Advanced Chiral Resolution Strategy for Industrial Rotigotine Production and Commercial Scale-Up
Advanced Chiral Resolution Strategy for Industrial Rotigotine Production and Commercial Scale-Up
The pharmaceutical landscape for Parkinson's disease treatment has been significantly transformed by dopamine agonists, with Rotigotine (CAS 92206-54-7) standing out as a critical transdermal therapeutic agent. As detailed in patent CN101717392A, a groundbreaking methodology has been established to synthesize Rotigotine and its derivatives with superior efficiency and environmental safety profiles. This technical disclosure addresses the longstanding challenges associated with the stereochemical control and purification of this complex tetrahydronaphthalene derivative. By leveraging a novel chiral phosphoric acid resolution strategy, the process circumvents the toxicity and low yield issues plaguing earlier synthetic routes. For R&D directors and procurement specialists seeking a reliable rotigotine intermediate supplier, understanding the mechanistic nuances of this patent is essential for securing a stable supply chain of high-purity active pharmaceutical ingredients.
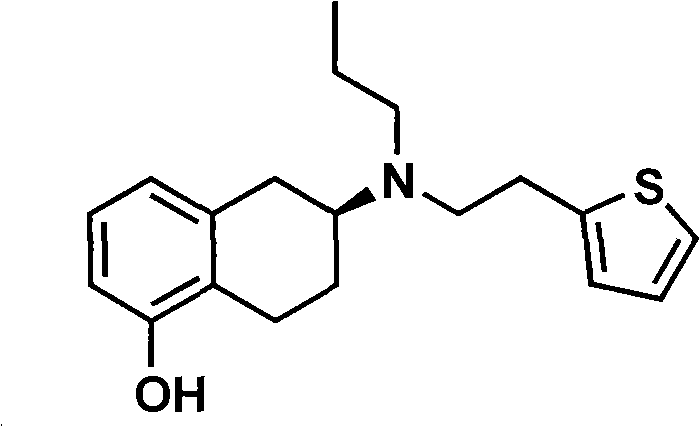
The commercial viability of any API intermediate hinges on the robustness of its synthetic pathway, particularly regarding impurity profiles and optical purity. The traditional synthesis of Rotigotine often involves cumbersome protection-deprotection sequences and the use of hazardous alkylating agents that complicate downstream processing. In contrast, the methodology outlined in this patent streamlines the construction of the chiral center early in the synthesis. This strategic shift not only simplifies the operational workflow but also ensures that the final product meets the stringent enantiomeric excess requirements necessary for neurological applications. The ability to produce high-purity rotigotine consistently is a decisive factor for pharmaceutical manufacturers aiming to minimize regulatory hurdles and accelerate time-to-market for generic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for Rotigotine have historically relied on the use of benzylamine as a protecting group or intermediate, which introduces significant toxicity concerns and necessitates rigorous removal steps to meet safety standards. Furthermore, conventional chiral resolution techniques often employ tartaric acid derivatives, which frequently result in suboptimal optical purity and require multiple recrystallization cycles to achieve acceptable enantiomeric ratios. These inefficiencies lead to substantial material loss and increased solvent waste, driving up the overall cost of goods sold. Additionally, the use of reagents such as iodopropane and 2-butanone in large quantities poses environmental hazards and requires specialized handling protocols that can bottleneck production capacity. The cumulative effect of these drawbacks is a synthetic process that is economically burdensome and difficult to scale for commercial demand.
