Revolutionizing N-Boc-Piperazine Production: A Novel Ethylenediamine Route for Commercial Scale-Up
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for higher purity, lower costs, and more sustainable processes. A pivotal advancement in this domain is detailed in Chinese Patent CN113185478B, which discloses a novel preparation method for N-tert-butoxycarbonyl piperazine (N-Boc-piperazine). This compound is a critical building block for a vast array of bioactive molecules, including antitussives, antihistamines, and antipsychotics. Traditionally, the synthesis of this protected piperazine has been plagued by inefficiencies, particularly regarding selectivity and raw material utilization. The patented technology introduces a constructive synthetic strategy that bypasses these historical bottlenecks by utilizing ethylenediamine and chloroacetaldehyde as starting materials. This approach not only fundamentally alters the reaction pathway to avoid the formation of stubborn impurities but also aligns perfectly with the principles of green chemistry by improving atom economy. For R&D directors and procurement strategists, understanding this shift from direct functionalization to constructive ring synthesis is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in complex drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-Boc-piperazine has relied heavily on the direct reaction of piperazine with di-tert-butyl dicarbonate (Boc2O). As illustrated in the conventional process flow below, this method suffers from inherent thermodynamic and kinetic challenges regarding selectivity. Because piperazine possesses two chemically equivalent secondary amine groups, controlling mono-protection is notoriously difficult. To mitigate the formation of the unwanted bis-Boc-piperazine impurity, manufacturers are forced to employ a massive excess of piperazine, typically ranging from 3 to 5 equivalents relative to the protecting agent. This practice results in abysmal atom economy, where the majority of the valuable amine starting material remains unreacted and must be recovered. Furthermore, the recovery of anhydrous piperazine is technically demanding and energy-intensive, often requiring complex distillation setups that drive up operational expenditures. Additionally, even with excess amine, the reaction selectivity remains poor, often generating approximately 20% of the di-substituted impurity, which complicates downstream purification and lowers the overall yield to a suboptimal range of 61-70%.
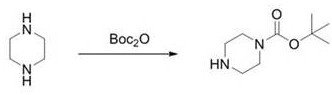
The Novel Approach
In stark contrast to the wasteful direct protection strategy, the method disclosed in patent CN113185478B employs a constructive synthesis route that builds the piperazine ring from simpler, cheaper precursors. By starting with ethylenediamine and chloroacetaldehyde, the process forms the heterocyclic core through a cyclization reaction followed by selective acylation and reduction. This strategic change effectively eliminates the possibility of forming the bis-Boc impurity because the second nitrogen atom is not available for reaction during the critical acylation step. The reaction sequence allows for precise control over the substitution pattern, ensuring that only the desired mono-protected product is formed. Moreover, the raw materials—ethylenediamine and chloroacetaldehyde—are commodity chemicals that are significantly less expensive and more readily available than high-purity anhydrous piperazine. The overall process is streamlined, with the potential for telescoping the acylation and reduction steps into a single pot, thereby reducing solvent consumption and processing time. This novel pathway achieves a remarkable total yield of over 81.5% with product purity exceeding 99.5%, representing a substantial leap forward in process efficiency.
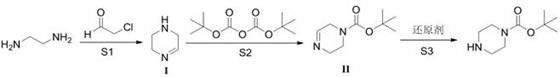
Mechanistic Insights into Constructive Ring Synthesis and Selective Protection
The core innovation of this technology lies in the mechanistic decoupling of ring formation and nitrogen protection. In the first step (S1), ethylenediamine reacts with chloroacetaldehyde under basic conditions to undergo cyclization. This reaction likely proceeds through the formation of an imine or enamine intermediate, resulting in an unsaturated piperazine derivative (Compound I). This intermediate is crucial because it presents a differentiated electronic environment compared to saturated piperazine. In the subsequent step (S2), this intermediate reacts with Boc2O. Due to the specific electronic properties and steric environment of the unsaturated ring, the acylation occurs selectively at one nitrogen center. Unlike saturated piperazine where both nitrogens are equally nucleophilic, the intermediate structure favors mono-substitution. Finally, in step (S3), a reducing agent such as sodium borohydride (NaBH4) or potassium borohydride (KBH4) is employed to reduce the double bond within the ring, saturating the piperazine backbone to yield the final N-Boc-piperazine. This reductive step occurs after the protecting group is already in place, locking the molecule in its mono-protected state and rendering the formation of the bis-Boc impurity chemically impossible.
