Advanced Chemical Synthesis of Cefprozil: Overcoming Impurity M for Global Compliance
Introduction to Advanced Cefprozil Manufacturing
The pharmaceutical landscape demands increasingly rigorous quality standards, particularly for second-generation cephalosporins like Cefprozil. Patent CN112694487B introduces a transformative preparation method that addresses the critical limitations of existing synthetic routes, specifically targeting the stringent requirements of the European Union Pharmacopoeia (EP). Traditional chemical synthesis often struggles with yield optimization and impurity profiles, frequently failing to meet international regulatory benchmarks without extensive purification. This novel approach leverages a sophisticated silanization strategy combined with a precisely controlled mixed anhydride acylation to deliver a product with exceptional purity and consistency. By integrating specific molar ratios of key reagents such as p-hydroxyphenylglycine dane potassium salt (HPDS) and ethyl chloroformate (CBE), the process effectively suppresses the formation of critical impurities while enhancing overall throughput.
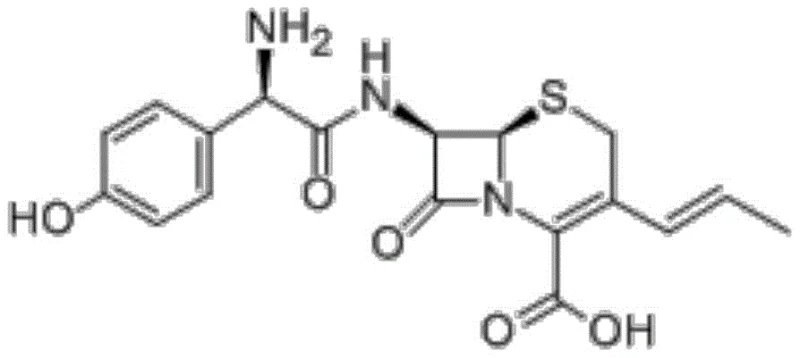
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for Cefprozil have historically relied on tetramethylguanidine (TMG) mediated acylation or enzymatic processes that, while effective to a degree, suffer from significant industrial drawbacks. The conventional chemical route typically involves dissolving APCA in TMG and reacting it with a mixed anhydride formed from HPDS and pivaloyl chloride at ultra-low temperatures. This method is plagued by low molar yields, often falling below 80%, and produces a faint yellow powder that, while compliant with domestic pharmacopoeias, fails to meet the stricter impurity limits of the EP and USP. Specifically, the uncontrolled reaction environment facilitates the generation of Impurity M, (Z)-Cefprozil Ethoxyformate, which frequently exceeds the allowable threshold of 0.30%. Furthermore, the reliance on specific enzymatic pathways, though greener, introduces variability in batch-to-batch consistency and scalability challenges that hinder reliable mass production for global supply chains.
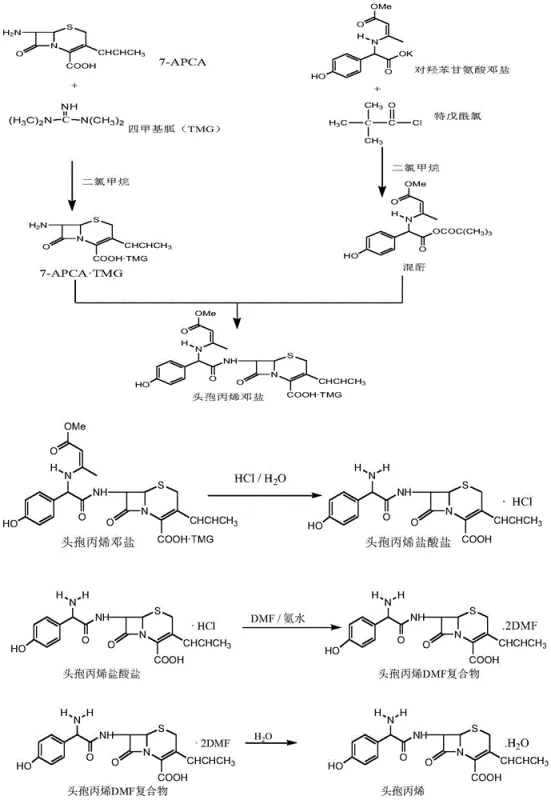
The Novel Approach
The patented methodology fundamentally reengineers the acylation landscape by introducing a silanization pre-step for the 7-APCA nucleus, utilizing trimethylchlorosilane (TMC) and hexamethyldisilazane (HMDS) in dichloromethane. This activation step creates a more reactive and stable intermediate that couples efficiently with the mixed anhydride generated from HPDS, DMF, and CBE. Crucially, the process incorporates the addition of a Cefprozil Naphthol compound during the crystallization of the DMF complex, which acts to enhance crystal growth and recovery from the aqueous phase. This dual-strategy of chemical activation and crystallization engineering results in a white, high-purity product with molar yields exceeding 85%. The rigorous control over reaction temperatures, maintained between -50°C and -75°C during acylation, ensures stereochemical integrity and minimizes side reactions, thereby securing compliance with international quality standards.
