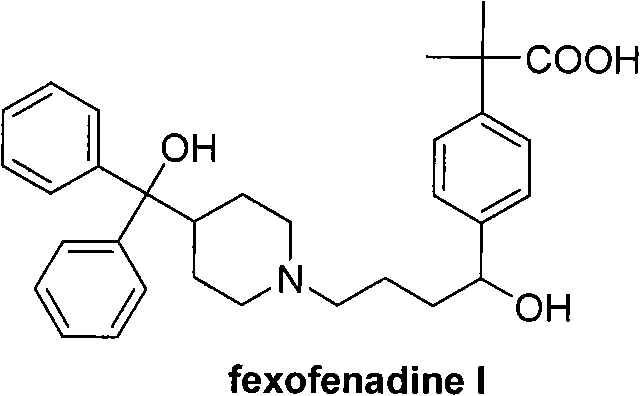
Advanced Synthetic Route for High-Purity Fexofenadine: Overcoming Isomer Challenges in Commercial Manufacturing
Introduction to Patent CN102070512A
The pharmaceutical industry constantly seeks robust manufacturing processes that guarantee high purity while minimizing production costs, particularly for third-generation antihistamines like fexofenadine. Patent CN102070512A discloses a groundbreaking synthesizing route and preparation method for high-purity fexofenadine and its key intermediates. This technology addresses the longstanding challenge of isomer separation inherent in traditional Friedel-Crafts acylation methods. By utilizing p-cyanomethylbenzoate as the primary raw material, the process ensures that the target molecule is constructed with the correct para-orientation from the very first step. This strategic choice eliminates the generation of meta-isomers, which are notoriously difficult to remove and significantly impact the quality of the final Active Pharmaceutical Ingredient (API). For R&D directors and procurement specialists, this represents a shift towards a more predictable and efficient supply chain for high-purity fexofenadine.
The significance of this patent lies in its ability to streamline the entire production workflow. Conventional methods often suffer from low selectivity, requiring complex purification steps that drive up costs and extend lead times. In contrast, the novel route described in CN102070512A employs a series of well-controlled reactions including methylation, condensation with gamma-butyrolactone, and subsequent hydrolysis and reduction. The result is a process that is not only chemically elegant but also commercially viable, offering a reliable solution for manufacturers aiming to secure a stable supply of this critical allergy medication intermediate without the burden of excessive impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fexofenadine has been plagued by regioselectivity issues. As detailed in the background technology of the patent, earlier methods such as US Patent 4254129 rely on Friedel-Crafts acylation between alpha,alpha-dimethyl phenyl acetic acid esters and chloroacetyl chloride derivatives. This approach fundamentally lacks precision, typically yielding a mixture of para and meta isomers in a ratio of approximately 85:15. Separating these structural analogues is an arduous task that often requires multiple recrystallizations or chromatographic separations, leading to substantial material loss and increased operational expenses. Furthermore, some alternative routes reported in literature involve the use of hazardous reagents like mercury oxide for oxidation steps, posing severe environmental and safety risks that are increasingly unacceptable in modern green chemistry standards.
Another significant drawback of traditional pathways is the complexity of the reaction conditions. Methods utilizing Grignard reagents or requiring strong acidic conditions for dehydration can lead to unpredictable side reactions and the formation of difficult-to-remove by-products. For a reliable API intermediate supplier, managing these variability factors is a major logistical hurdle. The accumulation of impurities not only affects the yield but also complicates the regulatory approval process for the final drug product. Consequently, there is a pressing industrial need for a synthesis strategy that bypasses these selectivity pitfalls and offers a cleaner, more direct path to the target molecule.