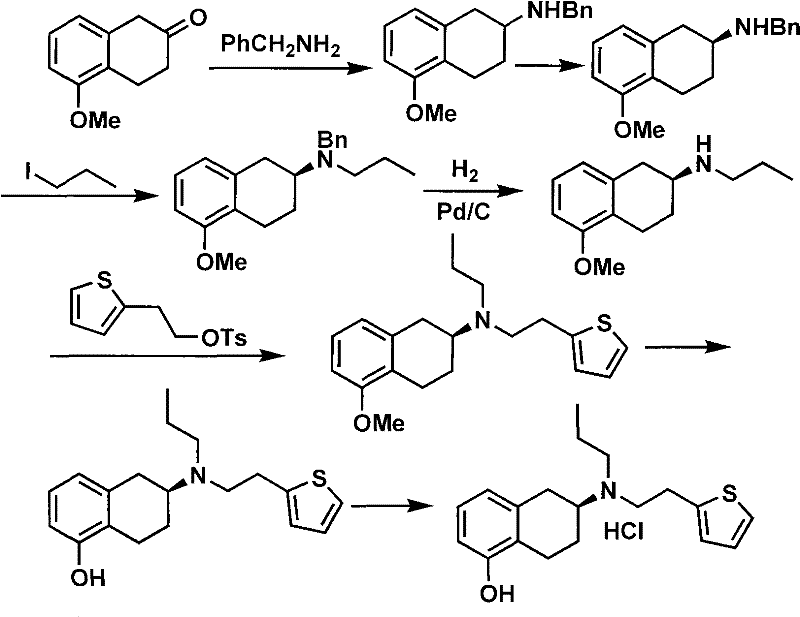
The Novel Approach
The innovative process described in patent CN101717392A fundamentally reengineers the synthesis by initiating with 5-methoxy-2-tetralone and employing a direct reductive amination strategy. This approach eliminates the need for toxic benzylamine intermediates, thereby simplifying the purification train and reducing the environmental footprint of the manufacturing process. A pivotal advancement in this route is the introduction of a novel chiral organic phosphoric acid, derived from readily available chiral amines such as phenylglycine or proline, to resolve the racemic amine intermediate. This resolution step is highly efficient, yielding the desired (S)-enantiomer with exceptional optical purity in a single operation. The subsequent condensation with a thiophene ethyl sulfonate ester and final demethylation are conducted under mild conditions, ensuring high yields and minimizing the formation of deleterious side products.
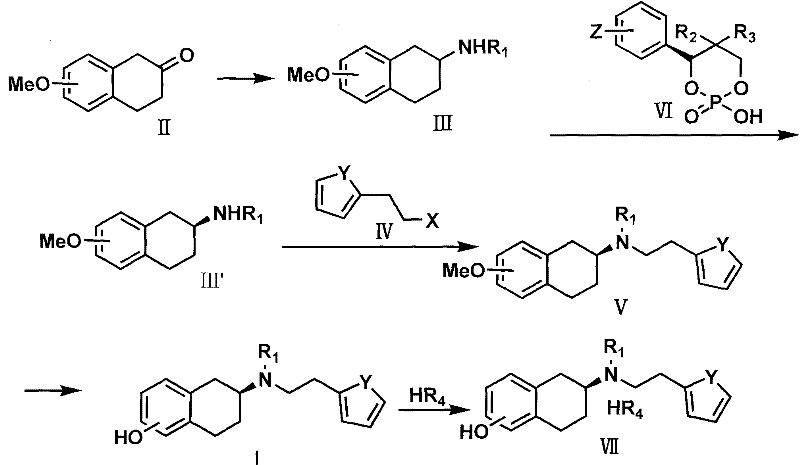
Mechanistic Insights into Chiral Phosphoric Acid Resolution
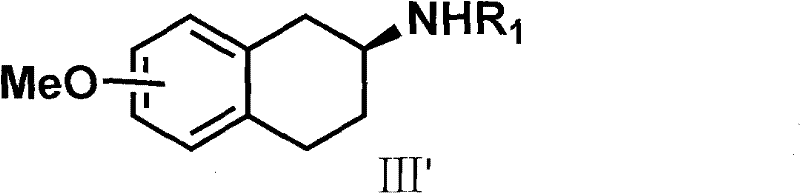
The cornerstone of this synthetic breakthrough lies in the sophisticated chiral resolution mechanism utilizing the phosphoric acid derivative designated as Formula VI. Unlike traditional salt formation methods that rely on weak acid-base interactions, the chiral phosphoric acid forms a highly stable diastereomeric complex with the racemic amine intermediate (Formula III). This interaction is governed by precise steric and electronic complementarity between the chiral phosphate anion and the ammonium cation of the amine. The differential solubility of these diastereomeric salts allows for the selective crystallization of the desired enantiomer, effectively filtering out the unwanted (R)-isomer. This mechanism ensures that the optical purity is established early in the synthesis, preventing the propagation of chiral impurities into the final API. The use of amino acid-derived phosphoric acids also offers a sustainable advantage, as these resolving agents can be synthesized from renewable chiral pools.
Furthermore, the stability of the chiral intermediate (Formula III') generated through this resolution is critical for the subsequent alkylation step. The patent demonstrates that maintaining the integrity of the chiral center during the nucleophilic substitution with the thiophene derivative is paramount. The reaction conditions, typically involving mild bases like sodium carbonate in polar aprotic solvents, are optimized to prevent racemization while facilitating efficient bond formation. The final demethylation step, often achieved using hydrobromic acid or boron tribromide, cleaves the methyl ether without compromising the stereochemical configuration of the adjacent chiral carbon. This robustness against racemization throughout the synthetic sequence is a key indicator of the process's reliability for producing clinical-grade material with consistent quality attributes.
