Advanced Roflumilast Synthesis: Eliminating Hazardous Reagents for Commercial Scale-Up
The global demand for Chronic Obstructive Pulmonary Disease (COPD) therapeutics continues to surge, placing immense pressure on the supply chains of key active pharmaceutical ingredients (APIs) like Roflumilast. As a selective phosphodiesterase-4 (PDE4) inhibitor, Roflumilast represents a critical treatment option, yet its manufacturing has historically been plagued by hazardous reagents and complex waste streams. Patent CN102850266A introduces a transformative approach to this synthesis, fundamentally altering the risk profile of the production line. By shifting from traditional chlorination-based activation to a carbonyldiimidazole (CDI) mediated coupling, this technology offers a pathway that aligns perfectly with modern green chemistry principles while maintaining high commercial viability. For procurement leaders and technical directors, understanding this shift is not merely about chemistry; it is about securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality without the volatility associated with dangerous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Roflumilast has relied heavily on aggressive chlorinating agents that pose significant operational hazards. As illustrated in the reaction scheme below, the conventional route typically involves the oxidation of the benzaldehyde precursor followed by conversion to an acid chloride using thionyl chloride (SOCl2). This step is notoriously problematic in large-scale manufacturing due to the evolution of toxic sulfur dioxide and hydrogen chloride gases, which require extensive scrubbing infrastructure. Furthermore, the subsequent amidation often necessitates the use of sodium hydride (NaH), a pyrophoric solid that demands rigorous inert atmosphere handling and specialized safety protocols. These factors collectively inflate the cost of goods sold (COGS) through increased safety overhead, expensive waste neutralization, and potential production downtime.
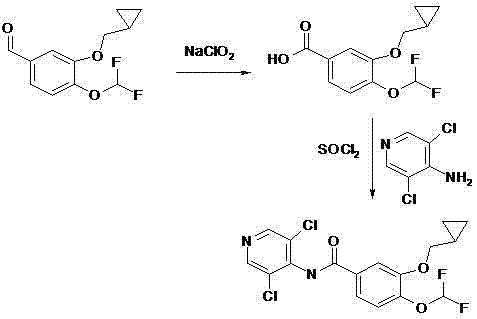
The Novel Approach
In stark contrast, the methodology disclosed in CN102850266A circumvents these bottlenecks by employing potassium permanganate (KMnO4) for the initial oxidation and CDI for the activation step. This strategic substitution eliminates the need for gaseous reagents and pyrophoric bases entirely. The oxidation proceeds smoothly in an aqueous alkaline environment at ambient temperatures, offering a much safer thermal profile than exothermic chlorinations. Subsequently, the activation of the carboxylic acid via CDI generates a reactive acyl imidazole intermediate in situ, which couples efficiently with the aminopyridine moiety. This approach not only streamlines the workflow by removing distinct chlorination and neutralization unit operations but also significantly enhances the overall atom economy, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into KMnO4 Oxidation and CDI-Mediated Coupling
The core of this technological advancement lies in the precise control of the oxidation state and the activation energy of the coupling reaction. In the first stage, potassium permanganate acts as a robust oxidant in a dilute sodium hydroxide solution (2-3% concentration). The alkaline medium facilitates the formation of the manganate ester intermediate, which rapidly decomposes to yield the corresponding benzoic acid salt. The patent specifies the use of phase transfer catalysts, such as tetrabutylammonium bromide, to enhance the interfacial contact between the organic aldehyde and the aqueous oxidant, ensuring complete conversion without over-oxidation or degradation of the sensitive difluoromethoxy group. This meticulous control is crucial for maintaining the integrity of the fluorine substituents, which are essential for the biological activity of the final drug.
