Advanced Synthetic Routes for Roflumilast Intermediates: Scaling High-Purity PDE4 Inhibitors
Advanced Synthetic Routes for Roflumilast Intermediates: Scaling High-Purity PDE4 Inhibitors
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatment has been significantly advanced by the development of selective phosphodiesterase 4 (PDE4) inhibitors, with Roflumilast standing out as a premier oral therapeutic agent. The efficient manufacturing of this critical active pharmaceutical ingredient (API) relies heavily on the robustness of its synthetic pathway. Patent CN102838533A discloses a highly optimized preparation method for Roflumilast and its key intermediates, addressing longstanding challenges in yield and purity that have plagued earlier production methodologies. This technical insight report analyzes the novel two-step synthesis strategy detailed in the patent, which involves the initial formation of a crucial amide intermediate followed by a selective etherification. By leveraging versatile starting materials and mild reaction conditions, this process offers a compelling value proposition for global supply chains seeking reliable pharmaceutical intermediates with consistent quality profiles.
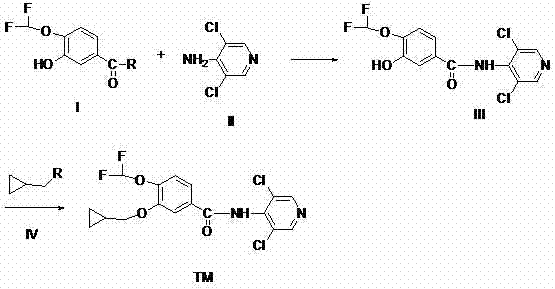
The core innovation lies in the strategic selection of precursors for the initial amidation step. Unlike rigid synthetic routes that depend on a single specific starting material, this methodology accommodates a diverse range of Compound I derivatives, including 3-hydroxy-4-(difluoromethoxy)benzoic acid, various esters such as t-butyl perbenzoate, and even the reactive benzoyl chloride. This flexibility allows manufacturers to adapt their procurement strategies based on market availability and cost fluctuations without compromising the integrity of the final product. The subsequent conversion of Intermediate III into the final Roflumilast structure (TM) via alkylation with cyclopropylmethyl halides demonstrates exceptional efficiency, achieving yields that significantly exceed industry averages for similar heterocyclic substitutions. For R&D Directors focused on impurity control, the mild thermal profile of this reaction minimizes the formation of thermal degradation byproducts, ensuring a cleaner crude profile that simplifies downstream purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex benzamide derivatives like Roflumilast has been fraught with inefficiencies that drive up costs and extend lead times. Traditional pathways often necessitate the use of expensive coupling reagents or harsh acidic conditions that can compromise the stability of the difluoromethoxy group, a critical pharmacophore for PDE4 inhibition. Furthermore, older methods frequently suffer from poor regioselectivity during the introduction of the cyclopropylmethoxy moiety, leading to difficult-to-remove isomeric impurities that require extensive chromatographic purification. These bottlenecks not only inflate the cost of goods sold (COGS) but also introduce significant variability in batch-to-batch consistency, posing a risk to supply chain reliability. Additionally, the reliance on specialized catalysts or multi-protection group strategies in conventional routes adds unnecessary operational complexity, increasing the environmental footprint through higher solvent consumption and waste generation.
The Novel Approach
In stark contrast, the methodology outlined in CN102838533A streamlines the synthesis into a direct and atom-economical sequence. By utilizing a direct amidation between the hydroxy-substituted benzoic acid derivatives and 3,5-dichloro-4-aminopyridine, the process eliminates the need for extraneous activation steps when using acid chloride or activated ester precursors. The reaction conditions are remarkably forgiving, operating effectively within a temperature range of 50 to 80 degrees Celsius, which is easily maintainable in standard glass-lined steel reactors. This approach drastically reduces the energy input required compared to high-temperature reflux methods. Moreover, the subsequent alkylation step utilizes common bases like triethylamine or sodium carbonate in polar aprotic solvents such as DMF or acetonitrile, facilitating a clean SN2 substitution. This simplicity translates directly into cost reduction in pharmaceutical intermediates manufacturing, as it lowers both the capital expenditure on specialized equipment and the operational expenditure on utilities and waste disposal.
Mechanistic Insights into Amidation and Etherification
The success of this synthetic route hinges on the precise control of nucleophilic attacks in both the amidation and alkylation stages. In the first step, the amino group of 3,5-dichloro-4-aminopyridine acts as a potent nucleophile, attacking the electrophilic carbonyl carbon of Compound I. When Compound I is an acid chloride, the reaction proceeds rapidly via an addition-elimination mechanism, releasing HCl which is scavenged by the added base. When esters are used, the mechanism involves a tetrahedral intermediate that collapses to expel the alkoxide leaving group. The presence of the electron-withdrawing difluoromethoxy group at the para-position enhances the electrophilicity of the carbonyl carbon, thereby accelerating the reaction rate and allowing for milder conditions. This electronic effect is crucial for maintaining the integrity of the sensitive pyridine ring, preventing unwanted side reactions such as nucleophilic aromatic substitution on the chlorine atoms.
