Advanced One-Pot Synthesis of Triazolo Pyrimidine Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for heterocyclic scaffolds that possess broad-spectrum biological activities, and patent CN112142750B presents a transformative approach to synthesizing 4,7-dihydro-[1,2,4]triazolo[1,5-a]pyrimidine derivatives. This specific class of compounds serves as a critical backbone for numerous bioactive agents, including herbicides like flumetsulam and bactericides such as ametoctradin, necessitating a production method that balances high yield with environmental sustainability. The disclosed invention utilizes a novel acidic ionic liquid catalyst within a specialized isopropanol-acetone aqueous solvent system to facilitate a three-component one-pot condensation reaction. By integrating aromatic aldehydes, active methylene compounds, and 3-amino-1,2,4-triazole under mild reflux conditions, this technology effectively overcomes the historical limitations of multi-step syntheses that often suffer from harsh conditions and low atom economy. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in process chemistry, offering a pathway to high-purity pharmaceutical intermediates with reduced operational complexity and enhanced green chemistry metrics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the preparation of 4,7-dihydro-[1,2,4]triazolo[1,5-a]pyrimidine derivatives has relied upon multi-step synthetic routes that are inherently inefficient and fraught with logistical challenges for large-scale manufacturing. Conventional methodologies often require the use of strong mineral acids or expensive transition metal catalysts that demand rigorous anhydrous conditions and elevated temperatures, leading to significant energy consumption and safety hazards in a plant setting. Furthermore, these older processes frequently generate complex mixtures of byproducts due to poor selectivity, necessitating tedious purification steps such as column chromatography or multiple recrystallizations that drastically reduce the overall yield and increase the cost of goods sold. The reliance on volatile organic solvents and the generation of heavy metal waste streams also pose severe environmental compliance issues, making it difficult for manufacturers to meet increasingly stringent global regulatory standards for pharmaceutical production. Consequently, the industry has long suffered from supply chain bottlenecks where the difficulty in scaling these complex routes results in inconsistent availability and inflated pricing for key drug intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN112142750B introduces a streamlined one-pot protocol that leverages the unique properties of SO3H-functionalized piperazinium-based dicationic ionic liquids to drive the condensation reaction with exceptional efficiency. This innovative approach allows for the direct combination of raw materials in a benign isopropanol-acetone-water solvent system, where the reaction proceeds smoothly under reflux to afford the target molecules in high yields without the need for intermediate isolation. The dual functionality of the solvent system is particularly noteworthy, as it not only supports the catalytic cycle but also induces the precipitation of the product upon cooling, thereby simplifying the work-up procedure to a mere filtration and washing step. 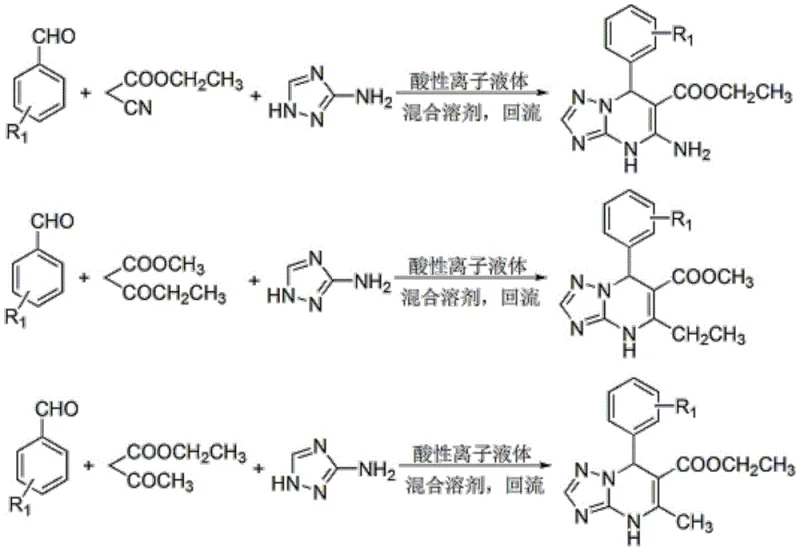
This elimination of extraction and complex purification stages translates directly into reduced processing time and lower solvent usage, addressing the core pain points of cost and throughput that plague conventional manufacturing. Moreover, the versatility of this method is demonstrated by its compatibility with a wide range of substituted aromatic aldehydes and active methylene compounds, enabling the rapid synthesis of diverse derivative libraries for drug discovery programs without requiring extensive process re-optimization for each new substrate.