From an impurity control perspective, this mechanism offers a robust solution to a persistent industry problem. In conventional methods, the bis-Boc impurity has similar polarity and solubility to the target product, making it difficult to remove via crystallization or standard extraction, often necessitating costly column chromatography or repeated recrystallizations. By designing a pathway where the bis-substituted species cannot form, the impurity profile is drastically simplified. The patent data highlights that the crude product obtained via this method exhibits a gas chromatography purity of 99.5% or higher directly after workup. This high level of intrinsic purity reduces the load on quality control laboratories and minimizes the loss of material during purification stages. For process chemists, this means a more predictable and scalable process with fewer variables to control, ensuring consistent batch-to-batch quality which is paramount for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize N-tert-butoxycarbonyl piperazine Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing high-quality N-Boc-piperazine. The process begins with the cyclization of ethylenediamine and chloroacetaldehyde in a solvent system such as methanol or water, utilizing bases like sodium hydroxide or potassium carbonate to drive the reaction at moderate temperatures (25-35°C). Following the isolation of the cyclic intermediate, the material is subjected to acylation with Boc2O in methanol at controlled low temperatures (10-20°C) to ensure selectivity. The final reduction step utilizes common hydride donors to complete the synthesis. Detailed standardized operating procedures, including specific molar ratios, addition rates, and workup parameters, are critical for replicating the high yields reported in the patent examples.
- Cyclization: React ethylenediamine with chloroacetaldehyde in a solvent medium (water/methanol) with a base to form the cyclic intermediate (Compound I).
- Acylation: Treat Compound I with di-tert-butyl dicarbonate (Boc2O) in methanol at low temperature (10-20°C) to install the protecting group.
- Reduction: Reduce the acylated intermediate using a hydride reducing agent (e.g., NaBH4) to yield the final saturated N-tert-butoxycarbonyl piperazine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ethylenediamine-based route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift away from anhydrous piperazine as a starting material addresses one of the most significant cost drivers in the traditional process. Anhydrous piperazine is a specialized, higher-cost reagent that often requires stringent storage conditions to prevent hydration, whereas ethylenediamine is a bulk commodity chemical with a stable and robust global supply chain. By eliminating the need for a 3-to-5-fold excess of amine, the new method drastically reduces the volume of raw materials required per kilogram of finished product. This reduction in material intensity directly translates to lower raw material costs and decreased waste disposal fees, contributing to a more sustainable and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The elimination of excess amine recovery is a major factor in lowering operational costs. In the conventional process, recovering and drying the unreacted piperazine requires significant energy input and specialized equipment. The new constructive route avoids this entirely, as the stoichiometry is much closer to theoretical ideals. Furthermore, the high selectivity of the reaction means that less solvent and fewer purification reagents are needed to remove impurities. The ability to telescope the acylation and reduction steps into a one-pot process further reduces cycle times and labor costs, allowing for higher throughput in existing reactor volumes without the need for capital expansion.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks like ethylenediamine and chloroacetaldehyde mitigates the risk of supply disruptions associated with specialized intermediates. These raw materials are produced on a massive scale for various industries, ensuring consistent availability and price stability. In contrast, the market for high-purity anhydrous piperazine can be more volatile and subject to supply constraints. By diversifying the raw material base to include widely available chemicals, manufacturers can secure a more resilient supply chain that is less susceptible to market fluctuations, ensuring continuous production schedules for downstream API synthesis.
- Scalability and Environmental Compliance: The improved atom economy of this process aligns with increasingly strict environmental regulations regarding waste generation. Traditional methods generate significant amounts of amine-contaminated waste streams that require treatment before disposal. The new method generates significantly less hazardous waste due to the absence of excess amine and the reduction of side products. This simplifies wastewater treatment protocols and reduces the environmental footprint of the facility. Additionally, the mild reaction conditions (mostly ambient to moderate temperatures) and the use of common solvents like methanol make the process highly scalable from pilot plant to multi-ton commercial production with minimal engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and technical specifications provided in patent CN113185478B, offering clarity on the feasibility and advantages of the technology for potential adopters and partners.
Q: Why is the ethylenediamine route superior to direct Boc-protection of piperazine?
A: Direct protection requires a large excess (3-5 equivalents) of expensive anhydrous piperazine to prevent di-substitution, leading to low atom economy and difficult recovery. The ethylenediamine route constructs the ring selectively, inherently preventing di-Boc impurities and utilizing cheaper commodity raw materials.
Q: What is the expected purity of N-Boc-piperazine using this new method?
A: The patent data indicates that this constructive synthesis method achieves product purity levels exceeding 99.5% (by GC), significantly reducing the burden on downstream purification processes compared to conventional methods which often struggle with bis-Boc impurities.
Q: Can steps S2 and S3 be combined for industrial efficiency?
A: Yes, the patent explicitly states that the acylation (S2) and reduction (S3) steps can be performed in a 'one-pot' manner. This telescoping of steps reduces solvent usage, processing time, and overall operational complexity, enhancing scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-tert-butoxycarbonyl piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of the ethylenediamine-based synthesis, ensuring that the stringent purity specifications demanded by the pharmaceutical industry are consistently met. With our rigorous QC labs and commitment to process optimization, we can deliver high-purity N-Boc-piperazine that adheres to the highest quality standards, supporting your drug development and commercial manufacturing needs with reliability and precision.
We invite you to explore how this advanced manufacturing technology can benefit your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic advantages of switching to this superior grade of intermediate. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us collaborate to optimize your production costs and secure a stable supply of this critical pharmaceutical building block.
