Optimizing Crizotinib Production: Advanced Chiral Resolution for Commercial Scale-up
Optimizing Crizotinib Production: Advanced Chiral Resolution for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes for high-value oncology therapeutics, and the synthesis of Crizotinib stands as a prime example of process innovation. Patent CN102532106A discloses a novel splitting process for the chiral isomer of a Crizotinib precursor, addressing critical bottlenecks in traditional manufacturing. This technical breakthrough focuses on the efficient preparation of (S)-1-(2,6-dichloro-3-fluorophenyl)ethanol, a pivotal intermediate that dictates the overall stereochemical integrity of the final drug substance. By leveraging a catalytic splitting method combining Boc-L-proline and tosic acid, the process circumvents the limitations of enzymatic hydrolysis and complex chromatographic separations. For a reliable pharmaceutical intermediate supplier, adopting such methodologies is essential to ensure consistent quality and supply continuity for global partners. The following analysis details how this patented approach transforms the production landscape for this vital antitumor molecular targeting medicament.
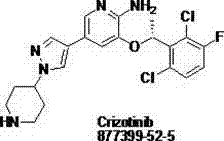
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral intermediates for Crizotinib has relied heavily on biological enzyme methods or cumbersome chemical resolution techniques that hinder industrial scalability. Conventional enzymatic hydrolysis, such as using pork liver esterase, often suffers from prolonged reaction times and severe sensitivity to reaction conditions, leading to inconsistent batch-to-batch performance. Furthermore, traditional laboratory-scale chemical resolutions frequently necessitate the use of protecting groups that require subsequent removal via harsh acidolysis, followed by purification through column chromatography. This reliance on chromatography is a significant drawback for large-scale manufacturing, as it introduces high solvent consumption, complex operational procedures, and difficulties in waste management. The low yields associated with these legacy methods, often hovering around 30% after extensive purification, create substantial economic inefficiencies and supply chain vulnerabilities for procurement teams managing API budgets.
The Novel Approach
The innovative strategy outlined in the patent data introduces a streamlined catalytic splitting system that fundamentally reshapes the efficiency of chiral intermediate production. By employing a combination of Boc-L-proline, tosic acid, and EDCI as a condensing agent, the process achieves a direct and highly selective resolution of the racemic alcohol without the need for enzymatic catalysts. This chemical approach allows for the separation of the S-alcohol and R-alcohol through crystallization and vacuum distillation, effectively bypassing the need for industrial column chromatography. The result is a dramatic simplification of the workflow, where the yield of the desired chiral intermediate reaches 60% with an enantiomeric excess exceeding 99%. This shift from biocatalysis to optimized chemical catalysis not only enhances the robustness of the reaction but also significantly reduces the environmental footprint by minimizing waste acid generation and solvent usage.
Mechanistic Insights into Boc-L-Proline Catalyzed Chiral Resolution
The core of this technological advancement lies in the precise mechanistic interaction between the chiral auxiliary and the racemic substrate during the esterification phase. The use of Boc-L-proline acts as a resolving agent that forms diastereomeric esters with the racemic 1-(2,6-dichloro-3-fluorophenyl)ethanol, creating distinct physical properties that facilitate separation. The addition of tosic acid as a catalyst plays a crucial role in activating the hydroxyl group, generating a highly reactive active ester intermediate that accelerates the reaction kinetics. Crucially, the reaction rate difference between the R and S configurations is amplified under these specific catalytic conditions, allowing the R-configuration to react faster while leaving the desired S-alcohol available for isolation. This kinetic resolution mechanism ensures that the unreacted S-enantiomer can be recovered with high optical purity, avoiding the racemization issues often seen in less controlled environments.
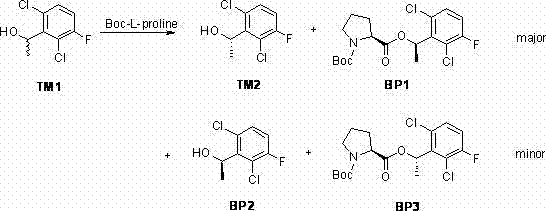
Impurity control is meticulously managed through the optimization of reagent stoichiometry and temperature profiles throughout the resolution process. Experimental data indicates that maintaining the Boc-L-proline equivalent at 0.6 and the EDCI equivalent at 0.8 maximizes the enantiomeric excess while preventing the formation of unwanted by-products. The process avoids the use of strong acidic solutions for deprotection in the early stages, which traditionally leads to the degradation of sensitive chiral centers. Instead, the method utilizes vacuum distillation followed by recrystallization in n-hexane at low temperatures, such as -20°C, to physically separate the target compound from minor impurities. This physical purification strategy ensures that the final intermediate meets stringent purity specifications without the risk of chemical degradation, providing R&D directors with confidence in the structural fidelity of the material.
