Advanced Synthetic Route for Fexofenadine Intermediates Enhancing Commercial Scalability
Advanced Synthetic Route for Fexofenadine Intermediates Enhancing Commercial Scalability
The pharmaceutical industry continuously seeks robust and scalable synthetic pathways for high-volume active pharmaceutical ingredients (APIs), particularly for non-sedating antihistamines like fexofenadine, commercially known as Allegra®. Patent CN1516689A presents a significant technological advancement in this domain by disclosing a novel method for preparing specific piperidine derivatives, including the critical intermediate structures required for fexofenadine synthesis. This intellectual property outlines a sophisticated chemical strategy that moves beyond simple alkylation, focusing instead on a controlled acylation and purification sequence that ensures high regioselectivity. The core innovation lies in the ability to effectively separate para- and meta-isomers through a unique salt formation technique, which is a pivotal improvement for maintaining stringent quality standards in API manufacturing. By leveraging this patented approach, manufacturers can achieve superior impurity profiles while utilizing standard industrial equipment, thereby bridging the gap between laboratory discovery and commercial viability.
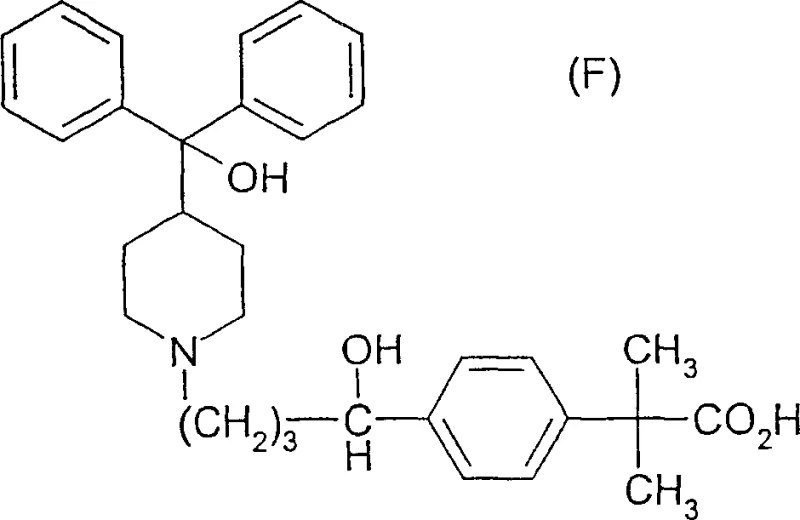
This technical disclosure is particularly relevant for stakeholders focused on the reliable supply of high-purity pharmaceutical intermediates, as it addresses common bottlenecks associated with isomeric mixtures. The process described not only yields the final active compound but also defines a series of novel synthetic intermediates, such as those of formulas VIII and IX, which serve as essential building blocks in the value chain. For R&D directors and process chemists, understanding the nuances of this pathway offers a blueprint for optimizing yield and reducing waste in existing production lines. Furthermore, the methodology emphasizes the use of accessible reagents and solvents, which aligns with modern green chemistry principles and cost-efficiency goals. As we delve deeper into the mechanistic details and commercial implications, it becomes evident that this patent represents a strategic asset for any organization aiming to secure a competitive edge in the antihistamine market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating aryl-alkyl ketone intermediates often suffer from poor regioselectivity during the initial carbon-carbon bond-forming steps, typically resulting in complex mixtures of ortho, meta, and para isomers. In the context of fexofenadine precursors, conventional Friedel-Crafts acylations frequently produce a statistical distribution of isomers that are notoriously difficult to separate using standard physical methods like distillation or simple recrystallization. This lack of selectivity necessitates the use of resource-intensive purification techniques, such as preparative chromatography, which are economically unfeasible for multi-ton commercial production. Moreover, the presence of meta-isomers can lead to downstream impurities that are structurally similar to the target molecule, posing significant challenges for regulatory compliance and final drug safety. The reliance on harsh reaction conditions without effective differentiation mechanisms often results in lower overall yields and increased environmental burden due to excessive solvent consumption and waste generation.
