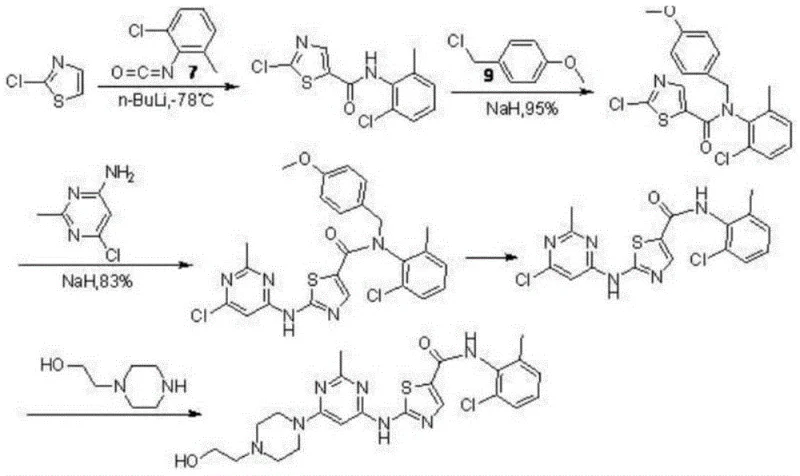
The Novel Approach
In stark contrast to these legacy methods, the process described in CN109369638B utilizes a convergent and温和 (mild) strategy centered on ethyl 3-oxopropionate as a stable starting material. The innovation lies in the direct construction of the thiazole ring via a copper bromide-mediated bromination followed by cyclization with thiourea, catalyzed by a reusable heteropolyacid salt. This eliminates the need for isolating unstable acyl chloride intermediates and avoids the use of cryogenic reagents entirely. The subsequent coupling with the pyrimidine moiety is achieved in a one-pot fashion using a copper iodide and N,N-dimethylglycine ligand system. This approach not only simplifies the operational workflow by reducing unit operations but also significantly enhances the overall atom economy. The transition from multi-step, hazardous sequences to a streamlined, catalytic process represents a paradigm shift in the manufacturing of this kinase inhibitor.
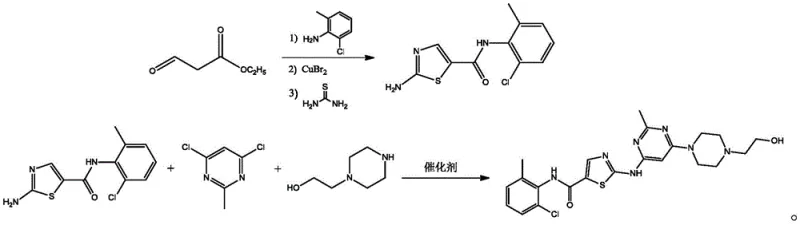
Mechanistic Insights into Copper-Mediated Thiazole Formation and Coupling
The core chemical innovation of this patent resides in the efficient construction of the 2-amino-N-(2-chloro-6-methylphenyl)thiazole-5-carboxamide scaffold. The mechanism initiates with the condensation of ethyl 3-oxopropionate and 2-chloro-6-methylaniline under alkaline conditions to form an enaminone intermediate. Crucially, the subsequent bromination step utilizes copper(II) bromide (CuBr2) rather than molecular bromine or NBS. In this context, CuBr2 acts as both an oxidant and a bromine source, facilitating the alpha-bromination of the carbonyl compound under reflux conditions. This is followed by the nucleophilic attack of thiourea on the brominated intermediate. The presence of the heteropolyacid salt catalyst, such as ammonium phosphomolybdate, accelerates the cyclization and dehydration steps required to aromatize the thiazole ring. The heteropolyacid functions as a solid superacid that can be easily removed by filtration, preventing acid corrosion issues common with liquid mineral acids and ensuring the catalyst can be recovered and reused, thereby aligning with green chemistry principles.
Following the formation of the thiazole intermediate, the final assembly of the Dasatinib molecule involves a nucleophilic aromatic substitution on the 4,6-dichloro-2-methylpyrimidine ring. The patent specifies the use of CuI coupled with N,N-dimethylglycine as a ligand in dimethylformamide (DMF) solvent. This catalytic system likely operates via a copper-ligand complex that activates the chloropyrimidine towards nucleophilic attack by the amino group of the thiazole intermediate and subsequently by the piperazine nitrogen. The choice of N,N-dimethylglycine as a bidentate ligand stabilizes the copper species in solution, preventing precipitation and maintaining catalytic activity at the elevated reaction temperature of 120°C. This specific ligand acceleration effect is critical for achieving high conversion rates without requiring excessive catalyst loading or prolonged reaction times, which directly translates to improved throughput in a commercial setting.
How to Synthesize Dasatinib Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-purity Dasatinib suitable for pharmaceutical applications. The process is divided into two primary stages: the formation of the thiazole carboxamide intermediate and the final coupling with the pyrimidine-piperazine fragment. The procedure emphasizes precise stoichiometric control, particularly regarding the ratio of copper bromide to the ketone precursor, and the careful selection of base (sodium methoxide or potassium phosphate) to drive the reactions to completion. Detailed standard operating procedures regarding temperature ramps, addition rates, and workup protocols are essential for replicating the reported yields of over 90% for the intermediate and 88% for the final API. For a comprehensive breakdown of the exact experimental parameters, please refer to the structured guide below.
