Advanced Synthesis of Clethodim Intermediates via Zeolite Catalysis for Commercial Scale-up
Advanced Synthesis of Clethodim Intermediates via Zeolite Catalysis for Commercial Scale-up
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for broad-spectrum solutions like clethodim. Patent CN111217730A introduces a transformative preparation method that addresses critical bottlenecks in the existing supply chain for this vital crop protection agent. Traditionally, the synthesis of clethodim has been plagued by multi-step processes involving harsh conditions, low atom utilization, and complex purification requirements that inflate production costs and environmental liabilities. This new technical disclosure outlines a streamlined pathway that leverages controlled acidification and heterogeneous zeolite catalysis to achieve superior yields and purity profiles. By fundamentally re-engineering the conversion of key cyclic intermediates, this method offers a robust solution for reliable agrochemical intermediate supplier networks seeking to optimize their manufacturing footprint. The following analysis details the mechanistic advantages and commercial implications of adopting this next-generation synthetic route.
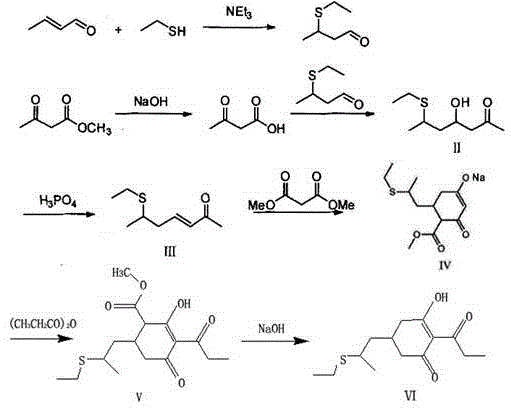
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for clethodim, such as those described in US6300281 and CN201710535450.4, suffer from inherent inefficiencies that compromise both economic viability and environmental compliance. The conventional pathway typically involves the formation of a cyclic intermediate which must then undergo a rearrangement reaction using homogeneous amine catalysts like 4-dimethylaminopyridine (DMAP). As illustrated in comparative data, this rearrangement step is notoriously problematic; it often results in yields hovering around 77% with significant formation of tar-like byproducts during subsequent hydrolysis. Furthermore, the introduction of amine catalysts necessitates rigorous post-treatment protocols, including multiple washing, extraction, and delamination steps to remove residual nitrogenous compounds. This not only extends the production cycle time but also generates wastewater with high ammonia nitrogen content, imposing a heavy burden on effluent treatment facilities and increasing the overall cost reduction in agrochemical intermediates manufacturing.
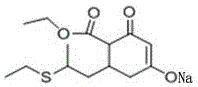
The Novel Approach
In stark contrast, the methodology disclosed in CN111217730A bypasses these traditional pitfalls through a clever manipulation of reaction conditions and catalyst selection. The core innovation lies in the direct conversion of the acidified intermediate (Compound B) to the acylated product (Compound D) using a solid acid catalyst, specifically ZSM-5 or H-ZSM-5 molecular sieves. This approach eliminates the discrete rearrangement step entirely, collapsing what was previously a multi-stage operation into a seamless, one-pot transformation. By avoiding the use of soluble amine catalysts and preventing the introduction of water during the acylation phase, the new process effectively suppresses hydrolysis side reactions that lead to tar formation. The result is a dramatic improvement in process simplicity, where operations such as washing and layering between intermediate stages are rendered unnecessary. This streamlining not only accelerates the reaction timeline but also ensures that the resulting intermediate possesses a purity profile that is far more conducive to high-yield final coupling reactions.
Mechanistic Insights into Zeolite-Catalyzed Acylation and Acidification
The technical superiority of this new route is rooted in the precise control of acidity and the unique properties of the zeolite catalyst. The process initiates with the acidification of Compound A to obtain Compound B, a step that serves a dual purpose: protonation of the enolate species and simultaneous purification. By adjusting the pH to a specific range, typically between 3 and 11, impurities generated during the prior cyclization are effectively separated, either through filtration of salts or phase separation. This pre-conditioning of the substrate is critical, as it ensures that the subsequent acylation occurs on a high-purity starting material, thereby minimizing the propagation of errors downstream. The use of inorganic acids like hydrochloric acid or organic acids like formic acid allows for fine-tuning of the reaction environment, ensuring that the intermediate remains stable prior to the next transformation.
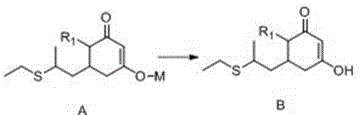
Following acidification, the reaction proceeds to the acylation stage where the ZSM-5 molecular sieve acts as a heterogeneous acid catalyst. Unlike homogeneous catalysts that dissolve in the reaction medium and require difficult removal, the zeolite provides active sites on its surface that facilitate the nucleophilic attack of the intermediate on propionyl chloride or propionic anhydride. The microporous structure of the ZSM-5 sieve likely imposes shape selectivity, favoring the formation of the desired ketone product while sterically hindering the formation of bulky polymeric byproducts. Furthermore, because the reaction is conducted in an anhydrous organic solvent environment, such as toluene or benzene, the risk of hydrolyzing the sensitive ester or ketone functionalities is virtually eliminated. This mechanistic precision results in yields exceeding 95% for the key intermediate, a figure that stands in sharp contrast to the sub-80% yields of legacy methods, demonstrating a clear path toward cost reduction in herbicide manufacturing.
