Advanced Cefprozil Mother Liquor Recycling Technology for Commercial API Production
The pharmaceutical industry is currently facing intensifying pressure to optimize the manufacturing processes of broad-spectrum antibiotics, particularly second-generation cephalosporins like Cefprozil. Patent CN108659008B introduces a groundbreaking methodology for the mechanical application and recycling of cefprozil mother liquor, addressing critical inefficiencies in traditional synthesis routes. This innovation focuses on the direct reintegration of treated mother liquor into the synthesis reaction stream, bypassing complex and solvent-heavy recovery stages that have historically plagued production lines. By implementing a strategic pH adjustment using saturated ferric chloride (FeCl3) solutions, the process effectively complexes and removes stubborn impurities such as 7-aminocephalosporanic acid (7-ACA) derivatives before they can contaminate the final active pharmaceutical ingredient (API). This technical advancement not only streamlines the operational workflow but also delivers substantial environmental benefits by minimizing the discharge of hazardous organic solvents. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and economically viable antibiotic manufacturing, ensuring that high-purity standards are met while simultaneously reducing the ecological footprint of large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial processes for cefprozil production often suffer from significant resource inefficiencies, primarily due to the handling of mother liquors generated during the refinement stages. In standard protocols, the mother liquor contains a non-negligible amount of residual cefprozil along with various by-products and solvents, necessitating a dedicated recovery process to reclaim the valuable API. This recovery typically involves the extensive use of dimethylformamide (DMF) and acetone to precipitate cefprozil dimethylformamide solvates, followed by further refining steps to obtain the final product. Such methods are not only capital intensive, requiring additional reactor volume and energy for solvent evaporation and recycling, but they also generate substantial volumes of chemical waste that require costly treatment. Furthermore, the repeated thermal stress and chemical exposure during these recovery loops can lead to product degradation, potentially compromising the impurity profile and failing to meet the stringent requirements of modern pharmacopoeias. The reliance on excessive solvent volumes also exacerbates safety risks associated with flammability and toxicity, creating a bottleneck for manufacturers aiming to enhance their environmental, social, and governance (ESG) performance metrics.
The Novel Approach
The novel approach disclosed in the patent data revolutionizes this workflow by treating the mother liquor not as waste to be processed separately, but as a valuable feedstock to be directly integrated into the primary synthesis reaction. Instead of isolating the residual cefprozil through solvent-heavy precipitation, the process employs a targeted chemical treatment where saturated FeCl3 is added to the mother liquor to adjust the pH to a highly acidic range of 0.8 to 1.2. This specific chemical environment promotes the formation of complexes with key impurities, effectively sequestering them and preventing their interference in the subsequent reaction steps. The treated mother liquor is then introduced directly into the cefprozil synthesis reaction solution immediately after the condensation reaction is complete. This integration allows the residual cefprozil in the mother liquor to act as a seed or simply accumulate with the newly formed product, thereby boosting the overall yield without the need for intermediate isolation. By eliminating the standalone recovery unit operation, the process drastically cuts down on the consumption of DMF and acetone, simplifying the plant layout and reducing the total cycle time for batch production while maintaining a product quality that conforms to new pharmacopoeia standards.
Mechanistic Insights into FeCl3-Mediated Impurity Removal and Condensation
The core chemical innovation lies in the precise manipulation of coordination chemistry to purify the reaction stream in situ. When saturated FeCl3 solution is introduced to the cefprozil mother liquor, the ferric ions act as a Lewis acid, coordinating with electron-rich functional groups present on impurity molecules such as unreacted 7-ACA or degraded beta-lactam fragments. This complexation alters the solubility properties of these impurities, facilitating their separation during the subsequent liquid-liquid extraction phase. The pH control is critical; maintaining the acidity between 0.8 and 1.2 ensures that the beta-lactam ring of the desired cefprozil remains stable while maximizing the binding affinity of iron for the contaminants. Following this purification step, the treated stream enters the main reaction vessel where the condensation of the protected cephalosporin nucleus with the activated side chain occurs. The presence of the recycled material does not inhibit the reaction kinetics; rather, the controlled temperature profile of 5-8°C during the mixing phase ensures thermodynamic stability. The subsequent addition of ammonia water to adjust the pH to a near-neutral range of 6.3 to 6.8 triggers the crystallization of the cefprozil crude product, leveraging the supersaturation created by the combined mass of newly synthesized and recycled API.