How to Synthesize Rotigotine Efficiently
The synthesis of Rotigotine via this patented route involves a logical sequence of transformations designed to maximize yield and purity while minimizing operational complexity. The process begins with the reductive amination of 5-methoxy-2-tetralone, followed by the critical chiral resolution step that defines the stereochemistry of the final product. Subsequent functionalization with the thiophene side chain and removal of the methyl protecting group complete the synthesis of the free base, which is then converted to the pharmacologically active hydrochloride salt. Each step has been optimized in the patent embodiments to demonstrate feasibility on a laboratory scale, providing a clear blueprint for process chemists. For detailed operational parameters, including specific stoichiometry, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
- Perform reductive amination on 5-methoxy-2-tetralone using propylamine and a reducing agent like sodium borohydride to form the racemic amine intermediate.
- Execute chiral resolution of the racemic amine using a specifically prepared chiral organic phosphoric acid to isolate the optically active (S)-enantiomer with high purity.
- Conduct a condensation reaction between the chiral amine and a 2-thiophene ethyl sulfonate ester under alkaline conditions to attach the side chain.
- Finalize the synthesis by demethylating the methoxy group using hydrobromic acid or boron tribromide, followed by salt formation to yield Rotigotine Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of toxic and regulated reagents such as benzylamine and iodopropane simplifies the sourcing of raw materials and reduces the regulatory burden associated with hazardous chemical handling. This streamlining of the supply chain mitigates the risk of production delays caused by strict environmental compliance checks or shortages of controlled substances. Moreover, the high efficiency of the chiral resolution step means that less starting material is wasted in the pursuit of high optical purity, leading to a more atom-economical process. These factors collectively contribute to a more predictable and cost-effective manufacturing model that aligns with the strategic goals of modern pharmaceutical supply chains.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the synthetic sequence and the improved efficiency of the chiral resolution. By avoiding the use of expensive and inefficient resolving agents like tartaric acid derivatives, the process significantly lowers the cost of raw materials per kilogram of API produced. Additionally, the high yields reported in the patent embodiments reduce the volume of solvent required for purification and recrystallization, resulting in substantial savings in utility and waste disposal costs. The overall reduction in processing steps also translates to lower labor and equipment occupancy costs, enhancing the overall profitability of the manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 5-methoxy-2-tetralone and common amino acids, ensures a robust and resilient supply chain. Unlike processes that depend on custom-synthesized or exotic reagents, this route leverages commodity chemicals that are readily accessible from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks and price volatility. Furthermore, the mild reaction conditions and standard equipment requirements facilitate technology transfer across different manufacturing sites, ensuring continuity of supply even in the event of regional disruptions or capacity constraints at a specific facility.
- Scalability and Environmental Compliance: The design of this synthetic route inherently supports scalability, with reaction conditions that are easily adaptable from laboratory to pilot and commercial scales. The avoidance of extreme temperatures and pressures minimizes the engineering challenges associated with scale-up, allowing for faster ramp-up times to meet market demand. From an environmental standpoint, the reduction in hazardous waste generation and the use of less toxic reagents align with green chemistry principles, facilitating easier permitting and compliance with increasingly stringent environmental regulations. This sustainability profile not only reduces liability but also enhances the corporate social responsibility standing of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Rotigotine. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the process capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their own production needs or for assessing the quality of suppliers utilizing this method. The insights provided here bridge the gap between theoretical chemistry and practical industrial application.
Q: What distinguishes this rotigotine synthesis method from prior art?
A: Unlike conventional methods that rely on toxic benzylamine and inefficient tartaric acid resolution, this patent utilizes a novel chiral phosphoric acid resolution system. This approach significantly enhances optical purity (up to 99% ee) and eliminates hazardous reagents, making it safer and more suitable for industrial scaling.
Q: How does the chiral resolution step impact overall production costs?
A: The use of recyclable or efficiently synthesized chiral phosphoric acids derived from common amino acids reduces the reliance on expensive, single-use resolving agents. Furthermore, the high enantiomeric excess achieved minimizes the need for repeated recrystallization, thereby lowering solvent consumption and processing time.
Q: Is this synthetic route scalable for commercial API manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrialized production. The reaction conditions utilize standard reagents like sodium borohydride and common solvents such as toluene and ethanol, avoiding extreme pressures or temperatures that typically hinder large-scale operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rotigotine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving Parkinson's disease medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate chiral resolution and demethylation steps described in this patent are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Rotigotine intermediate meets the highest international standards for optical purity and chemical identity. Our commitment to excellence ensures that our partners receive materials that facilitate smooth regulatory filings and consistent clinical outcomes.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced synthetic technology for their Rotigotine projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall cost of goods.