How to Synthesize Crizotinib Efficiently
The synthesis pathway described integrates several unit operations into a cohesive flow that prioritizes yield and purity at every stage. Starting from the reduction of 2,6-dichloro-3-fluoroacetophenone, the route proceeds through the critical chiral resolution step before advancing to etherification and ring construction. Detailed standardized synthesis steps see the guide below, which outlines the specific parameters for reagents, temperatures, and workup procedures required to replicate this high-efficiency process. Adhering to these protocols ensures that the complex stereochemistry is maintained throughout the multi-step sequence, ultimately delivering a final product that complies with global pharmacopeial standards.
- Perform catalytic splitting of racemic 1-(2,6-dichloro-3-fluorophenyl)ethanol using Boc-L-proline and tosic acid to isolate the S-enantiomer.
- Execute Mitsunobu etherification followed by nitro reduction and selective bromination to construct the pyridine core.
- Complete the synthesis via palladium-catalyzed coupling and final acidolysis to yield Crizotinib with >99% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized synthesis route offers tangible benefits that extend beyond mere technical feasibility into the realm of strategic sourcing. The elimination of column chromatography and the reduction in reaction cycles directly translate to a shorter manufacturing lead time, allowing for more responsive inventory management and faster time-to-market for downstream drug products. By removing the dependency on expensive enzymatic catalysts and reducing the volume of organic solvents required for purification, the overall cost of goods sold is significantly lowered, enabling more competitive pricing structures for long-term supply agreements. Furthermore, the robustness of the chemical catalytic system ensures high batch consistency, reducing the risk of production delays caused by failed batches or out-of-specification results.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive enzymatic reagents with readily available chemical catalysts and eliminating the capital-intensive chromatography step. The improved yield of 60% in the resolution step, compared to lower yields in prior art, means less raw material is wasted per kilogram of final product, optimizing the utilization of starting materials. Additionally, the simplified workup procedure reduces labor hours and energy consumption associated with solvent recovery and waste treatment, contributing to a leaner operational expenditure model for the manufacturing facility.
- Enhanced Supply Chain Reliability: Utilizing stable chemical reagents like tosic acid and EDCI ensures that the supply chain is not vulnerable to the biological variability or shelf-life limitations associated with enzyme-based processes. The ability to scale this reaction from laboratory benchtop to multi-ton production without significant re-engineering provides supply chain heads with the assurance of continuity even during periods of high market demand. This reliability is critical for maintaining the production schedules of life-saving oncology medications, where interruptions can have severe consequences for patient care and regulatory compliance.
- Scalability and Environmental Compliance: The avoidance of strong acid deprotection steps and the reduction in solvent usage align with modern green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. The process generates less hazardous waste, simplifying the disposal process and reducing the environmental liability for the manufacturing site. This eco-friendly profile not only mitigates regulatory risk but also enhances the corporate social responsibility standing of the supply chain, making it a preferred choice for multinational pharmaceutical companies with stringent sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method in a GMP environment. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential manufacturing partners. Understanding these details is crucial for evaluating the feasibility of technology transfer and the integration of this route into existing production lines.
Q: How does the new catalytic splitting method improve upon enzymatic hydrolysis?
A: The novel chemical catalytic splitting method eliminates the need for expensive enzymes and lengthy reaction times, offering a more robust and scalable alternative with simplified purification.
Q: What is the optical purity achieved with this synthesis route?
A: The optimized process consistently achieves an enantiomeric excess (ee) of greater than 99%, meeting stringent regulatory requirements for oncology therapeutics.
Q: Does this process require column chromatography for purification?
A: No, the method utilizes vacuum distillation and recrystallization, effectively removing the need for industrial-scale column chromatography and reducing solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crizotinib Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate complex patented synthesis routes like this into commercial reality for our global clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch, guaranteeing that the chiral integrity and impurity profiles meet the highest industry standards.
We invite you to collaborate with us to leverage these process innovations for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals for Crizotinib and related oncology intermediates.