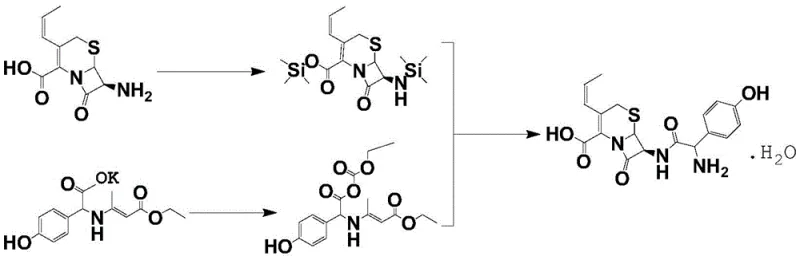
Furthermore, the impurity control mechanism extends to the crystallization dynamics. By cooling the aqueous phase to below -5°C before the addition of DMF and acetone wash liquids, the process maximizes the recovery of the product while keeping soluble impurities in the supernatant. The washing steps with DMF and acetone are optimized to remove residual solvents and salts, such as ammonium chloride formed during neutralization, without dissolving the product cake. The final refining step involves dissolving the crude product in purified water at controlled temperatures (35-45°C) and re-crystallizing upon cooling. This recrystallization acts as a final polishing step, ensuring that the metal residues from the FeCl3 treatment are reduced to acceptable limits and that the crystal habit is suitable for downstream formulation. The result is a high-purity API with reported purity levels exceeding 99.7%, demonstrating that the recycling strategy does not compromise the stringent quality specifications required for injectable or oral antibiotic formulations.
How to Synthesize Cefprozil Efficiently
The implementation of this mother liquor recycling protocol requires strict adherence to temperature controls and stoichiometric ratios to ensure reproducibility and safety. The process begins with the preparation of the protected nucleus via silylation of 7-APRA using hexamethyldisilazane and trimethylchlorosilane, followed by the activation of the p-hydroxyphenylglycine side chain using ethyl chloroformate. Once the condensation is complete, the treated mother liquor is introduced, marking the divergence from traditional linear synthesis. Operators must monitor the exotherm during the acid addition and maintain the stirring times precisely (typically 30 minutes) to allow for complete complexation and phase separation. The detailed standardized synthetic steps, including specific reagent grades and equipment specifications required for GMP compliance, are outlined in the technical guide below.
- Treat the cefprozil mother liquor with saturated FeCl3 solution and adjust pH to 0.8-1.2 to complex and remove impurities like 7-ACA.
- Introduce the treated mother liquor directly into the cefprozil synthesis reaction solution after condensation, maintaining temperature at 5-8°C.
- Separate the aqueous phase, cool below -5°C, add DMF and acetone wash liquid, then adjust pH with ammonia water to crystallize the crude product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into immediate and tangible operational improvements that impact the bottom line. The most significant advantage is the drastic reduction in solvent procurement costs, as the process eliminates the need to purchase large volumes of DMF and acetone specifically for mother liquor recovery. Since these solvents are among the most expensive and regulated chemicals in pharmaceutical manufacturing, reducing their consumption directly lowers the variable cost per kilogram of API produced. Additionally, the simplification of the workflow reduces the burden on waste management teams, as there is less hazardous waste to treat and dispose of, leading to lower environmental compliance costs. The ability to reuse the mother liquor directly also means that the facility can achieve higher throughput with the existing reactor infrastructure, effectively increasing capacity without capital expenditure on new tanks or distillation columns. This operational leanness enhances the resilience of the supply chain against fluctuations in raw material availability and pricing.
