Optimizing Capecitabine Production: A Scalable Route from Furtulon for Global Pharma Supply Chains
The global demand for high-quality oncology therapeutics continues to drive innovation in the synthesis of critical active pharmaceutical ingredients (APIs). Among these, Capecitabine stands out as a pivotal oral fluoropyrimidine carbamate, widely utilized for the treatment of breast and colorectal cancers. However, traditional manufacturing routes have often been plagued by harsh reaction conditions and complex purification requirements. A breakthrough disclosed in patent CN102260310A introduces a transformative approach to synthesizing Capecitabine starting from Furtulon (5'-deoxy-5-fluoro-uridine). This novel methodology replaces aggressive chemical treatments with a sequence of mild, highly controllable reactions, specifically leveraging orthoester protection strategies. For pharmaceutical manufacturers and procurement leaders, this represents a significant opportunity to enhance supply chain stability while reducing the environmental footprint associated with API production. The process is characterized by its rational sequence, stable raw material availability, and exceptional yield profiles, making it an ideal candidate for modern, green chemistry-compliant facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Capecitabine and related nucleoside analogs has relied heavily on protecting group strategies that involve acetyl, isopropylidene, or benzoyl groups at the 2' and 3' hydroxyl positions. While chemically valid, the removal of these protecting groups typically necessitates the use of strong acids or highly basic reflux conditions. These harsh environments frequently induce unwanted side reactions, such as glycosidic bond cleavage or base degradation, which severely compromise the integrity of the nucleoside scaffold. Consequently, the resulting crude products often exhibit low purity and complex impurity profiles, necessitating laborious and costly purification processes like repeated recrystallization or column chromatography. Furthermore, the poor controllability of these exothermic and aggressive reactions poses safety risks and limits the feasibility of scaling up to multi-ton production levels, creating bottlenecks for reliable API intermediate suppliers.
The Novel Approach
In stark contrast, the process outlined in CN102260310A utilizes a sophisticated orthoester protection mechanism that fundamentally alters the reaction landscape. By employing trimethyl orthoformate or orthoacetate under catalytic acidic conditions, the 2' and 3' hydroxyl groups are protected efficiently at ambient temperatures. This mild protection strategy avoids the thermal stress associated with traditional esterification. More critically, the subsequent deprotection step utilizes dilute acid solutions at low temperatures rather than harsh reflux, preserving the delicate fluorinated pyrimidine ring. This approach not only streamlines the workflow by allowing intermediates to be used directly in the next step without isolation but also ensures that the final Capecitabine product achieves high purity specifications with minimal downstream processing. The result is a robust, linear synthesis pathway that maximizes atom economy and operational safety.
Mechanistic Insights into Orthoester-Mediated Nucleoside Modification
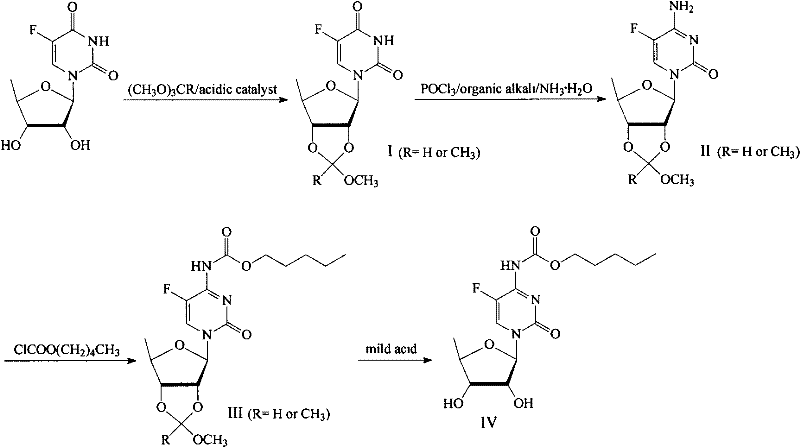
The core of this synthetic innovation lies in the precise manipulation of the ribose moiety through orthoester chemistry. In the initial step, 5'-deoxy-5-fluoro-uridine reacts with a trimethyl orthoester in the presence of a catalyst like p-toluenesulfonic acid. This reaction forms a cyclic methoxymethylene bridge across the 2' and 3' oxygens, effectively locking the ribose conformation and preventing unwanted nucleophilic attacks at these positions during subsequent transformations. This regioselective protection is crucial because it directs the reactivity exclusively to the N3 position of the uracil ring during the amination phase. Following protection, the conversion to the cytidine analog is achieved via activation with phosphorus oxychloride followed by ammonolysis. The mild conditions ensure that the orthoester bridge remains intact, providing a stable scaffold for the introduction of the amino group without risking hydrolysis of the sugar moiety.