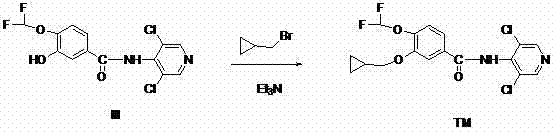
Following the formation of Intermediate III, the second stage involves the O-alkylation of the phenolic hydroxyl group. The phenolic oxygen, once deprotonated by the base to form a phenoxide anion, becomes a strong nucleophile capable of displacing the halide in cyclopropylmethyl bromide or iodide. The choice of solvent plays a pivotal role here; polar aprotic solvents like DMF stabilize the transition state without solvating the nucleophile too strongly, thus enhancing its reactivity. The steric bulk of the cyclopropyl group is managed effectively by the linear geometry of the methylene linker, ensuring high conversion rates. This mechanistic understanding allows for fine-tuning of reaction parameters to suppress potential N-alkylation side products, ensuring that the final high-purity Roflumilast meets stringent regulatory specifications for API intermediates.
How to Synthesize Roflumilast Efficiently
The practical execution of this synthesis requires careful attention to stoichiometry and solvent selection to maximize throughput. The patent details specific embodiments where the molar ratios of reactants are optimized to drive the equilibrium towards completion, particularly in the reversible ester aminolysis reactions. For instance, using a slight excess of the amine or the alkylating agent ensures that the limiting reagent is fully consumed, minimizing the burden on purification units. The workup procedures described, involving simple aqueous washes and filtration, highlight the robustness of the solid-state properties of the intermediates, which precipitate cleanly from the reaction mixture. Detailed standardized synthesis steps for implementing this route in a GMP environment are provided below.
- React Compound I (various benzoic acid derivatives) with Compound II (3,5-dichloro-4-aminopyridine) in an organic solvent at 50-80°C to form Intermediate III.
- Purify Intermediate III via filtration or crystallization to ensure high purity before the next step.
- Alkylate Intermediate III with Compound IV (cyclopropylmethyl halide) in the presence of a base like triethylamine or sodium carbonate to yield Roflumilast (TM).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the diversification of the raw material base; since Compound I can be sourced as an acid, an ester, or an acid chloride, buyers are not locked into a single supplier or a volatile commodity market. This flexibility acts as a hedge against supply disruptions, ensuring continuous production schedules. Furthermore, the elimination of exotic catalysts and the use of commodity solvents like methanol, ethanol, and toluene significantly lower the barrier to entry for contract manufacturing organizations (CMOs), fostering a more competitive bidding environment for production services.
- Cost Reduction in Manufacturing: The streamlined two-step process inherently reduces the number of unit operations, which directly correlates to lower labor costs and reduced facility occupancy time. By avoiding the use of precious metal catalysts often found in cross-coupling alternatives, the process eliminates the costly and time-consuming step of heavy metal scavenging and residual analysis. The high yields reported in the patent embodiments, often exceeding 80-90%, mean that less raw material is wasted per kilogram of final product, driving down the effective cost per unit. Additionally, the ability to recycle solvents like THF and acetonitrile further contributes to substantial cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 3,5-dichloro-4-aminopyridine and cyclopropylmethyl bromide ensures that the supply chain is resilient to geopolitical or logistical shocks. These chemicals are produced at scale by multiple global vendors, reducing the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility quality, such as steam pressure fluctuations, making it easier to transfer technology between different manufacturing sites without loss of efficiency. This stability is critical for maintaining long-term supply agreements with major pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns well with green chemistry principles by maximizing atom economy and minimizing waste generation. The absence of toxic heavy metals simplifies wastewater treatment protocols, reducing the environmental compliance burden on manufacturing facilities. The process is inherently scalable, as demonstrated by the successful transition from gram-scale laboratory experiments to multi-kilogram pilot batches without significant re-optimization. This scalability ensures that the commercial scale-up of complex PDE4 inhibitors can be achieved rapidly to meet surging market demand, providing a distinct competitive advantage in the fast-paced respiratory therapy sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this route for their own production pipelines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance.
Q: What are the preferred starting materials for Compound I in this synthesis?
A: The patent specifies several viable precursors including 3-hydroxy-4-(difluoromethoxy)benzoic acid, its esters (t-butyl, ethyl, isopropyl), and the acid chloride, offering flexibility based on cost and availability.
Q: How does this method improve upon traditional Roflumilast synthesis?
A: This method simplifies the process into two high-yielding steps with mild reaction conditions (50-80°C), avoiding harsh reagents and significantly improving overall product quality and yield compared to older multi-step routes.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common industrial solvents like THF, DMF, and acetonitrile, along with readily available alkylating agents, makes this route highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the delivery of life-saving medications. Our technical team has extensively analyzed the methodology presented in CN102838533A and possesses the expertise to optimize these reactions for industrial-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of high-purity Roflumilast intermediate we produce meets the stringent purity specifications required by global regulatory bodies.
We invite pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments that demonstrate how our optimized processes can enhance your supply chain resilience and profitability.