The Novel Approach
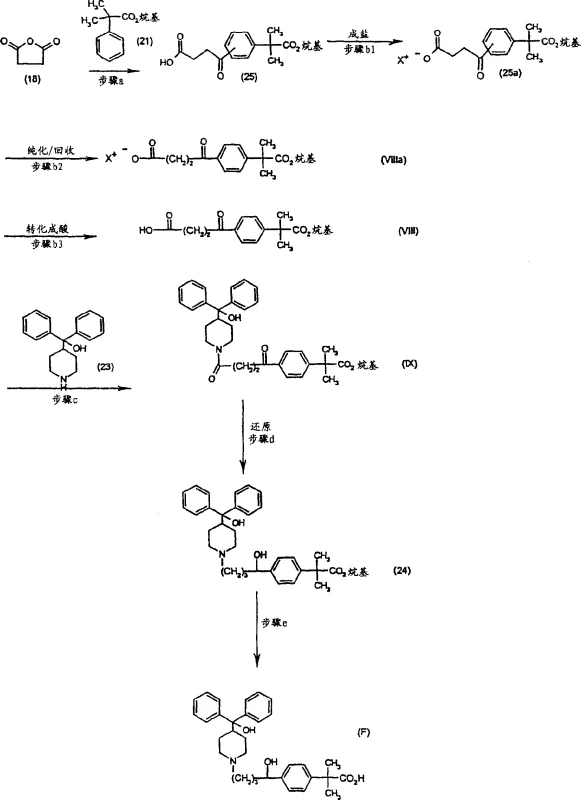
In stark contrast, the methodology disclosed in patent CN1516689A introduces a transformative solution by integrating a selective salt formation step immediately following the acylation reaction. Instead of attempting to separate the free acid isomers directly, the process converts the crude mixture of regioisomers into phenethylamine salts, which exhibit distinct solubility properties in alcoholic solvents like isopropanol. This clever manipulation of physicochemical characteristics allows for the preferential crystallization of the desired para-isomer salt, effectively purifying the intermediate at an early stage of the synthesis. By isolating the pure para-isomer salt (Formula VIIIa') before proceeding to subsequent coupling reactions, the process ensures that downstream steps are fed with high-quality starting material, thereby minimizing the propagation of impurities. This approach not only simplifies the purification workflow but also significantly enhances the overall efficiency of the synthesis, making it a superior choice for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Friedel-Crafts Acylation and Regioselective Crystallization
The chemical foundation of this novel process rests on a carefully optimized Friedel-Crafts acylation followed by a thermodynamic control step via crystallization. In the initial acylation phase, alpha,alpha-dimethylphenylacetic acid ester reacts with succinic anhydride in the presence of a Lewis acid catalyst, typically aluminum chloride (AlCl3), within an anhydrous aprotic solvent system such as carbon disulfide or dichloromethane. While this reaction inherently generates a mixture of regioisomers (approximately 60% para and 40% meta), the true brilliance of the invention lies in the subsequent workup. The crude acid mixture is treated with phenethylamine to form ammonium carboxylate salts, where the para-isomer salt demonstrates significantly lower solubility in hot isopropanol compared to its meta-counterpart. Upon cooling, the para-isomer salt precipitates out of the solution with high purity, a phenomenon driven by the specific crystal lattice energy and solvation dynamics of the phenethylamine complex. This fractional crystallization acts as a powerful filter, removing the unwanted meta-isomer which remains in the mother liquor, thus achieving a level of purity that would otherwise require multiple chromatographic passes.
Following the isolation of the pure acid intermediate, the synthesis proceeds through an amide coupling reaction with a piperidine derivative, specifically 4-(hydroxydiphenylmethyl)piperidine. This step utilizes standard peptide coupling activation strategies, employing reagents such as ethyl chloroformate or dicyclohexylcarbodiimide (DCC) to activate the carboxylic acid moiety. The resulting keto-amide intermediate is then subjected to a reduction step using borane complexes, such as borane-dimethylsulfide, which selectively reduces both the ketone and the amide functionalities to yield the final hydroxy-alkyl chain. The mechanistic precision here ensures that the stereochemistry and functional group integrity are maintained throughout the transformation. By controlling the reaction temperature and stoichiometry, the process minimizes side reactions such as over-reduction or elimination, ensuring that the final product meets the rigorous specifications required for pharmaceutical applications. This detailed understanding of the reaction mechanism allows process engineers to fine-tune parameters for maximum yield and minimal byproduct formation.