Mechanistic Insights into Silanization and Mixed Anhydride Acylation
The core of this technological breakthrough lies in the precise manipulation of the acylation mechanism through silanization. By converting the amino group of the 7-APCA nucleus into a silylated derivative, the nucleophilicity is modulated to favor the desired amide bond formation while reducing susceptibility to hydrolysis or rearrangement. The subsequent reaction with the mixed anhydride, formed in situ from HPDS and ethyl chloroformate at cryogenic temperatures (-60°C to -70°C), proceeds with high regioselectivity. The patent specifies a narrow molar window for HPDS relative to APCA (1.05 to 1.20 equivalents) and CBE relative to HPDS (0.95 to 1.02 equivalents). This stoichiometric precision is vital; an excess of ethyl chloroformate can lead to over-acylation or the formation of ethoxyformate esters, directly correlating to the generation of Impurity M. The mechanism ensures that the electrophilic attack occurs exclusively at the intended nitrogen center, preserving the delicate beta-lactam ring structure essential for antibiotic activity.
Impurity control is further achieved through a unique crystallization intervention involving the Cefprozil Naphthol compound. During the workup phase, after acidification and layering, DMF is added to the aqueous phase along with this naphthol derivative. This addition promotes the selective precipitation of the Cefprozil DMF complex, effectively excluding impurities from the crystal lattice. The naphthol compound can be recovered from the mother liquor and recycled in subsequent batches, creating a closed-loop system that minimizes waste. This step is critical for reducing Impurity M levels to below 0.10%, far surpassing the EP requirement of 0.30%. The final conversion step, where the DMF complex is hydrolyzed in water at 30-38°C, releases the free acid form of Cefprozil with high structural fidelity, ensuring the final API possesses the correct (6R, 7R) stereochemistry required for biological efficacy.
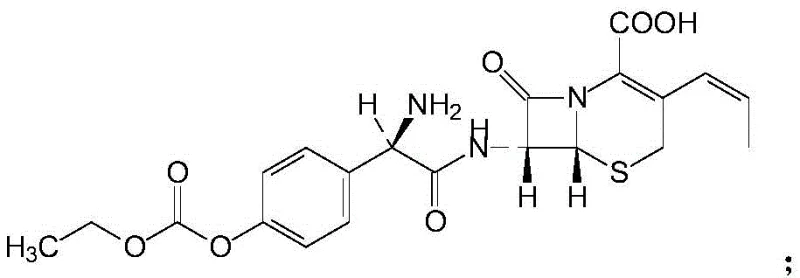
How to Synthesize Cefprozil Efficiently
The synthesis of Cefprozil via this patented route requires meticulous attention to thermal gradients and reagent addition rates to maximize yield and purity. The process begins with the silanization of the cephalosporin nucleus, followed by the generation of a reactive mixed anhydride species under inert atmosphere. These two streams are then converged under strictly controlled cryogenic conditions to effect acylation. Following the reaction, a specialized workup involving acidification, solvent extraction, and seeded crystallization with the naphthol additive isolates the intermediate DMF complex. Finally, a温和 hydrolytic conversion yields the target API. For laboratory and pilot-scale execution, adhering to the specific molar ratios and temperature profiles outlined in the patent is essential to replicate the high-purity results. Detailed standardized operating procedures for each unit operation are provided in the technical guide below.