The Novel Approach
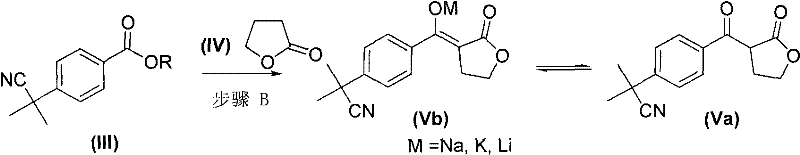
The innovative route presented in CN102070512A fundamentally reengineers the synthesis by starting with a pre-functionalized aromatic ring. By selecting p-cyanomethylbenzoate (Compound II) as the starting material, the process inherently guarantees the para-substitution pattern required for fexofenadine. This eliminates the isomer problem at the source, ensuring that every subsequent step builds upon the correct molecular scaffold. The synthesis proceeds through a key intermediate (Compound Va or its salt Vb), formed via condensation with gamma-butyrolactone. This step effectively constructs the four-carbon ketone chain necessary for the final structure without introducing regio-ambiguity.
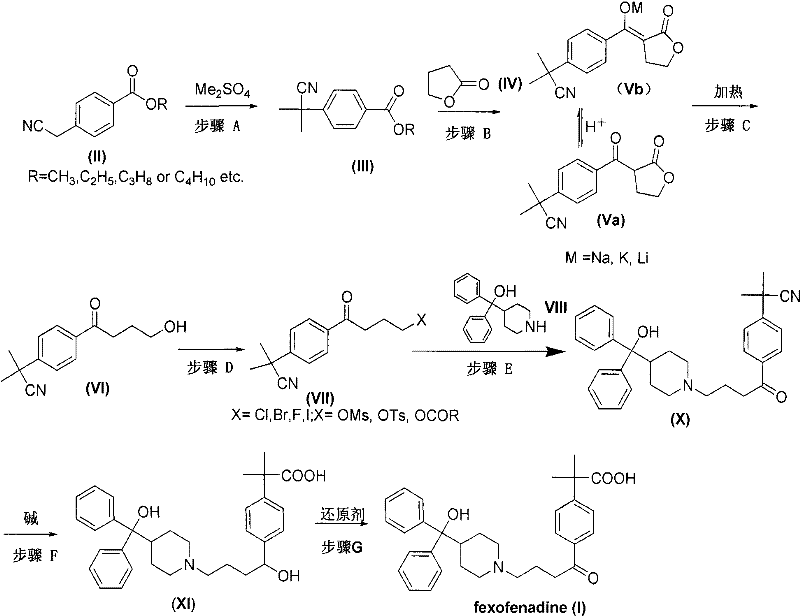
Moreover, the novel approach emphasizes operational simplicity and scalability. The patent describes conditions that allow for 'one-pot' reactions, where multiple transformation steps can be performed sequentially without isolating unstable intermediates. For instance, the conversion from Compound X to Compound XI and finally to Fexofenadine (I) can be achieved in a continuous sequence. This reduction in unit operations translates directly to cost reduction in pharmaceutical manufacturing, as it lowers solvent consumption, energy usage, and labor requirements. The ability to produce high-purity products with simplified post-processing makes this route highly attractive for large-scale commercial production, offering a distinct competitive advantage over legacy technologies.
Mechanistic Insights into Lactone Condensation and Decarboxylation
A critical aspect of this synthesis is the formation of the ketone side chain via the condensation of the ester (III) with gamma-butyrolactone (IV). In Step B, a strong base such as sodium hydride is used to deprotonate the alpha-position of the ester, generating a nucleophilic enolate. This enolate attacks the carbonyl carbon of the lactone ring, leading to ring opening and the formation of the beta-keto ester salt (Vb). This mechanism is highly efficient because the lactone serves as a protected form of the four-carbon acid chain, preventing self-condensation side reactions that might occur with open-chain equivalents. The stability of the intermediate salt (Vb) allows for easy handling and purification, often simply by extraction or crystallization, which is crucial for maintaining high throughput in a manufacturing setting.
Following the condensation, Step C involves a hydrolysis and decarboxylation sequence that converts the beta-keto ester into the desired gamma-keto alcohol (VI). Under acidic or alkaline conditions with heating, the ester group is hydrolyzed to the carboxylic acid, which then undergoes thermal decarboxylation due to the activation provided by the adjacent ketone group. This mechanistic pathway is clean and high-yielding, avoiding the need for harsh reducing agents or toxic catalysts often seen in older methods. The resulting hydroxy-ketone (VI) is then activated in Step D (via halogenation or esterification) to become an electrophile (VII), ready for nucleophilic attack by the piperidine derivative (VIII). This logical progression of functional group interconversions demonstrates a deep understanding of organic reactivity, ensuring that each step maximizes yield while minimizing impurity generation.