How to Synthesize Fexofenadine Intermediates Efficiently
The synthesis of these critical piperidine derivatives requires a disciplined approach to reaction conditions and purification protocols to ensure reproducibility and high quality. The process begins with the strict control of moisture during the Friedel-Crafts acylation, as the presence of water can deactivate the Lewis acid catalyst and lead to hydrolysis of the anhydride. Following the acylation, the formation of the phenethylamine salt must be conducted under controlled cooling rates to promote the growth of large, pure crystals of the para-isomer, which facilitates easier filtration and washing. The subsequent coupling and reduction steps demand anhydrous conditions and careful monitoring of exotherms to prevent thermal runaway and ensure operator safety. Detailed standardized operating procedures for each of these stages are essential for transferring this technology from the laboratory to the pilot plant and eventually to full-scale commercial production.
- Perform Friedel-Crafts acylation of alpha,alpha-dimethylphenylacetic acid ester with succinic anhydride using AlCl3 to generate regioisomer mixtures.
- Separate the desired para-isomer by forming phenethylamine salts and utilizing fractional crystallization in isopropanol.
- Couple the purified acid intermediate with a piperidine derivative using activating agents like DCC or ethyl chloroformate, followed by borane reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthetic route offers tangible benefits that extend far beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification process, which eliminates the need for expensive and time-consuming chromatographic separations that often bottleneck production capacity. By relying on crystallization—a unit operation that is easily scalable and cost-effective—manufacturers can significantly reduce the operational expenditure associated with solvent recovery and waste disposal. Furthermore, the starting materials utilized in this process, such as succinic anhydride and simple phenylacetic acid esters, are commodity chemicals with stable and robust global supply chains, mitigating the risk of raw material shortages. This reliability ensures consistent production schedules and protects against market volatility, making the supply of high-purity pharmaceutical intermediates more predictable and secure for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of complex purification steps like preparative HPLC translates directly into substantial cost savings, as the process relies on inexpensive crystallization techniques and readily available reagents. The ability to recover the desired isomer with high efficiency reduces the overall material input required per kilogram of final product, optimizing the cost of goods sold. Additionally, the use of standard solvents like isopropanol and ethyl acetate, which are easily recycled, further lowers the environmental and financial burden of the manufacturing process. This economic efficiency makes the technology highly attractive for generic drug producers looking to maximize margins while maintaining competitive pricing strategies in the global marketplace.
- Enhanced Supply Chain Reliability: By utilizing widely available commodity chemicals as starting materials, the process minimizes dependency on specialized or custom-synthesized reagents that may have long lead times. The robustness of the synthetic route means that production can be scaled up rapidly to meet surges in demand without compromising on quality or delivery timelines. This flexibility is crucial for maintaining continuity of supply in the pharmaceutical sector, where interruptions can have significant clinical and commercial consequences. The method's compatibility with existing infrastructure in most fine chemical plants further accelerates the timeline from technology transfer to commercial availability, ensuring a steady flow of critical intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions and workup procedures that are safe and manageable on a multi-ton scale. The avoidance of heavy metal catalysts or toxic reagents simplifies waste treatment and helps facilities meet increasingly stringent environmental regulations. The high selectivity of the crystallization step reduces the volume of organic waste generated, contributing to a greener manufacturing footprint. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing organization, appealing to environmentally conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic methodology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production needs. The answers highlight the specific advantages of the salt formation technique and the robustness of the reduction steps, offering clarity on how this technology solves traditional manufacturing pain points.
Q: How does this patent improve impurity control compared to traditional methods?
A: The patent introduces a specific salt formation step using phenethylamine, which allows for the efficient fractional crystallization of the desired para-isomer from the meta-isomer mixture, significantly enhancing purity without chromatography.
Q: What are the key starting materials for this synthesis?
A: The process utilizes readily available commodity chemicals such as succinic anhydride, alpha,alpha-dimethylphenylacetic acid methyl ester, and 4-(hydroxydiphenylmethyl)piperidine, ensuring robust supply chain continuity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method relies on standard unit operations like crystallization and filtration rather than complex preparative HPLC, making it highly scalable for industrial manufacturing of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fexofenadine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensively analyzed technologies like patent CN1516689A to ensure we can deliver high-purity intermediates that meet the most demanding specifications. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of fexofenadine intermediate we supply is ready for immediate use in API synthesis. We are committed to being a strategic partner who understands the complexities of bringing life-saving medications to market.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized processes can reduce your overall production costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to streamline your supply chain and accelerate your time to market with reliable, high-quality chemical solutions.