Following the formation of the cytidine core, the N4-position is acylated using n-pentyl chloroformate to install the essential carbamate side chain required for Capecitabine's prodrug activity. The final step involves the strategic removal of the orthoester protecting group. Unlike benzoyl or acetyl groups that require vigorous hydrolysis, the methoxymethylene bridge is labile to mild acidic conditions. By carefully controlling the pH and temperature during this deprotection, the process releases the free 2' and 3' hydroxyl groups while leaving the N4-carbamate and the glycosidic bond untouched. This selectivity is the key to the high yields reported in the patent examples, as it virtually eliminates the formation of degradation byproducts that typically plague nucleoside synthesis.
How to Synthesize Capecitabine Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and temperature control, particularly during the phosphorylation and deprotection stages. The patent details a streamlined four-step sequence that begins with the protection of Furtulon, proceeds through the formation of the cytidine intermediate, followed by carbamoylation, and concludes with deprotection. Each step is designed to be telescoped where possible, minimizing solvent exchanges and handling time. For process chemists looking to adopt this methodology, the key lies in maintaining the integrity of the orthoester intermediate and ensuring complete conversion during the amination step to prevent carryover of uridine impurities. Detailed standardized operating procedures for each reaction stage, including specific solvent choices and workup protocols, are essential for replicating the high purity outcomes described in the intellectual property.
- Protect the 2',3'-hydroxyl groups of 5'-deoxy-5-fluoro-uridine using trimethyl orthoformate and an acid catalyst to form the methoxymethylene intermediate.
- Convert the uridine derivative to the cytidine analog using phosphorus oxychloride and ammonia water under controlled低温 conditions.
- Perform N4-acylation using n-pentyl chloroformate and an organic base to introduce the carbamate side chain.
- Remove the protecting groups using mild acid hydrolysis to yield high-purity Capecitabine without harsh reflux conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the drastic simplification of the manufacturing workflow. By eliminating the need for harsh reflux conditions and complex purification steps between intermediates, the process significantly reduces the consumption of solvents, energy, and labor hours. This efficiency translates directly into a more competitive cost structure for the final API intermediate. Furthermore, the use of stable, commercially available starting materials like Furtulon ensures a reliable supply base, mitigating the risks associated with sourcing exotic or unstable reagents. The robustness of the reaction conditions also implies a lower rate of batch failures, enhancing overall supply continuity for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The elimination of cumbersome purification treatments between steps is a major driver for cost savings. Traditional routes often require isolation and extensive washing of intermediates to remove side products generated by harsh conditions. In this new process, the high selectivity and mild conditions allow intermediates to be carried forward directly, or with minimal workup. This reduction in unit operations lowers the demand for equipment time, filtration media, and solvent recovery capacity. Additionally, the avoidance of expensive catalysts or specialized high-pressure equipment further contributes to a leaner manufacturing cost profile, allowing for substantial margin improvements in high-volume production scenarios.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the predictability of the manufacturing process. The described method operates under mild conditions (often at room temperature or 0°C), which reduces the thermal load on reactors and minimizes the risk of runaway reactions or equipment corrosion associated with strong acids and bases. This operational safety allows for longer campaign runs and reduced maintenance downtime. Moreover, the raw materials specified, such as trimethyl orthoformate and n-pentyl chloroformate, are commodity chemicals with stable global supply chains. This ensures that production schedules are not disrupted by the scarcity of niche reagents, providing a consistent flow of high-purity Capecitabine to meet market demand.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding pharmaceutical waste and solvent emissions, this process offers a greener alternative. The high yields achieved at each step mean less raw material is wasted as byproduct. The ability to perform reactions in common solvents like acetonitrile or dichloromethane, followed by simple aqueous workups, facilitates easier solvent recycling and waste treatment. The absence of heavy metal catalysts or toxic reagents simplifies the environmental compliance burden. From a scalability perspective, the linear nature of the synthesis and the robustness of the intermediates make it highly amenable to scale-up from pilot plant quantities to multi-ton commercial production without the need for significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Capecitabine synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing route.
Q: What are the key advantages of this Capecitabine synthesis route over traditional methods?
A: Unlike conventional methods that require harsh strong acid or base reflux for deprotection—often leading to side reactions and low yields—this novel process utilizes mild orthoester protection and gentle acidic deprotection. This results in significantly higher crude product purity (up to 99.5%) and eliminates the need for cumbersome purification steps between intermediates.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights the process's suitability for industrial production. The reaction conditions are mild (often at room temperature or 0°C), the raw materials like Furtulon are stable and easily obtainable, and the high yields across all four steps ensure economic viability for commercial scale-up.
Q: How does this method impact the impurity profile of the final API?
A: By avoiding extreme pH conditions and high-temperature reflux during the deprotection phase, the formation of degradation byproducts is minimized. The method allows for direct use of intermediates in subsequent steps without isolation, which reduces exposure to environmental contaminants and simplifies the overall impurity control strategy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN102260310A can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to monitor every critical parameter of the synthesis, from the initial orthoester protection to the final crystallization of Capecitabine. Our commitment to quality assurance ensures that every batch meets the exacting standards required for oncology API manufacturing.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact us today to request specific COA data for our Capecitabine intermediates and to discuss route feasibility assessments for your upcoming projects. Let us help you optimize your supply chain with a process that delivers both economic and technical superiority.
