Scalable Synthesis of Triazolopyrimidine Derivatives Using Acidic Ionic Liquids for Global Pharma Supply
Introduction to Advanced Green Synthesis Technologies
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for producing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN112142750A, which discloses a novel preparation method for 4,7-dihydro-[1,2,4]triazolo[1,5-a]pyrimidine derivatives. These compounds serve as critical building blocks in the development of broad-spectrum biologically active agents, including herbicides like flumetsulam and fungicides such as ametoctradin. The disclosed technology leverages an acidic ionic liquid catalyst within a specialized isopropanol-acetone aqueous solution to facilitate a one-pot multicomponent condensation reaction. This approach represents a paradigm shift from traditional multi-step syntheses, offering a robust solution for reliable pharmaceutical intermediate supplier networks aiming to enhance supply chain resilience while adhering to stricter environmental regulations.
For R&D directors and process chemists, the significance of this patent lies in its ability to overcome the inherent limitations of conventional routes, such as harsh reaction conditions, low atom economy, and difficult purification protocols. By utilizing a recyclable catalytic system that functions effectively under mild reflux conditions, the process ensures high product yields and exceptional purity without the need for extensive downstream processing. This technological advancement not only streamlines the manufacturing workflow but also aligns with the global push towards green chemistry principles, making it an attractive option for cost reduction in API manufacturing where margin compression is a constant challenge.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazolopyrimidine derivatives has been plagued by inefficient multi-step procedures that rely on hazardous organic solvents and stoichiometric amounts of reagents. Traditional methods often involve separate steps for Knoevenagel condensation followed by cyclization, requiring intermediate isolation and purification via column chromatography or repeated recrystallization. These processes are not only time-consuming and labor-intensive but also generate substantial quantities of chemical waste, leading to high disposal costs and environmental liabilities. Furthermore, the use of strong acids or bases in conventional catalysis can lead to side reactions, resulting in complex impurity profiles that complicate regulatory approval and increase the burden on quality control laboratories.
Another critical drawback of existing technologies is the poor scalability and limited substrate scope. Many conventional catalysts suffer from deactivation or difficult recovery, preventing their reuse and driving up the overall cost of goods sold (COGS). The reliance on volatile organic compounds (VOCs) as solvents further exacerbates safety risks in large-scale production facilities. Consequently, manufacturers face significant hurdles in achieving consistent batch-to-batch reproducibility, which is essential for maintaining the stringent quality standards required by global regulatory bodies. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and reduced responsiveness to market demands for key pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology described in CN112142750A introduces a highly efficient one-pot strategy that consolidates multiple reaction steps into a single operational unit. By employing an acidic ionic liquid catalyst, specifically a piperazinium-based dicationic species, the reaction proceeds with remarkable selectivity and speed. The use of an isopropanol-acetone-water ternary solvent system is particularly innovative, as it serves a dual purpose: facilitating the reaction kinetics and acting as an anti-solvent for product precipitation. This eliminates the need for energy-intensive distillation or extraction steps, allowing the high-purity solid product to simply crystallize out of the solution upon cooling.
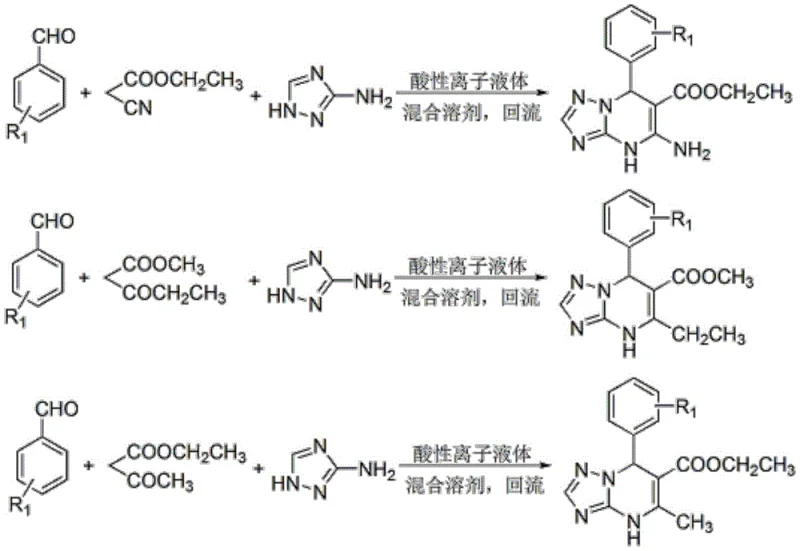
The versatility of this novel approach is demonstrated by its broad substrate tolerance, accommodating various aromatic aldehydes and active methylene compounds to produce a diverse library of derivatives. As illustrated in the reaction scheme, the process seamlessly integrates aromatic aldehydes, active methylene components like ethyl cyanoacetate or beta-keto esters, and 3-amino-1,2,4-triazole. The result is a streamlined workflow that drastically reduces solvent consumption and simplifies the isolation procedure. This efficiency translates directly into commercial advantages, enabling manufacturers to scale up production rapidly while maintaining tight control over impurity levels and ensuring a stable supply of high-quality intermediates for downstream drug synthesis.
