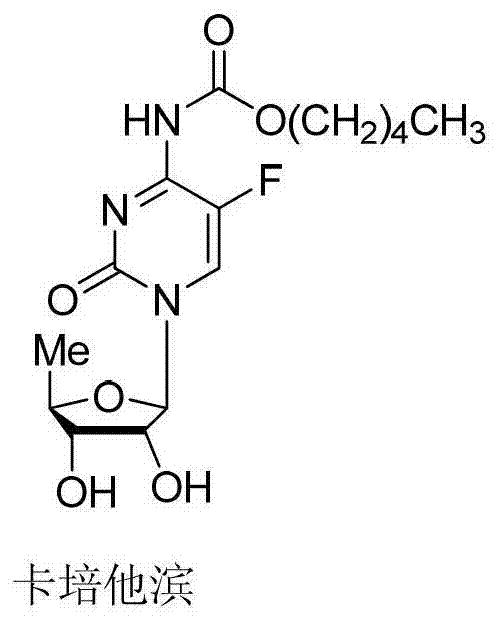
Optimizing Capecitabine Production: A Technical Analysis of High-Yield Glycosylation Strategies
Optimizing Capecitabine Production: A Technical Analysis of High-Yield Glycosylation Strategies
The global demand for high-purity oncology therapeutics continues to drive innovation in the synthesis of key pharmaceutical intermediates. Patent CN103113441A presents a significant advancement in the preparation of Capecitabine, an oral cytotoxic prodrug widely used in the treatment of breast and colorectal cancers. This proprietary methodology addresses critical bottlenecks in traditional manufacturing by introducing a streamlined four-step sequence: silicon-based protection, Lewis acid-catalyzed glycosylation, condensation, and hydrolysis. Unlike conventional routes that suffer from low yields or reliance on prohibitively expensive chiral starting materials, this novel approach leverages a robust silylation-glycosylation strategy to achieve a total yield ranging from 54% to 67%. For R&D directors and process chemists, the introduction of ammonium sulfate as a silylation catalyst and the optimization of recrystallization solvents represent tangible improvements in impurity profiling and process robustness.
From a supply chain perspective, the ability to produce pharmacopeia-grade Capecitabine with such efficiency translates directly into enhanced reliability for downstream API manufacturers. The patent details specific conditions, such as the use of tin tetrachloride (SnCl4) at controlled low temperatures (0 to 10°C), which ensures high stereoselectivity during the critical glycosidic bond formation. Furthermore, the strategic selection of solvents like toluene for silylation and methylene dichloride (DCM) for glycosylation aligns with standard industrial solvent recovery systems, facilitating easier scale-up. This technical disclosure provides a blueprint for reducing lead time for high-purity pharmaceutical intermediates by minimizing the number of purification steps required between the initial coupling and the final deprotection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Capecitabine has been plagued by economic and technical inefficiencies that hinder large-scale adoption. Early methodologies, such as those utilizing 5'-deoxy-5-fluorocytidine as a starting material, necessitated multiple acylation and hydrolysis steps that not only increased operational costs but also introduced significant opportunities for yield loss during isolation. Other reported routes relied on the use of pentyloxy formyl chloride for simultaneous acylation of hydroxyl and amino groups, followed by selective hydrolysis; however, achieving the necessary selectivity without compromising the integrity of the fluoropyrimidine ring often required harsh conditions or expensive reagents. Perhaps most critically, methods employing racemic triacetoxy ribofuranose suffered from inherent stereochemical inefficiencies, leading to difficult separation of diastereomers and ultimately limiting the overall yield of the desired beta-anomer. These legacy processes often resulted in complex impurity profiles that were challenging to purge to meet stringent regulatory standards.
The Novel Approach
The methodology outlined in CN103113441A fundamentally restructures the synthetic logic to overcome these historical barriers. By initiating the sequence with an in-situ silylation of 5-fluorocytosine using hexamethyldisilazane (HMDS), the process activates the nucleobase for subsequent coupling without the need for isolating sensitive intermediates. The core innovation lies in the glycosylation step, where the silylated base reacts with 1,2,3-triacetoxy-5-deoxy-D-ribose under the influence of a Lewis acid catalyst. As illustrated in the reaction scheme below, this direct coupling bypasses the need for pre-activated nucleoside precursors, significantly shortening the synthetic timeline. The subsequent condensation with n-amyl chloroformate and final hydrolysis are optimized to maximize recovery, with the patent reporting isolated yields of up to 88.7% for the glycosylation intermediate and 77% for the final hydrolysis step. This streamlined pathway offers a clear route for cost reduction in pharmaceutical intermediates manufacturing by consolidating steps and improving material throughput.