How to Synthesize Fexofenadine Efficiently
The synthesis of fexofenadine via this patented route involves a sequence of precise chemical transformations designed to maximize yield and purity. The process begins with the methylation of the starting benzoate, followed by the critical lactone condensation to build the carbon skeleton. Subsequent steps involve activating the alcohol for coupling with the piperidine moiety, followed by hydrolysis of the nitrile and ester groups to reveal the final carboxylic acid and alcohol functionalities. The detailed standardized synthesis steps for implementing this route in a GMP environment are outlined below.
- Methylation of p-cyanomethylbenzoate (II) using dimethyl sulfate under alkaline conditions to form compound (III).
- Condensation of compound (III) with gamma-butyrolactone (IV) using a strong base like sodium hydride to form the key intermediate salt (Vb).
- Hydrolysis and decarboxylation of intermediate (V) to generate the keto-alcohol (VI), followed by halogenation and coupling with piperidinol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By eliminating the formation of meta-isomers, the process removes the need for expensive and time-consuming separation technologies such as preparative HPLC or multiple fractional crystallizations. This directly correlates to a significant reduction in production costs and a shorter overall cycle time from raw material intake to finished goods. Furthermore, the use of readily available starting materials like p-cyanomethylbenzoate ensures a stable supply base, reducing the risk of raw material shortages that can plague more exotic synthetic pathways.
- Cost Reduction in Manufacturing: The elimination of isomer separation steps results in substantial cost savings by reducing solvent usage, energy consumption for distillation, and labor hours associated with complex purification. Additionally, the avoidance of toxic heavy metal catalysts like mercury oxide lowers waste disposal costs and mitigates environmental compliance risks. The high yields reported in the patent embodiments further contribute to better atom economy, ensuring that a larger proportion of raw materials are converted into saleable product rather than waste.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, makes the process less susceptible to minor fluctuations in operating parameters. This reliability ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with pharmaceutical clients. The ability to perform 'one-pot' reactions also reduces the number of intermediate storage requirements, streamlining the logistics of the manufacturing facility and allowing for faster turnaround times on orders.
- Scalability and Environmental Compliance: The synthetic route is designed with industrial scale-up in mind, utilizing common reagents and standard reactor configurations. The absence of hazardous reagents simplifies the safety profile of the plant, reducing the need for specialized containment equipment. From an environmental perspective, the cleaner reaction profile means less chemical waste is generated per kilogram of product, aligning with global sustainability goals and facilitating easier permitting for capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fexofenadine synthesis route. These answers are derived directly from the technical specifications and comparative data provided in patent CN102070512A, offering clarity on how this method outperforms traditional alternatives in terms of purity, safety, and operational efficiency.
Q: How does this new route solve the isomer separation problem in Fexofenadine synthesis?
A: Traditional Friedel-Crafts acylation methods produce a mixture of para and meta isomers (approx. 85:15), which are difficult and costly to separate. This patent utilizes p-cyanomethylbenzoate as a starting material, which already possesses the correct para-substitution pattern, thereby completely eliminating the formation of meta-isomers and ensuring high product purity from the outset.
Q: What are the environmental advantages of this synthesis compared to older methods?
A: Older routes, such as those reported by Kawai, often require toxic heavy metal oxidants like mercury oxide (red precipitate) to convert alkynes to ketones. The present invention avoids these hazardous reagents entirely, utilizing safer alkylation and condensation reactions that result in significantly reduced environmental pollution and simpler waste treatment protocols.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial applicability. It features simple reaction conditions, uses readily available raw materials, and allows for 'one-pot' sequences (such as converting compound X to XI to I without isolation). These factors drastically simplify post-processing and enhance the feasibility of commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fexofenadine Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in patent literature can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fexofenadine intermediate meets the highest global regulatory standards, providing you with peace of mind regarding product quality and consistency.
We invite you to collaborate with us to optimize your supply chain for this critical antihistamine intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the pharmaceutical market through superior chemical manufacturing.