Mechanistic Insights into Acidic Ionic Liquid Catalyzed Condensation
The cornerstone of this advanced synthesis lies in the sophisticated design of the acidic ionic liquid catalyst, which features a piperazinium dicationic core functionalized with sulfonic acid groups to provide strong Brønsted acidity. 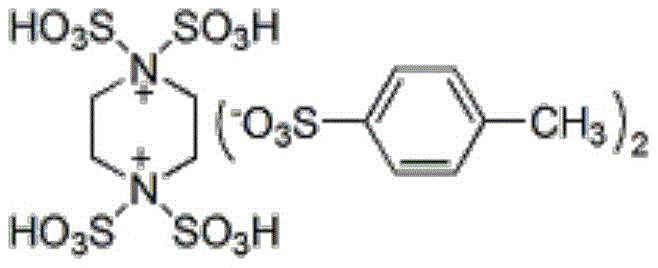
Unlike traditional homogeneous acid catalysts that are difficult to recover, this ionic liquid forms a stable catalytic system that activates the carbonyl group of the aromatic aldehyde and the active methylene compound simultaneously, facilitating the Knoevenagel condensation and subsequent cyclization with 3-amino-1,2,4-triazole. The dicationic nature of the catalyst enhances its solubility in the polar reaction medium while maintaining high thermal stability, ensuring that the catalytic activity remains consistent throughout the reaction duration even at reflux temperatures. From a mechanistic standpoint, the sulfonic acid protons act as efficient proton donors to accelerate the nucleophilic attack and dehydration steps, which are typically the rate-determining steps in such heterocyclic formations. This precise activation mechanism minimizes side reactions such as polymerization or hydrolysis, resulting in a cleaner reaction profile that is essential for producing pharmaceutical-grade intermediates with tight impurity specifications.
Furthermore, the interaction between the ionic liquid and the isopropanol-acetone-water solvent creates a microenvironment that stabilizes the transition states of the reaction, further lowering the activation energy required for the cyclization to occur. This synergistic effect allows the reaction to proceed rapidly, often completing within 30 to 60 minutes, which is a dramatic improvement over the prolonged reaction times associated with conventional solid acid catalysts. The high selectivity of this catalytic system ensures that the final product crystallizes with high purity directly from the reaction mixture, effectively acting as a self-purifying process that reduces the burden on downstream quality control laboratories. For process chemists, understanding this mechanism is crucial for troubleshooting and optimizing the reaction parameters, such as the molar ratio of reactants and the specific composition of the solvent mixture, to maximize yield and minimize waste generation in a commercial setting.
How to Synthesize 4,7-Dihydro-[1,2,4]triazolo[1,5-a]pyrimidine Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters regarding solvent ratios and catalyst loading to ensure reproducibility and safety. The process begins with the preparation of the ternary solvent system, followed by the sequential addition of substrates and the catalyst under controlled stirring to prevent localized exotherms. Detailed standard operating procedures regarding temperature ramping rates and reflux monitoring are critical to achieving the high yields reported in the patent data, and operators must be trained to recognize the endpoint of the reaction via thin-layer chromatography to avoid over-processing.
- Prepare the reaction vessel by adding an isopropanol-acetone aqueous solution, followed by the sequential addition of aromatic aldehyde, active methylene compound, and 3-amino-1,2,4-triazole under stirring at room temperature.
- Introduce the acidic ionic liquid catalyst and heat the mixture to reflux temperature in an oil bath, maintaining uniform heating until the raw material point disappears via TLC monitoring.