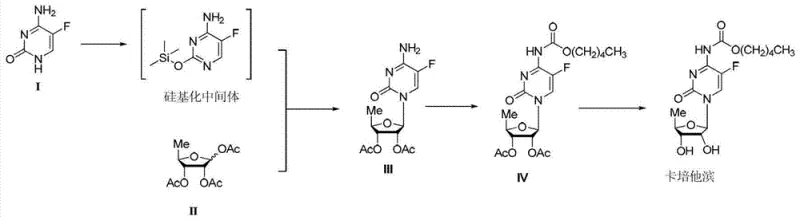
Mechanistic Insights into Lewis Acid-Catalyzed Glycosylation
The success of this synthetic route hinges on the precise control of the glycosylation mechanism, which is facilitated by the use of tin tetrachloride (SnCl4) as a potent Lewis acid. In this transformation, the silylated 5-fluorocytosine acts as a nucleophile, attacking the anomeric center of the ribose derivative. The presence of the Lewis acid coordinates with the acetate leaving group at the C1' position of the sugar, generating a highly reactive oxocarbenium ion intermediate. This activation lowers the energy barrier for the nucleophilic attack, ensuring that the reaction proceeds efficiently even at low temperatures (0 to 30°C). The stereochemical outcome is heavily influenced by the neighboring group participation of the C2' acetate, which directs the formation of the desired beta-glycosidic linkage through a transient cyclic acyloxonium ion. This mechanistic pathway is crucial for R&D teams focused on impurity control, as it minimizes the formation of the alpha-anomer and other regioisomers that typically complicate downstream purification.
Furthermore, the patent highlights a critical safety and quality control mechanism during the workup of the glycosylation reaction. Traditional Lewis acid quenches can be exothermic and generate hazardous gases; however, this protocol specifies a sequential quenching procedure where water is added first to decompose the Lewis acid complex, followed by the addition of a mild base like sodium bicarbonate. This two-stage neutralization prevents the violent evolution of gas and ensures that the reaction mixture remains stable during processing. Additionally, the use of mixed solvent systems for recrystallization, specifically isopropanol and methyl tert-butyl ether (MTBE), allows for the selective precipitation of the desired intermediate while keeping soluble impurities in the mother liquor. This level of control over the solid-state properties of the intermediate is essential for maintaining a consistent impurity profile throughout the campaign, ensuring that the final API meets the rigorous specifications required for oncology drugs.
How to Synthesize Capecitabine Efficiently
The synthesis of Capecitabine via this patented route requires strict adherence to temperature controls and stoichiometric ratios to replicate the high yields reported in the literature. The process begins with the silylation of 5-fluorocytosine, followed by the critical glycosylation step which dictates the overall stereochemical purity. Detailed operational parameters, including the specific molar equivalents of tin tetrachloride and the precise timing of the quenching agents, are essential for successful execution. For process engineers looking to implement this technology, the following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility and safety at scale.
- Perform silylation of 5-fluorocytosine using HMDS and ammonium sulfate catalyst in toluene at reflux.
- Conduct glycosylation with 1,2,3-triacetoxy-5-deoxy-D-ribose using SnCl4 in DCM, followed by careful quenching and recrystallization.
- Execute condensation with n-amyl chlorocarbonate in the presence of pyridine, followed by acid wash and hydrolysis with NaOH.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for expensive, pre-functionalized nucleoside starting materials found in older methods, manufacturers can source cheaper, commodity-grade chemicals like 5-fluorocytosine and triacetoxy ribose. This shift significantly reduces the cost of goods sold (COGS) and mitigates the risk of supply disruptions associated with niche specialty reagents. Moreover, the high overall yield of 54-67% implies a much lower material intensity per kilogram of finished product, which directly correlates to reduced waste disposal costs and a smaller environmental footprint. These factors combine to create a more resilient supply chain capable of withstanding market volatility.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in favor of inexpensive Lewis acids like tin tetrachloride, combined with the use of common industrial solvents such as toluene and DCM, drastically lowers the operational expenditure. The process avoids the need for costly chromatographic purifications by relying on efficient crystallization techniques, which are inherently cheaper and more scalable. This optimization ensures that the production of high-purity pharmaceutical intermediates remains economically competitive even in a price-sensitive generic market.
- Enhanced Supply Chain Reliability: By utilizing robust chemistry that tolerates minor variations in reaction conditions, the manufacturing process becomes less prone to batch failures. The specific workup procedures described, such as the controlled neutralization of the glycosylation mixture, reduce the likelihood of safety incidents that could halt production lines. This reliability allows supply chain planners to forecast delivery timelines with greater confidence, ensuring continuous availability of this critical oncology intermediate for downstream API synthesis.
- Scalability and Environmental Compliance: The synthetic route is designed with industrial scalability in mind, avoiding exotic reagents or extreme conditions that are difficult to manage in large reactors. The ability to recycle solvents like toluene and DCM further enhances the sustainability profile of the process. Additionally, the high purity of the intermediates reduces the burden on wastewater treatment facilities, as fewer organic impurities need to be degraded. This alignment with green chemistry principles facilitates smoother regulatory approvals and long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Capecitabine synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for potential licensing or contract manufacturing opportunities.
Q: What are the key advantages of the silylation-glycosylation route for Capecitabine?
A: This route avoids expensive starting materials used in prior art and utilizes a robust Lewis acid catalysis system (SnCl4) that achieves high yields (up to 88.7% in glycosylation step) and superior purity through optimized recrystallization protocols.
Q: How does this method address safety concerns during glycosylation?
A: The process incorporates a specific quenching sequence where water is added first to terminate the reaction, followed by alkali neutralization. This controlled approach prevents the rapid evolution of large amounts of gas, mitigating safety risks associated with wayward reactions in industrial settings.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the method is explicitly designed for industrial suitability. It utilizes common solvents like toluene and DCM, avoids complex purification of intermediates where possible, and achieves a total yield of 54-67%, making it economically viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in CN103113441A can be faithfully reproduced on an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of Capecitabine intermediate meets the stringent purity specifications required by global regulatory bodies.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market for this vital anticancer agent.
