Advanced Synthesis of Bactericidal Thiazolo-Pyrimidinone Intermediates for Commercial Scale-Up
Introduction to Novel Bactericidal Thiazole Architectures
The escalating demand for potent antibacterial agents has driven significant innovation in heterocyclic chemistry, particularly within the realm of fused nitrogen-sulfur systems. Patent CN110172067B discloses a groundbreaking technical solution for synthesizing a specific class of thiazole drug molecules exhibiting robust bactericidal activity against pathogens such as Escherichia coli and Bacillus subtilis. This intellectual property outlines a sophisticated six-step synthetic pathway that transforms simple acetophenone derivatives into complex thiazolo[3,2-a]pyrimidin-5-one scaffolds. The core structural motif, characterized by a fused thiazole and pyrimidinone ring system with variable substituents (R = methyl, hydroxyl, or nitro), represents a high-value target for pharmaceutical development. By leveraging a cascade of cyclization, functionalization, and oxidative coupling reactions, this methodology offers a streamlined approach to accessing biologically active intermediates that were previously difficult to manufacture with high efficiency.
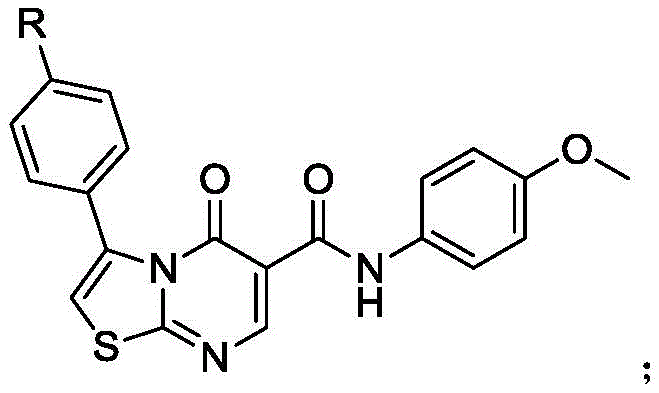
The strategic importance of this synthesis lies in its ability to introduce diverse functional groups at the para-position of the phenyl ring, allowing for fine-tuning of the biological activity profile. As demonstrated in the associated biological assays, the nitro-substituted variant exhibits superior inhibitory effects compared to its methyl and hydroxyl counterparts, highlighting the structure-activity relationship (SAR) potential inherent in this chemical series. For R&D directors and process chemists, understanding the nuances of this route is critical for developing next-generation antibiotics. The following analysis dissects the mechanistic intricacies and commercial viability of this patented process, providing a comprehensive roadmap for reliable pharmaceutical intermediate supplier engagements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing fused thiazole-pyrimidine systems often suffer from significant drawbacks that hinder their application in cost reduction in API manufacturing. Conventional routes frequently rely on harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents like phosphorus oxychloride for ring closure, which generate substantial toxic waste streams. Furthermore, older methodologies often lack regioselectivity, leading to complex mixtures of isomers that require extensive and yield-depleting purification steps. The reliance on stoichiometric amounts of heavy metal oxidants for side-chain functionalization not only inflates raw material costs but also introduces stringent environmental compliance burdens regarding heavy metal residue limits in final drug substances. These inefficiencies create bottlenecks in the supply chain, extending lead times and compromising the economic feasibility of scaling these molecules for commercial production.
The Novel Approach
In stark contrast, the methodology described in CN110172067B introduces a refined, catalytic strategy that addresses these historical pain points through milder conditions and higher atom economy. The novel approach utilizes a dual-catalyst system involving pyrrolidine and p-toluenesulfonic acid to facilitate the initial thiazole ring formation under reflux, eliminating the need for exotic reagents. A key innovation is the employment of a two-stage oxidation protocol for converting the hydroxymethyl intermediate to the carboxylic acid; first using silver nitrate and potassium persulfate, followed by a green oxidation utilizing molecular oxygen and N-hydroxyphthalimide (NHPI). This shift towards aerobic oxidation significantly reduces the chemical footprint and waste generation. Additionally, the final amidation step employs standard peptide coupling reagents (EDC/HOBt), ensuring high conversion rates and minimizing side reactions. This holistic process design enhances the overall yield and purity, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Thiazole-Pyrimidine Cyclization
The heart of this synthetic route lies in the construction of the bicyclic core, a process that demands precise control over reaction kinetics and thermodynamics. The initial formation of the 4-(4-methylbenzene)-thiazole-2-amine involves a Hantzsch-like thiazole synthesis where the enolizable ketone reacts with sulfur and cyanamide. The presence of pyrrolidine acts as a base to generate the enamine intermediate, while p-toluenesulfonic acid likely activates the cyanamide electrophile, facilitating the nucleophilic attack and subsequent cyclization. This step is critical as it establishes the sulfur-nitrogen heterocycle which serves as the nucleophile for the subsequent ring fusion. Following isolation, this amine undergoes condensation with ethyl 3-oxopropionate. Under the influence of p-toluenesulfonic acid and sodium methoxide, the amine attacks the keto-ester, followed by intramolecular cyclization and dehydration to form the stable thiazolo[3,2-a]pyrimidin-5-one skeleton. The use of a water separator during this reflux in toluene is a crucial engineering control to drive the equilibrium towards product formation by continuously removing the water byproduct.