Mechanistic Insights into Acidic Ionic Liquid Catalysis
The core of this technological advancement lies in the unique properties of the acidic ionic liquid catalyst, whose structure is depicted below. This dicationic species features multiple sulfonic acid groups (-SO3H) that provide strong Brønsted acidity, essential for activating the carbonyl group of the aromatic aldehyde and the active methylene compound. The ionic nature of the catalyst enhances its solubility in the polar reaction medium while allowing for potential phase separation or retention in the mother liquor for recycling. The cooperative interaction between the acidic protons and the reactants lowers the activation energy for the initial Knoevenagel condensation, facilitating the formation of the key olefinic intermediate which subsequently undergoes Michael addition with the triazole ring.
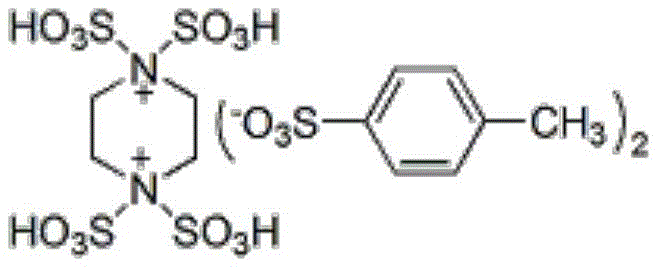
Furthermore, the catalyst plays a pivotal role in the cyclization step, promoting the intramolecular nucleophilic attack that closes the pyrimidine ring. The high selectivity observed in this process minimizes the formation of by-products, a common issue in acid-catalyzed condensations. This mechanistic efficiency is reflected in the exceptional purity of the final products, often exceeding 98% as confirmed by HPLC analysis. For instance, the synthesis of ethyl 5-amino-7-(4-chlorophenyl)-4,7-dihydro-[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylate yields a white solid with a melting point of 190-192°C and a purity of 98.8%, demonstrating the robustness of the catalytic system in controlling reaction pathways.
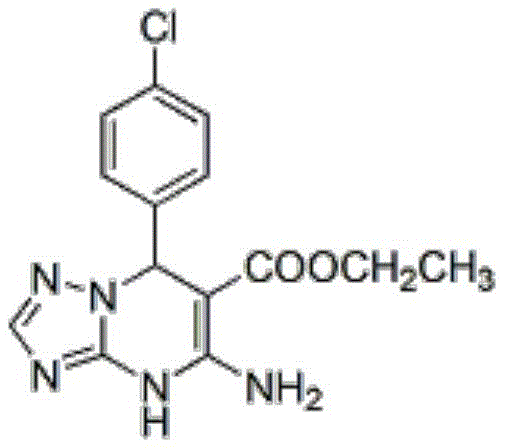
From an impurity control perspective, the mild reaction conditions prevent the degradation of sensitive functional groups, such as nitro or methoxy substituents on the aromatic ring. This is crucial for maintaining the structural integrity of complex intermediates intended for high-value API synthesis. The ability of the ionic liquid to stabilize transition states without promoting side reactions ensures a clean reaction profile, reducing the burden on purification units. This level of control is vital for meeting the rigorous specifications of international pharmacopoeias, thereby reducing the risk of batch rejection and ensuring a reliable supply of materials for clinical and commercial manufacturing.
How to Synthesize 4,7-Dihydro-[1,2,4]triazolo[1,5-a]pyrimidine Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and catalyst longevity. The process begins with the preparation of the ternary solvent system, followed by the sequential addition of reactants and the catalyst. Maintaining the correct molar ratios and reflux conditions is critical to driving the reaction to completion within the short timeframe of 30 to 60 minutes. The simplicity of the work-up procedure, involving mere cooling and filtration, makes this method highly accessible for both laboratory-scale optimization and industrial-scale production.