- Perform silanization on 7-APCA using TMC and HMDS in dichloromethane under reflux conditions.
- Generate a mixed anhydride solution by reacting p-hydroxyphenylglycine dane potassium salt (HPDS) with ethyl chloroformate (CBE) at low temperatures.
- Conduct acylation by mixing the silanized solution with the anhydride, followed by acidification, DMF crystallization, and hydrolytic conversion to obtain the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this synthesis route offers substantial operational benefits beyond mere regulatory compliance. The shift from enzymatic or low-yield chemical processes to this robust silanization-based method stabilizes the supply of high-purity Cefprozil intermediates. By eliminating the variability associated with biological catalysts and optimizing chemical yields through stoichiometric control, manufacturers can guarantee consistent delivery schedules. The ability to recover and recycle the Cefprozil Naphthol compound from mother liquors significantly reduces raw material consumption, translating to direct cost efficiencies in large-scale manufacturing. Furthermore, the process utilizes common industrial solvents like dichloromethane and DMF, avoiding the need for exotic or hard-to-source reagents that often bottleneck production timelines.
- Cost Reduction in Manufacturing: The optimized stoichiometry and the recycling of the naphthol additive drastically reduce the consumption of expensive starting materials like HPDS and APCA. By achieving molar yields greater than 85% compared to the sub-80% yields of conventional methods, the cost per kilogram of the active ingredient is significantly lowered. Additionally, the elimination of complex purification steps required to remove Impurity M reduces downstream processing costs and solvent usage, leading to a leaner and more economical production profile.
- Enhanced Supply Chain Reliability: This chemical route is highly scalable and less susceptible to the fluctuations that affect enzymatic processes, such as enzyme stability or fermentation batch failures. The use of standard chemical reagents ensures that raw material sourcing remains stable even during market volatility. The robust nature of the silanization and acylation steps allows for seamless scale-up from pilot plants to multi-ton commercial reactors, ensuring that supply commitments to global pharmaceutical partners can be met without interruption.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by maximizing atom economy through high yields and minimizing waste via the recovery of the naphthol compound. The reduction in total impurities means less solvent is required for recrystallization and purification, lowering the environmental footprint of the manufacturing facility. This alignment with sustainability goals not only meets regulatory expectations but also positions the supply chain favorably for future environmental audits and compliance certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefprozil preparation method. These insights are derived directly from the experimental data and claims within patent CN112694487B, providing clarity on how this technology resolves historical manufacturing pain points. Understanding these specifics is crucial for technical teams evaluating the feasibility of adopting this route for commercial API production.
Q: How does this method control Impurity M levels?
A: By strictly controlling the molar ratios of HPDS to APCA (1.05-1.20:1) and CBE to HPDS (0.95-1.02:1), alongside precise temperature management during acylation, the formation of (Z)-Cefprozil Ethoxyformate (Impurity M) is suppressed to below 0.10%.
Q: What is the role of the Cefprozil Naphthol compound in this process?
A: The Cefprozil Naphthol compound is added during the DMF crystallization step to act as a seed or co-crystallizer, significantly increasing the yield of the Cefprozil DMF complex and allowing for recovery from mother liquors.
Q: Does this process meet European Pharmacopoeia standards?
A: Yes, unlike conventional methods which often fail EP standards due to high Impurity M content, this patented route consistently achieves purity greater than 99.5% and Impurity M levels well below the 0.30% limit.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefprozil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and proven scalability. Our team specializes in the commercialization of complex antibiotic intermediates, leveraging advanced chemical engineering to bridge the gap between laboratory innovation and industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate temperature controls and stoichiometric precision required for this Cefprozil synthesis are maintained at every scale. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch meets stringent purity specifications and international pharmacopoeia standards.
We invite global pharmaceutical partners to collaborate with us to optimize their Cefprozil supply chain. By leveraging our technical capabilities, you can secure a reliable source of high-quality API intermediates that comply with the strictest global regulations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
