Revolutionizing Amine Production: Safe, Scalable Rearrangement of Hydroxamic Acids for Commercial Manufacturing
Revolutionizing Amine Production: Safe, Scalable Rearrangement of Hydroxamic Acids for Commercial Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical manufacturing, particularly regarding safety, selectivity, and environmental impact. Patent CN101208292A introduces a transformative methodology for the preparation of unprotected or carbamate-protected amines from hydroxamic acids, utilizing alkylphosphonic anhydrides as the key activating agents. This technology addresses critical limitations found in classical rearrangement reactions by enabling transformations under exceptionally mild temperature ranges, typically between -100°C and +120°C, with a preferred operational window of -30°C to +30°C. For R&D directors and process chemists, this represents a paradigm shift away from hazardous reagents toward a more controlled, high-fidelity synthetic route that preserves stereochemical integrity while delivering near-quantitative yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of carboxylic acid derivatives into primary amines has relied on established but problematic protocols such as the Hofmann, Curtius, Schmidt, and Lossen decompositions. These traditional pathways are fraught with significant safety hazards and operational complexities that pose severe risks in a commercial setting. For instance, the Hofmann degradation necessitates the use of aggressive and toxic halogens like bromine or chlorine alongside strong alkali metals, which can degrade sensitive functional groups present in complex multifunctional molecules. Similarly, the Curtius and Schmidt rearrangements involve the handling of hydrazine or azido acid, substances known for their explosive potential and high toxicity. Furthermore, the drastic conditions often required for these classical methods frequently lead to unwanted epimerization of stereocenters, compromising the optical purity essential for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique reactivity of cyclic alkylphosphonic anhydrides, such as 2,4,6-substituted 1,3,5,2,4,6-trioxatriphosphines, to activate hydroxamic acids gently and efficiently. This method eliminates the need for explosive azides or corrosive halogens, thereby drastically improving the safety profile of the manufacturing process. The reaction proceeds with remarkable chemoselectivity, allowing for the conversion of specific carboxylic groups even in the presence of other sensitive functionalities without affecting them. By operating at lower temperatures, the process effectively suppresses side reactions and prevents the epimerization of chiral centers, ensuring that the final amine product retains the desired stereochemistry. This level of control is paramount for the synthesis of high-value pharmaceutical intermediates where structural fidelity is non-negotiable.
Mechanistic Insights into Phosphonic Anhydride-Mediated Rearrangement
The core of this technological advancement lies in the activation mechanism facilitated by the phosphonic anhydride. When the hydroxamic acid reacts with the cyclic anhydride, it forms a highly reactive acyloxyphosphonium intermediate. This activation step is crucial as it lowers the energy barrier for the subsequent rearrangement, allowing the migration of the R1 group from the carbonyl carbon to the nitrogen atom to occur under mild thermal conditions. Unlike traditional activation methods that might generate unstable isocyanates prone to polymerization or side reactions, this pathway ensures a smooth transition to the amine or carbamate species. The presence of an alcohol (R2OH) in the reaction mixture allows for the immediate trapping of the intermediate, directly yielding the carbamate-protected amine (R1-NHCO2R2) in a single pot, which simplifies the overall synthetic sequence.
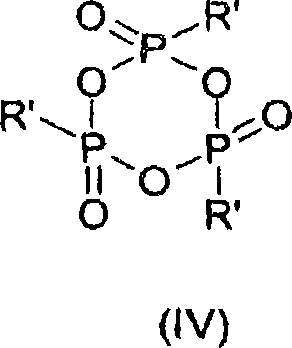
Furthermore, the mechanistic pathway inherently supports exceptional impurity control, which is a primary concern for quality assurance teams. The byproducts generated from the phosphonic anhydride reagent are typically phosphonic acid derivatives that exhibit high water solubility. This physicochemical property is exploited during the workup phase, where simple hydrolysis and phase separation effectively remove these impurities from the organic layer containing the desired product. Consequently, the need for extensive chromatographic purification is often eliminated, leading to a cleaner crude product with HPLC purities frequently exceeding 97%. This inherent cleanliness of the reaction profile reduces the burden on downstream processing units and minimizes the risk of introducing foreign contaminants, thereby streamlining the path to GMP-compliant manufacturing.