- Prepare the reaction mixture by combining aromatic aldehyde, active methylene compound, and 3-amino-1,2,4-triazole in an isopropanol-acetone aqueous solution within a three-neck flask equipped with a condenser.
- Add the acidic ionic liquid catalyst (6-9 mol%) to the mixture at room temperature, stir uniformly, and then heat to reflux temperature (approx. 31-57 minutes) until TLC indicates completion.
- Cool the reaction naturally to room temperature to precipitate the solid product, filter under vacuum, wash with anhydrous ethanol, and dry to obtain the pure derivative; the filtrate can be recycled.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-catalyzed process offers transformative benefits in terms of cost structure and operational reliability. The elimination of complex purification steps such as column chromatography significantly reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in raw materials and waste disposal. Moreover, the ability to recycle the catalytic system directly from the filtrate means that the effective cost of the catalyst per kilogram of product decreases dramatically with each cycle, enhancing the overall economic viability of the manufacturing process.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this synthesis eliminates the need for intermediate isolation and multiple solvent exchanges, which are major cost drivers in traditional chemical manufacturing. By reducing the number of unit operations, facilities can lower their energy consumption for heating and cooling, as well as minimize labor costs associated with handling and transferring materials. The high atom economy of the reaction ensures that a greater proportion of raw materials ends up in the final product, reducing waste generation and the associated environmental compliance costs. This holistic reduction in operational expenses allows for more competitive pricing strategies in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system, which can be reused for at least six to seven cycles without significant loss of activity, ensures a consistent and predictable production output. This stability is crucial for long-term supply agreements, as it mitigates the risk of production delays caused by catalyst replenishment or process failures. Additionally, the use of commercially available and inexpensive starting materials, such as substituted benzaldehydes and common beta-keto esters, reduces dependency on specialized or scarce reagents. This accessibility strengthens the supply chain against geopolitical disruptions and raw material shortages, ensuring continuous availability of critical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in standard three-neck flask setups that translate well to larger reactor vessels. The use of an aqueous-organic solvent system reduces the volatility and flammability risks associated with pure organic solvents, improving workplace safety and lowering insurance premiums. Furthermore, the significant reduction in chemical waste aligns with increasingly stringent environmental regulations, facilitating easier permitting and community acceptance of manufacturing sites. This green chemistry profile enhances the corporate sustainability image, a key factor for partnerships with major multinational pharmaceutical companies committed to carbon neutrality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing lines or for sourcing these intermediates from external partners. The answers are derived directly from the experimental data and technical specifications provided in the patent literature.
Q: Can the acidic ionic liquid catalyst be recycled in this synthesis process?
A: Yes, the patent data demonstrates that the catalytic system, comprising the acidic ionic liquid and the isopropanol-acetone aqueous solvent, can be directly recycled after filtration. Experimental results show the system maintains high catalytic activity and product purity over at least 6 to 7 consecutive cycles without significant degradation.
Q: What are the advantages of the isopropanol-acetone-water solvent system over traditional organic solvents?
A: This specific ternary solvent system acts both as a reaction medium and a recrystallization solvent. Unlike traditional methods requiring extraction and column chromatography, this system allows the product to precipitate directly upon cooling, significantly simplifying purification, reducing energy consumption, and minimizing environmental waste.
Q: What is the typical purity and yield range for these triazolopyrimidine derivatives?
A: According to the provided examples, the process consistently achieves high purity levels exceeding 97% (often reaching 98-99% by HPLC) and reaction yields ranging from 81% to 94%, depending on the specific substituents on the aromatic aldehyde and the active methylene compound used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,7-Dihydro-[1,2,4]triazolo[1,5-a]pyrimidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and sustainable synthesis routes for complex heterocyclic intermediates. Our team of expert process chemists has extensively evaluated the technology described in CN112142750A and possesses the capability to implement these advanced ionic liquid-catalyzed protocols at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in the lab are faithfully reproduced in our manufacturing facilities. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Whether you require custom synthesis of specific triazolopyrimidine derivatives or optimization of existing routes, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can support your R&D and commercial manufacturing goals with reliable, high-quality intermediates.