- React ethyl 3-oxopropionate with 2-chloro-6-methylaniline under alkaline reflux, followed by copper bromide addition and thiourea cyclization using a heteropolyacid salt catalyst to form the thiazole intermediate.
- In a separate vessel, dissolve 4,6-dichloro-2-methylpyrimidine in DMF with N-hydroxyethyl piperazine and the thiazole intermediate.
- Add potassium phosphate, CuI catalyst, and N,N-dimethylglycine ligand, then heat to 120°C to effect the final coupling and obtain high-purity dasatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the process described in CN109369638B offers tangible strategic benefits beyond mere chemical elegance. The most immediate impact is observed in the reduction of raw material costs and the mitigation of supply risks associated with specialty reagents. By replacing expensive and hazardous reagents like n-butyllithium and palladium catalysts with commodity chemicals like copper bromide and thiourea, the bill of materials is significantly optimized. Furthermore, the elimination of cryogenic processing (-78°C) removes the need for specialized low-temperature infrastructure, allowing production to occur in standard glass-lined or stainless steel reactors. This flexibility enhances asset utilization and reduces the capital expenditure required for facility upgrades, making the technology accessible to a broader range of contract manufacturing organizations.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by the substitution of high-cost catalysts and reagents. Traditional methods often rely on palladium-based cross-coupling catalysts (e.g., Pd(OAc)2/BINAP) which are not only expensive but also require stringent removal steps to meet residual metal specifications in the final drug product. The switch to a copper-based catalytic system drastically lowers catalyst costs, as copper salts are orders of magnitude cheaper than palladium complexes. Additionally, the use of a filterable heteropolyacid catalyst simplifies the purification process, reducing the consumption of silica gel or activated carbon and minimizing product loss during filtration. These cumulative efficiencies lead to a lower cost of goods sold (COGS) without compromising the quality of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. The reliance on ethyl 3-oxopropionate and 2-chloro-6-methylaniline ensures a stable supply base, as these are bulk chemicals produced by multiple vendors globally. In contrast, routes dependent on custom-synthesized intermediates like (E)-3-ethoxyacryloyl chloride create single points of failure; if a supplier of this volatile intermediate faces production issues, the entire Dasatinib supply chain could be disrupted. By synthesizing the core scaffold in-house from stable precursors, manufacturers gain greater control over their production schedules and can better respond to fluctuations in market demand, ensuring continuous availability for downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner profile that facilitates regulatory approval and community acceptance. The avoidance of N-bromosuccinimide (NBS) eliminates the generation of succinimide waste, which can be difficult to treat in wastewater systems. Moreover, the reaction conditions are inherently safer; operating at reflux or moderate temperatures (120°C) rather than extreme cold or high pressure reduces the risk of thermal runaway incidents. The ability to recycle the heteropolyacid catalyst further minimizes the volume of hazardous waste generated per kilogram of product. These factors collectively simplify the permitting process for new manufacturing lines and reduce the long-term liability associated with hazardous waste disposal, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific improvements and data points highlighted in the patent documentation, focusing on yield optimization, impurity control, and operational safety. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for quality assurance teams establishing control strategies.
Q: How does this process improve upon traditional n-butyllithium routes?
A: Traditional routes often require cryogenic conditions (-78°C) and hazardous reagents like n-butyllithium. This patented process operates at mild temperatures (20-25°C for cyclization, reflux for initial steps) and utilizes stable copper salts, significantly reducing safety risks and energy consumption associated with deep freezing.
Q: What are the purity specifications achievable with this method?
A: The patent data indicates that the final dasatinib product can achieve purity levels exceeding 99.9% after simple recrystallization. The use of filterable heteropolyacid catalysts minimizes metal contamination, simplifying the purification workflow compared to homogeneous acid catalysts.
Q: Why is the elimination of N-bromosuccinimide (NBS) significant for manufacturing?
A: NBS is a solid brominating agent that can be difficult to handle on large scales due to potential exotherms and the generation of succinimide by-products which complicate waste streams. Replacing it with copper bromide in solution allows for better thermal control and easier downstream processing, enhancing overall process robustness.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dasatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN109369638B process are fully realized in a GMP environment. We are equipped with state-of-the-art reactors capable of handling the specific thermal profiles required for this copper-catalyzed synthesis, along with stringent purity specifications enforced by our rigorous QC labs. Our commitment to quality ensures that every batch of Dasatinib intermediate meets the exacting standards required by global regulatory bodies, providing our partners with peace of mind regarding product consistency and safety.
We invite pharmaceutical companies and generic manufacturers to explore the potential of this optimized synthesis route for their supply chains. By leveraging our technical expertise, you can achieve significant operational efficiencies and secure a reliable source of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the oncology therapeutic area.
