Advanced Post-Treatment Technology for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Post-Treatment Technology for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic efficiency, particularly for critical building blocks like N-tert-butoxycarbonyl piperazine. Patent CN114478439A introduces a groundbreaking post-treatment technology that fundamentally alters the economics of Boc-piperazine production by addressing the longstanding challenge of raw material recovery. This innovation is not merely a laboratory curiosity but a scalable solution designed for reliable pharmaceutical intermediate supplier operations aiming to reduce environmental footprints while maximizing yield. By re-engineering the workup phase, the process achieves a stable reaction environment where unreacted anhydrous piperazine can be physically separated and recycled rather than lost to aqueous waste streams. 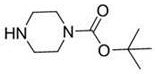 This structural integrity is maintained throughout the novel purification sequence, ensuring that the final API intermediate meets the stringent quality standards required for downstream drug synthesis.
This structural integrity is maintained throughout the novel purification sequence, ensuring that the final API intermediate meets the stringent quality standards required for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis protocols for Boc-piperazine typically rely on a significant excess of anhydrous piperazine, often ranging from three to five equivalents, to suppress the formation of the undesired di-substituted byproduct. While this stoichiometric imbalance effectively drives selectivity, it creates a massive bottleneck during the purification stage, where the standard practice involves washing the reaction mixture with large volumes of water. Since piperazine is highly soluble in water but difficult to extract back into organic solvents, the excess starting material is essentially dissolved into the wastewater, rendering it unrecoverable and leading to substantial raw material wastage. Furthermore, this aqueous workup generates significant volumes of contaminated effluent that requires costly treatment before discharge, thereby inflating both the direct material costs and the environmental compliance burden for manufacturers. The inability to efficiently recycle the expensive piperazine feedstock means that the overall atom economy of the process remains suboptimal, directly impacting the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology disclosed in the patent circumvents these inefficiencies by replacing the initial aqueous wash with a strategic organic solvent precipitation step using a mixture of ethyl acetate and petroleum ether. Instead of dissolving the excess piperazine, this specific solvent combination induces the precipitation of unreacted anhydrous piperazine as a solid, which can then be easily separated via simple filtration. This physical separation allows for the direct recovery and reuse of the starting material, with experimental data indicating a recovery rate of approximately 85%, which dramatically improves the overall process economics. Following the removal of the solid piperazine, the filtrate undergoes a controlled concentration and a subsequent water wash specifically timed to remove the di-Boc impurity without sacrificing the mono-protected product. This sequential decoupling of impurity removal and raw material recovery represents a paradigm shift in process chemistry, offering a pathway for high-purity Boc-piperazine production that is both economically superior and environmentally sustainable.
Mechanistic Insights into Solvent Engineering and Selective Precipitation
The core of this technological advancement lies in the precise manipulation of solubility parameters to achieve selective crystallization of the starting material. In the conventional aqueous workup, the polarity of water matches the high polarity of piperazine, leading to irreversible dissolution; however, the novel process exploits the differential solubility of piperazine in non-polar to moderately polar organic mixtures. By utilizing a specific volume ratio of ethyl acetate to petroleum ether, typically around 1:5, the solvent system creates an environment where the Boc-protected product remains in solution while the free amine (piperazine) reaches its saturation limit and precipitates out. This phenomenon is driven by the lack of solvation energy provided by the hydrocarbon-rich petroleum ether component, which cannot effectively stabilize the polar amine groups of the piperazine molecules. Consequently, the unreacted piperazine aggregates into a filterable solid, allowing for its mechanical separation from the reaction liquor containing the desired mono-Boc product and the di-Boc byproduct.
Furthermore, the mechanism for impurity control is elegantly staged to ensure maximum purity without compromising yield. Once the bulk of the unreacted piperazine is removed via filtration, the remaining filtrate contains the target Boc-piperazine along with trace amounts of the di-substituted impurity. The process then introduces water at this specific stage, leveraging the fact that the di-Boc impurity can be effectively separated or that the product can be extracted into an organic phase like dichloromethane while leaving polar contaminants behind. The final recrystallization from petroleum ether serves as a polishing step, removing any residual solvent or minor organic impurities to deliver a white crystalline powder suitable for sensitive pharmaceutical applications. This multi-stage purification logic ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed with consistent quality, minimizing the risk of batch failures due to impurity carryover.