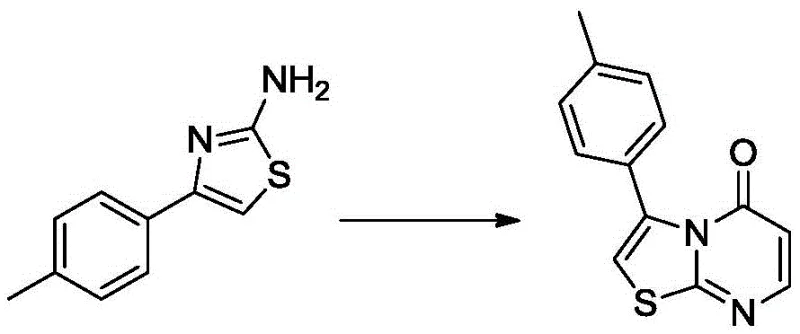
Impurity control is meticulously managed throughout the mechanism, particularly during the oxidation phases. The conversion of the hydroxymethyl group to the aldehyde using silver nitrate and potassium persulfate must be carefully monitored to prevent over-oxidation or degradation of the sensitive heterocyclic ring. The subsequent oxidation of the aldehyde to the carboxylic acid using NHPI and oxygen gas represents a radical-mediated mechanism that is highly selective for the aldehyde functionality. This selectivity is paramount for maintaining the integrity of the thiazole and pyrimidine rings, which could otherwise be susceptible to oxidative cleavage under harsher conditions. By optimizing the oxygen pressure to 0.1-0.2 MPa and maintaining room temperature, the process ensures that the desired carboxylic acid is generated with minimal byproduct formation, thereby simplifying downstream purification and enhancing the overall quality of the high-purity pharmaceutical intermediate.
How to Synthesize Thiazolo[3,2-a]pyrimidin-5-one Efficiently
Executing this synthesis requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the thiazole amine precursor, followed by the critical ring-closing step to form the pyrimidinone core. Subsequent functionalization involves hydroxymethylation with paraformaldehyde, followed by the sequential oxidation steps to install the carboxylic acid handle. The final stage involves coupling this acid with p-anisidine to generate the target amide. Each step utilizes common industrial solvents such as cyclohexane, toluene, methanol, and acetonitrile, facilitating easy solvent recovery and recycling. Detailed standard operating procedures regarding temperature ramps, addition rates, and workup protocols are essential for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the guide below.
- Cyclize acetophenone derivatives with sulfur and cyanamide using pyrrolidine/p-TsOH catalysis to form thiazole amines.
- Condense the thiazole amine with ethyl 3-oxopropionate under reflux to construct the fused pyrimidinone core.
- Perform hydroxymethylation with paraformaldehyde, followed by two-step oxidation (Ag/K-persulfate then NHPI/O2) to generate the carboxylic acid.
- Couple the resulting acid with p-anisidine using EDC/HOBt mediation to yield the final bactericidal amide target.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented process offers compelling advantages that directly impact the bottom line and operational resilience. The reliance on commodity chemicals such as acetophenones, elemental sulfur, and cyanamide ensures a robust and diversified supply base, mitigating the risk of raw material shortages that often plague specialty chemical manufacturing. Furthermore, the elimination of cryogenic conditions and the use of atmospheric or slightly pressurized oxygen for oxidation steps drastically simplify the reactor requirements, allowing for production in standard glass-lined or stainless steel vessels without the need for specialized Hastelloy equipment. This accessibility translates to broader manufacturing capacity availability and reduced capital expenditure for contract manufacturing organizations (CMOs).
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive stoichiometric oxidants with catalytic aerobic oxidation systems. The use of molecular oxygen as the terminal oxidant in the presence of NHPI eliminates the generation of heavy metal waste associated with traditional chromium or manganese-based oxidations, thereby reducing waste disposal costs. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the consumption of silica gel and solvents during purification. This streamlined workflow results in substantial cost savings per kilogram of produced intermediate, enhancing the overall margin profile for the final API.
- Enhanced Supply Chain Reliability: The synthetic route is designed with supply chain continuity in mind, utilizing reagents that are globally sourced and available in bulk quantities. The avoidance of proprietary or single-source catalysts ensures that production is not vulnerable to supplier monopolies. Moreover, the robustness of the reaction conditions, which tolerate minor fluctuations in temperature and stoichiometry without significant yield loss, contributes to consistent batch-to-batch quality. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand fluctuations and ensuring uninterrupted supply for downstream drug formulation.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated effectively from gram to multi-gram scales in the patent examples with consistent results. The use of green chemistry principles, such as aerobic oxidation and the minimization of hazardous byproducts, aligns with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and permitting timelines associated with new manufacturing lines. The ability to scale this process from 100 kgs to 100 MT annual commercial production without fundamental changes to the chemistry makes it an attractive asset for long-term strategic sourcing agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these thiazole derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for assessing the feasibility of integrating this chemistry into existing production portfolios.
Q: What are the critical starting materials for this thiazole-pyrimidine synthesis?
A: The process utilizes readily available substituted acetophenones (methyl, nitro, or hydroxy variants) as the primary feedstock, reacting them with elemental sulfur and cyanamide to establish the heterocyclic foundation.
Q: How does this method ensure high purity for pharmaceutical applications?
A: Purity is rigorously maintained through specific recrystallization protocols using methanol and silica gel column chromatography at critical intermediate stages, ensuring the final compound achieves >98% purity suitable for biological testing.
Q: Is this synthetic route suitable for large-scale manufacturing?
A: Yes, the methodology employs standard industrial solvents like toluene and acetonitrile with reflux conditions and avoids cryogenic temperatures or ultra-high pressure, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiazolo[3,2-a]pyrimidin-5-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced heterocyclic intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical techniques to verify the identity and quality of every batch. Our expertise in handling complex cyclization and oxidation chemistries positions us as a preferred partner for companies seeking to secure a stable supply of critical antibiotic intermediates.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific drug development needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget. Let us be your trusted ally in bringing high-quality bactericidal agents to the global market.