- Cool the reaction mixture naturally to room temperature to precipitate the solid product, then separate via filtration, wash with anhydrous ethanol, and dry under vacuum to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-catalyzed process offers a compelling value proposition centered around cost reduction in pharmaceutical intermediate manufacturing and enhanced supply security. The ability to recycle the catalytic system and the solvent mixture directly after product separation means that the consumption of fresh raw materials is drastically minimized, leading to substantial long-term savings on input costs without compromising product quality. Unlike processes that rely on precious metal catalysts which are subject to volatile market pricing and supply constraints, the organic ionic liquid used here is synthesized from readily available precursors, ensuring a stable and predictable supply chain for the catalyst itself. The simplification of the purification process eliminates the need for expensive chromatography resins and large volumes of organic solvents for extraction, which significantly lowers the operational expenditure related to waste disposal and solvent recovery infrastructure.
- Cost Reduction in Manufacturing: The elimination of complex purification steps such as column chromatography and multiple recrystallizations directly reduces labor hours and utility consumption, resulting in a leaner manufacturing process with a lower cost base. By avoiding the use of expensive transition metal catalysts, the process removes the necessity for costly metal scavenging steps and rigorous heavy metal testing, further driving down the total cost of production. The high atom economy of the one-pot reaction ensures that the majority of the starting materials are converted into the desired product, minimizing the financial loss associated with unreacted feedstock and byproduct formation. Additionally, the mild reaction conditions reduce the energy load on heating and cooling systems, contributing to a more energy-efficient production facility that aligns with modern sustainability goals.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in raw material quality ensures consistent output, reducing the risk of batch failures that can disrupt supply schedules for downstream API manufacturers. Since the catalyst can be reused multiple times with minimal loss of activity, the dependency on frequent catalyst replenishment is reduced, smoothing out the procurement cycle and inventory management requirements. The use of common industrial solvents like isopropanol and acetone, rather than exotic or regulated chemicals, mitigates the risk of supply interruptions due to regulatory changes or logistics bottlenecks. This reliability makes the method particularly attractive for long-term supply agreements where continuity of supply is a critical key performance indicator for pharmaceutical clients.
- Scalability and Environmental Compliance: The one-pot nature of the reaction simplifies the equipment requirements, allowing for easy scale-up from laboratory glassware to large-scale stainless steel reactors without significant engineering modifications. The reduction in solvent usage and the absence of heavy metal contaminants simplify the wastewater treatment process, ensuring that the facility remains compliant with strict environmental discharge regulations. The biodegradable nature of the ionic liquid catalyst further enhances the environmental profile of the process, supporting corporate social responsibility initiatives and improving the company's standing with eco-conscious stakeholders. This scalability ensures that the method can meet the growing global demand for triazolo pyrimidine derivatives without encountering the technical barriers often associated with scaling complex multi-step syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology, providing clarity for potential partners evaluating its feasibility for their specific applications. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for assessing the fit of this technology within existing manufacturing portfolios and for planning future capacity expansions.
Q: What are the primary advantages of using acidic ionic liquids in this synthesis?
A: The use of SO3H-functionalized piperazinium-based dicationic ionic liquids provides superior catalytic activity and selectivity compared to traditional solid acids, significantly reducing reaction time and byproduct formation while allowing for catalyst recyclability.
Q: How does the solvent system contribute to process efficiency?
A: The specific isopropanol-acetone-water mixture acts not only as a reaction medium but also facilitates product precipitation upon cooling, eliminating the need for complex extraction or recrystallization steps and simplifying downstream processing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for scalability with mild reaction conditions, easy catalyst recovery, and a simplified work-up procedure that minimizes waste generation, making it highly viable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,7-Dihydro-[1,2,4]triazolo[1,5-a]pyrimidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN112142750B for securing the supply of critical pharmaceutical intermediates in a competitive global market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and risk-mitigated. We are committed to delivering high-purity 4,7-dihydro-[1,2,4]triazolo[1,5-a]pyrimidine derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging our expertise in green chemistry and ionic liquid technologies, we can help your organization optimize its supply chain and achieve significant cost efficiencies in the manufacturing of complex heterocyclic building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements and volume needs. Please contact us to request a Customized Cost-Saving Analysis that details the potential economic benefits of switching to this greener, more efficient process for your specific application. Our team is ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and sourcing decisions, ensuring that you have all the necessary information to move forward with confidence.