How to Synthesize Boc-Piperazine Efficiently
The implementation of this synthesis route requires careful attention to solvent ratios and temperature control to maximize the recovery of the starting amine. The process begins with the amidation reaction in dichloromethane at ambient temperature, followed by solvent removal and the critical precipitation step. Operators must adhere to the specified solvent volumes to ensure complete precipitation of the piperazine, as deviations could lead to product loss or incomplete recovery. The detailed standardized synthesis steps, including specific stoichiometric ratios and agitation times, are outlined below to facilitate immediate technology transfer and process validation.
- Conduct amidation reaction between anhydrous piperazine and Boc-anhydride in dichloromethane at room temperature.
- Concentrate the solvent and wash the residue with an ethyl acetate and petroleum ether mixture to precipitate and recover unreacted piperazine.
- Extract the filtrate with dichloromethane after water washing to remove di-Boc impurities, followed by recrystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this post-treatment technology translates directly into enhanced margin protection and supply security. The ability to recover and reuse approximately 85% of the anhydrous piperazine significantly reduces the net consumption of raw materials per kilogram of finished product, which acts as a powerful hedge against volatility in amine pricing. Unlike traditional methods where excess reagents are treated as sunk costs, this process treats them as recoverable assets, effectively lowering the variable cost of goods sold without requiring capital-intensive equipment upgrades. Moreover, the reduction in wastewater volume simplifies environmental compliance procedures, reducing the overhead costs associated with effluent treatment and disposal, which is increasingly critical in regions with strict environmental regulations.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic reduction in raw material waste through the efficient recycling of anhydrous piperazine. By avoiding the loss of expensive starting materials into aqueous waste streams, manufacturers can achieve substantial cost savings that compound over large production volumes. Additionally, the simplified workup procedure reduces the consumption of processing solvents and energy required for extensive aqueous extractions, further contributing to a leaner manufacturing cost structure. This efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a distinct advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is essential for maintaining uninterrupted supply lines to downstream API manufacturers. The simplicity of the filtration-based recovery step reduces the risk of operational errors compared to complex multi-stage extractions, leading to higher first-pass yields and more predictable production schedules. Furthermore, the reduced dependency on massive volumes of process water mitigates risks associated with water scarcity or utility fluctuations, ensuring that production can continue reliably even under resource-constrained conditions.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns perfectly with green chemistry principles by maximizing atom utilization and minimizing waste generation. The elimination of large-scale aqueous washing steps significantly reduces the volume of hazardous wastewater, easing the burden on treatment facilities and lowering the environmental footprint of the manufacturing site. This eco-friendly profile not only aids in meeting regulatory requirements but also enhances the brand value of the supply chain, appealing to global partners who prioritize sustainable sourcing practices in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented post-treatment process. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method outperforms legacy synthesis routes. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this technology for their own production lines.
Q: How does the new post-treatment process improve raw material utilization compared to conventional methods?
A: Conventional methods require washing with large amounts of water, which dissolves excess piperazine and makes recovery difficult. The patented process uses an organic solvent mixture (ethyl acetate/petroleum ether) to precipitate unreacted piperazine as a solid, allowing for filtration and direct reuse with a recovery rate of approximately 85%.
Q: What is the primary mechanism for removing di-Boc-piperazine impurities in this synthesis?
A: After recovering the unreacted piperazine via organic solvent washing, the process utilizes a specific water washing step on the concentrated filtrate. This step effectively separates the di-Boc-piperazine impurity through filtration before the final extraction and recrystallization, ensuring high product purity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It operates at room temperature, avoids complex cryogenic conditions, and simplifies the workup procedure by replacing difficult aqueous extractions of piperazine with straightforward filtration steps, significantly reducing wastewater generation and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boc-Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and proven scaling capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN114478439A can be fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Boc-piperazine meets the exacting standards required for modern pharmaceutical synthesis, delivering consistency that R&D directors can trust.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain for maximum efficiency. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your volume requirements and operational constraints. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and reliability in your pharmaceutical intermediate sourcing strategy.