Following the isolation of the high-purity acid intermediate, the process leverages the unique reactivity of N,N'-carbonyldiimidazole. Upon heating to 40-50°C in dimethylformamide (DMF), CDI reacts with the carboxylic acid to release carbon dioxide and imidazole, forming an electrophilic acyl imidazole species. This activated intermediate is sufficiently reactive to undergo nucleophilic attack by the weakly nucleophilic 3,5-dichloro-4-aminopyridine at elevated temperatures (60-75°C), yet stable enough to minimize side reactions such as racemization or hydrolysis. This mechanism effectively bypasses the harsh conditions required for acid chloride formation, resulting in a cleaner reaction profile with fewer by-products. For R&D teams, this implies a simplified purification strategy, often requiring only standard aqueous workups and solvent swaps to achieve the target specification of ≥99% purity.
How to Synthesize Roflumilast Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and temperature gradients outlined in the patent to maximize yield and safety. The process is divided into four distinct operational phases: oxidation, extraction/purification of the acid, activation, and final coupling. Each step is designed to be telescoped where possible, minimizing material handling and exposure. The following guide summarizes the critical operational parameters derived from the patent examples, serving as a foundational reference for process engineers looking to adapt this chemistry for pilot or commercial scale.
- Oxidize 3-(cyclopropylmethoxy)-4-(difluoromethoxy)benzaldehyde using potassium permanganate in alkaline solution at room temperature.
- Extract the resulting benzoic acid with ethyl acetate, wash, dry, and purify to obtain a crystalline solid.
- Activate the acid in DMF using N,N'-carbonyldiimidazole (CDI) at 40-50°C to form the reactive intermediate.
- Couple with 3,5-dichloro-4-aminopyridine at 60-75°C to finalize the Roflumilast structure.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain stakeholders, the transition to this novel synthetic route offers profound logistical and financial benefits beyond simple yield improvements. By removing the dependency on thionyl chloride and sodium hydride, manufacturers can drastically reduce their regulatory burden and insurance premiums associated with storing and handling hazardous materials. The elimination of corrosive gas emissions simplifies environmental compliance, allowing facilities to operate with greater continuity and less risk of regulatory shutdowns. Furthermore, the use of common, stable reagents like potassium permanganate and CDI ensures a more resilient supply chain, less susceptible to the market volatility often seen with specialty chlorinating agents.
- Cost Reduction in Manufacturing: The removal of thionyl chloride and sodium hydride eliminates the need for specialized corrosion-resistant reactors and complex gas scrubbing systems, leading to substantial capital expenditure savings. Additionally, the simplified workup procedure reduces solvent consumption and labor hours per batch, driving down the variable cost of production significantly without compromising quality.
- Enhanced Supply Chain Reliability: Utilizing stable, non-hazardous reagents mitigates the risk of transportation delays and storage restrictions. Potassium permanganate and CDI are widely available commodity chemicals, ensuring that production schedules are not disrupted by the supply constraints often associated with controlled or dangerous precursors, thereby guaranteeing consistent delivery timelines for downstream partners.
- Scalability and Environmental Compliance: The aqueous nature of the oxidation step and the absence of toxic gas evolution make this process inherently scalable from kilogram to multi-ton quantities. The reduction in hazardous waste generation aligns with increasingly stringent global environmental standards, facilitating easier permitting for capacity expansion and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Roflumilast synthesis method. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, providing clarity on performance metrics and operational safety.
Q: Why is the KMnO4 oxidation method preferred over sodium chlorite for Roflumilast production?
A: The KMnO4 method operates under milder alkaline conditions and avoids the generation of chlorine dioxide gas, significantly reducing safety risks and equipment corrosion compared to acidic sodium chlorite oxidation.
Q: How does replacing thionyl chloride with CDI impact industrial waste management?
A: Replacing thionyl chloride eliminates the release of corrosive SO2 and HCl gases, drastically simplifying scrubber requirements and reducing the volume of hazardous acidic wastewater generated during the acylation step.
Q: What purity levels can be achieved with this novel synthetic route?
A: According to patent data, this route consistently achieves HPLC purity levels of ≥98.5% for the acid intermediate and ≥99% for the final product, minimizing the need for complex recrystallization cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN102850266A requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the absence of critical impurities, guaranteeing that every batch of Roflumilast intermediate meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to leverage these technological advantages for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency and reduce total landed costs.