- Cost Reduction in Manufacturing: The elimination of the separate mother liquor recovery stage results in substantial cost savings by removing an entire unit operation from the production schedule. This reduction in processing steps lowers energy consumption associated with heating, cooling, and vacuum drying, while simultaneously decreasing labor hours required for monitoring and handling. The qualitative reduction in solvent usage means that the facility is less exposed to the volatility of petrochemical-derived solvent markets, providing more predictable budgeting for long-term production contracts. Furthermore, the increased overall yield means that more saleable product is generated from the same amount of expensive starting materials like 7-APRA, effectively spreading the fixed costs over a larger output volume and improving the gross margin profile of the manufacturing line.
- Enhanced Supply Chain Reliability: By streamlining the production process, the lead time for manufacturing batches is significantly shortened, allowing for faster response to market demand spikes. The robustness of the FeCl3 treatment ensures consistent product quality batch after batch, reducing the risk of failed quality control tests that could delay shipments. This reliability is crucial for maintaining continuous supply agreements with major pharmaceutical partners who require just-in-time delivery of critical antibiotic intermediates. The process also reduces dependency on external solvent recovery services, internalizing the efficiency and securing the production schedule against third-party logistical bottlenecks. Consequently, the supply chain becomes more agile and capable of scaling up production rapidly when needed without the lag time associated with complex solvent recycling loops.
- Scalability and Environmental Compliance: The technology is inherently scalable, utilizing standard mixing and filtration equipment that is readily available in most multipurpose API plants. This ease of scale-up facilitates the transition from pilot-scale validation to commercial production of complex pharmaceutical intermediates with minimal technical risk. From an environmental perspective, the significant reduction in organic solvent discharge aligns with increasingly strict global regulations on volatile organic compound (VOC) emissions. This proactive compliance posture mitigates the risk of regulatory fines and enhances the corporate reputation of the manufacturer as a sustainable partner. The lower E-factor (mass of waste per mass of product) achieved by this process makes it an attractive option for companies aiming to green their supply chains and meet sustainability targets set by international health organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cefprozil mother liquor recycling technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and quality implications for potential adopters. Understanding these details is essential for R&D teams evaluating the technology for technology transfer and for procurement officers assessing the long-term value proposition.
Q: How does the FeCl3 treatment improve product purity in cefprozil synthesis?
A: The addition of saturated FeCl3 solution facilitates the complexation of specific impurities, such as 7-aminocephalosporanic acid (7-ACA) derivatives, allowing them to be effectively separated from the desired product stream before crystallization, thereby ensuring high purity standards.
Q: What are the primary cost drivers reduced by this mother liquor application process?
A: This process significantly lowers operational costs by eliminating the need for a separate, energy-intensive mother liquor recovery unit operation and drastically reducing the consumption of expensive organic solvents like dimethylformamide (DMF) and acetone.
Q: Is this recycling method compatible with large-scale commercial manufacturing?
A: Yes, the method utilizes standard chemical engineering unit operations such as pH adjustment, liquid-liquid separation, and crystallization, making it highly scalable for industrial production ranging from pilot batches to multi-ton annual capacities without requiring specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefprozil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficient production of beta-lactam antibiotics is critical for global health security. Our technical team has extensively analyzed advanced recycling methodologies like the one described in CN108659008B to optimize our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every gram of cefprozil intermediate we supply meets stringent purity specifications. Our rigorous QC labs are equipped to detect trace impurities and residual solvents, guaranteeing that our products are ready for immediate use in downstream API synthesis without additional purification burdens. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the highest international standards.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency. Let us be your partner in achieving a more sustainable and cost-effective pharmaceutical manufacturing future.