How to Synthesize Clethodim Intermediate Efficiently
Implementing this advanced synthesis protocol requires careful attention to catalyst activation and temperature control to maximize the benefits of the zeolite system. The process begins with the preparation of the acidified intermediate solution, followed by the introduction of the activated molecular sieve catalyst at elevated temperatures. Operators must ensure that the catalyst is properly dispersed to maintain consistent reaction kinetics throughout the batch. The subsequent addition of the acylating agent should be controlled to manage exothermicity, ensuring safety and selectivity. Once the reaction reaches completion, indicated by the consumption of raw materials, the solid catalyst is simply filtered off, leaving a clean solution ready for the final hydrolysis and oxime formation steps. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis instructions below.
- Acidify Compound A using an inorganic or organic acid to obtain high-purity Compound B, removing impurities via filtration or layering.
- React Compound B with propionyl chloride or propionic anhydride in the presence of a ZSM-5 or H-ZSM-5 molecular sieve catalyst at 85-95°C.
- Filter to recover the catalyst and proceed directly to hydrolysis and decarboxylation to obtain the final clethodim precursor without intermediate rearrangement steps.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route represents a strategic opportunity to enhance margin stability and supply security. The elimination of complex purification steps and the reduction in reaction time directly translate to higher throughput capabilities within existing infrastructure. By removing the dependency on expensive and difficult-to-remove amine catalysts, manufacturers can significantly lower their raw material costs and reduce the inventory burden associated with specialized reagents. Moreover, the simplified workflow reduces the likelihood of batch failures caused by human error during tedious separation processes, thereby ensuring a more consistent and reliable supply of high-purity agrochemical intermediates for downstream formulation.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic simplification of the unit operations. By eliminating the need for multiple washing, extraction, and delamination steps, the process reduces labor hours, solvent consumption, and energy usage associated with heating and cooling cycles. Additionally, the high selectivity of the ZSM-5 catalyst minimizes the loss of valuable starting materials to tar and byproducts, effectively improving the overall mass balance of the production line. This efficiency gain allows for substantial cost savings without compromising on the quality specifications required for final herbicide registration.
- Enhanced Supply Chain Reliability: A shorter and more robust synthetic route inherently reduces the lead time for high-purity agrochemical intermediates. The removal of the rearrangement step, which was previously a bottleneck prone to variability, ensures that production schedules can be met with greater predictability. Furthermore, the use of commercially available and recyclable zeolite catalysts mitigates the risk of supply disruptions associated with specialized organic catalysts. This stability is crucial for maintaining continuous production flows, especially during peak agricultural seasons when demand for crop protection products surges globally.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with increasingly stringent global regulations regarding industrial effluent. The avoidance of amine catalysts means that wastewater streams do not require expensive ammonia nitrogen removal treatments, significantly lowering the operational expenditure on environmental compliance. The reduction in solvent usage and the ability to recycle the solid catalyst further contribute to a greener manufacturing profile. This sustainability advantage not only future-proofs the supply chain against regulatory changes but also enhances the brand value of the final agrochemical product in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel clethodim intermediate synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this technology outperforms legacy systems. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: How does the new acidification process improve intermediate purity compared to conventional methods?
A: The novel process utilizes a controlled acidification step to convert Compound A to Compound B, which effectively removes impurities formed during the initial cyclization. This pre-purification prevents the carryover of contaminants that typically form tar during subsequent hydrolysis, significantly enhancing the purity of the final clethodim intermediate.
Q: What are the environmental benefits of using ZSM-5 molecular sieves over amine catalysts?
A: Traditional methods often rely on amine catalysts like DMAP for rearrangement, which generate high ammonia nitrogen content in wastewater, requiring specialized treatment. The zeolite-catalyzed route eliminates the need for these amines and avoids water introduction during acylation, drastically reducing wastewater treatment burdens and three-waste discharge.
Q: Does this synthesis route require complex separation steps between intermediate stages?
A: No, one of the primary advantages of this method is the simplification of the workflow. The transition from Compound A to Compound D does not require complex operations such as separation, washing, layering, or rearrangement. The catalyst can be simply filtered off, allowing for a continuous and efficient production flow suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clethodim Supplier
As the agrochemical industry evolves towards more sustainable and efficient manufacturing practices, partnering with a technically proficient CDMO becomes essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN111217730A can be successfully translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of clethodim intermediate meets the exacting standards required for global herbicide registration and market entry.
We invite forward-thinking partners to collaborate with us on optimizing their supply chains through advanced catalytic technologies. By leveraging our expertise in process development, we can help you realize the full potential of this zeolite-catalyzed route. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and comprehensive route feasibility assessments tailored to your production volume requirements.