How to Synthesize Carbamate-Protected Amines Efficiently
The practical implementation of this synthesis route involves a straightforward sequence that is highly amenable to standard reactor configurations found in fine chemical plants. The process begins with the dissolution of the hydroxamic acid substrate and a base, such as triethylamine, in a suitable aprotic solvent like ethyl acetate or THF. Following this, the cyclic phosphonic anhydride is metered into the mixture at controlled low temperatures to manage the exotherm and ensure selective activation. Once the activation is complete, the trapping alcohol is introduced, and the mixture is warmed to facilitate the rearrangement and final product formation. Detailed standardized synthesis steps see the guide below.
- Preparation of the reaction mixture by dissolving hydroxamic acid and a base (such as triethylamine) in an aprotic solvent like ethyl acetate.
- Activation of the hydroxamic acid by metering in a cyclic alkylphosphonic anhydride (e.g., T3P) at controlled low temperatures (-30 to 0°C).
- Addition of the trapping alcohol (e.g., tert-butanol or benzyl alcohol) and heating to facilitate rearrangement and isolation of the protected amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this phosphonic anhydride-mediated rearrangement offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like azides and halogens significantly reduces the regulatory burden and insurance costs associated with storing and handling dangerous chemicals. Moreover, the simplified workup procedure, which relies on aqueous extraction rather than complex purification techniques, translates directly into reduced cycle times and lower consumption of expensive chromatography media. This efficiency gain allows manufacturing facilities to increase throughput without requiring additional capital investment in new equipment, thereby optimizing asset utilization and improving overall return on investment for the production line.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the purification workflow and the high atom economy of the transformation. Since the phosphonic byproducts are water-soluble, the reliance on costly silica gel columns or preparative HPLC is drastically minimized, leading to significant savings in material costs and waste disposal fees. Additionally, the high yields reported, often approaching quantitative levels, mean that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold. The ability to perform the reaction in common solvents like ethyl acetate further contributes to cost efficiency by avoiding the need for specialized or exotic solvent systems.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable reagents such as cyclic phosphonic anhydrides, which are produced on a large industrial scale by major chemical suppliers. Unlike specialized reagents that may suffer from supply bottlenecks or long lead times, these activators are robust and have a well-established global supply network. The mild reaction conditions also reduce the risk of batch failures due to thermal runaways or equipment corrosion, ensuring consistent production schedules. This reliability is critical for maintaining continuous supply to downstream pharmaceutical customers who depend on just-in-time delivery models for their own manufacturing operations.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of gas evolution or highly exothermic events that typically complicate scale-up efforts. The reaction can be safely conducted in standard stainless steel reactors without the need for specialized pressure vessels or cryogenic cooling beyond standard chillers. From an environmental standpoint, the generation of water-soluble byproducts simplifies wastewater treatment protocols, aiding in compliance with increasingly stringent environmental regulations. The reduction in hazardous waste generation aligns with green chemistry principles, enhancing the sustainability profile of the manufacturing site and appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced amine synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing process portfolios and identify potential areas for optimization.
Q: Why is the phosphonic anhydride method superior to traditional Hofmann or Curtius rearrangements?
A: Traditional methods often utilize hazardous reagents like bromine, chlorine, or explosive azides. The phosphonic anhydride method described in CN101208292A operates under mild conditions without toxic halogens or explosive intermediates, ensuring higher safety and stereoselectivity.
Q: What are the typical yields and purity levels achievable with this process?
A: The patent data indicates that this method typically achieves yields between 90% and 100%, with HPLC purities often exceeding 97%. The process minimizes side reactions and epimerization, resulting in highly pure products suitable for pharmaceutical applications.
Q: How does the workup procedure contribute to cost efficiency?
A: The byproducts generated from the phosphonic anhydride are generally water-soluble. This allows for simple phase separation and hydrolysis during workup, eliminating the need for complex chromatographic purification steps and significantly reducing processing time and waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbamate-Protected Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that balance technical excellence with commercial viability. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods like the phosphonic anhydride rearrangement are successfully translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity carbamate-protected amines meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality ensures that our clients receive materials that are ready for immediate use in subsequent synthetic steps without additional purification.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this route can optimize your budget. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Let us help you secure a reliable supply of complex intermediates while driving down your overall manufacturing costs through scientific innovation.
