Revolutionizing Dasatinib Production via Ionic Liquid Catalysis and Scalable Green Chemistry
Revolutionizing Dasatinib Production via Ionic Liquid Catalysis and Scalable Green Chemistry
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways for the production of critical oncology therapeutics. Patent CN109678853B introduces a groundbreaking preparation process for Dasatinib, a potent tyrosine kinase inhibitor used in treating chronic myelogenous leukemia. This technology represents a significant departure from legacy synthetic routes by leveraging a novel ionic liquid catalyst system and green solvent methodologies. For R&D directors and procurement specialists, this patent offers a compelling blueprint for reducing manufacturing complexity while enhancing product purity. The core innovation lies in a streamlined three-step sequence that avoids hazardous volatile intermediates and expensive transition metal catalysts, positioning it as a superior alternative for commercial scale-up. By integrating mild reaction conditions with high-yield transformations, this process addresses the critical pain points of supply chain reliability and cost efficiency in API manufacturing.
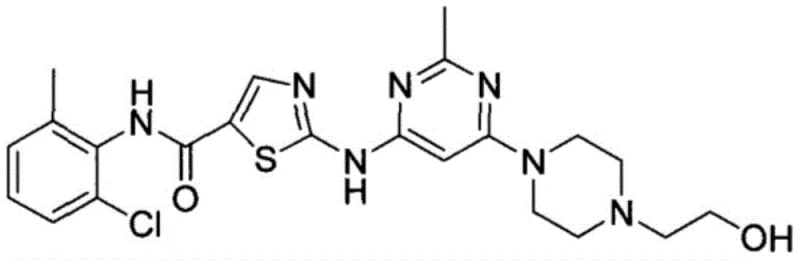
Dasatinib, chemically known as N-(2-chloro-6-methylphenyl)-2-[6-[4-(2-hydroxyethyl)-1-piperazinyl]-2-methyl-4-pyrimidinyl]amino-5-thiazolecarboxamide, is a high-value active pharmaceutical ingredient. The structural complexity of this molecule traditionally necessitates multi-step syntheses involving sensitive reagents. However, the methodology disclosed in CN109678853B simplifies this landscape by utilizing ethyl 3-oxopropionate as a stable starting material. This strategic choice bypasses the instability issues associated with traditional acryloyl chloride intermediates. Furthermore, the implementation of a one-pot synthesis strategy for the final coupling step drastically reduces unit operations, solvent consumption, and processing time. For a reliable API intermediate supplier, adopting such a process translates directly into enhanced production capacity and reduced lead times for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dasatinib has been plagued by severe operational hazards and economic inefficiencies. Prior art routes, such as those described in early medicinal chemistry literature, often rely on cryogenic conditions requiring n-butyllithium at temperatures as low as -78°C. Such extreme conditions impose immense energy burdens and require specialized reactor infrastructure that is difficult to maintain in large-scale industrial settings. Additionally, many conventional pathways utilize palladium-catalyzed cross-coupling reactions involving expensive ligands like BINAP. These noble metal catalysts not only drive up raw material costs but also introduce significant challenges in residual metal removal, a critical quality attribute for oncology drugs. Another prevalent issue in legacy processes is the reliance on volatile intermediates like (E)-3-ethoxyacryloyl chloride, which poses storage and handling risks due to its high volatility and instability.
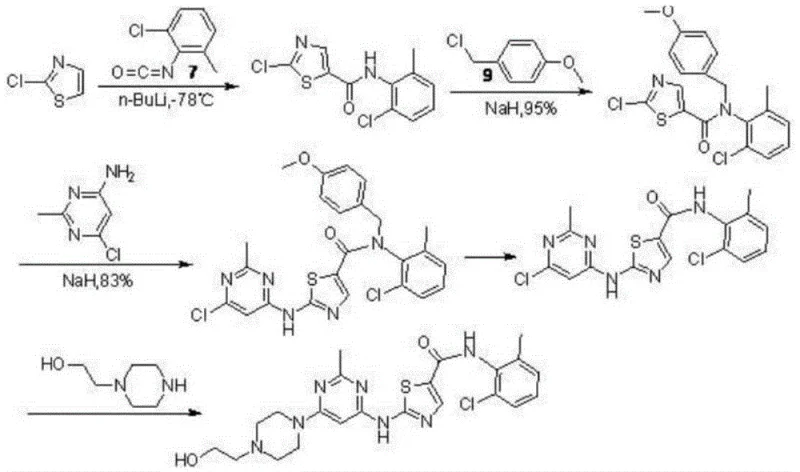
Furthermore, traditional methods often suffer from poor atom economy and low overall yields, sometimes dropping below 50% across the entire synthetic sequence. The use of hazardous solvents and the generation of substantial chemical waste further complicate regulatory compliance and environmental management. For instance, routes involving repeated use of sodium hydride or lithium reagents generate significant inorganic waste streams. The formation of difficult-to-separate by-products, such as bipyrimidine ring compounds during the coupling stages, necessitates complex purification protocols like column chromatography, which are impractical for ton-scale production. These cumulative factors render many historical synthetic routes economically unviable for modern generic competition and cost-sensitive markets.
The Novel Approach
In stark contrast, the process defined in CN109678853B offers a paradigm shift towards sustainable manufacturing. The novel approach initiates with a copper-mediated bromination of ethyl 3-oxopropionate, avoiding the need for hazardous N-bromosuccinimide (NBS) or volatile acryloyl chlorides. This step proceeds under reflux conditions in tetrahydrofuran, yielding the key intermediate with exceptional purity exceeding 99%. Subsequently, the thiazole ring construction is achieved in water, a truly green solvent, at room temperature. This eliminates the need for toxic organic solvents during cyclization and leverages the hydrophobic effect to drive the reaction forward efficiently. The culmination of this process is a sophisticated one-pot reaction where the pyrimidine core is assembled using an ionic liquid catalyst, 1-butyl-3-methylimidazole glycinate.
This one-pot strategy allows for the sequential addition of 4,6-dichloro-2-methylpyrimidine, N-hydroxyethyl piperazine, and the thiazole amine without isolating unstable intermediates. The use of potassium phosphate as a base in conjunction with the ionic liquid creates a highly active nucleophilic environment, facilitating rapid substitution at the pyrimidine 4-position followed by the 6-position. This telescoping of steps significantly reduces solvent usage and processing time. The result is a robust, high-yielding process that achieves final product purities greater than 99.9% with simple recrystallization. For procurement managers, this translates to cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material waste and maximizing throughput per batch.
Mechanistic Insights into Ionic Liquid Catalyzed Pyrimidine Coupling
The heart of this technological advancement lies in the unique role of the 1-butyl-3-methylimidazole glycinate ([Bmim][Gly]) ionic liquid. Unlike traditional organic bases or phase transfer catalysts, this task-specific ionic liquid acts as both a solvent modifier and a catalytic promoter. The glycinate anion serves as a mild yet effective base that deprotonates the amine nucleophiles without causing degradation of the sensitive pyrimidine ring. Simultaneously, the imidazolium cation stabilizes the transition state through hydrogen bonding interactions, lowering the activation energy for the nucleophilic aromatic substitution (SNAr) reactions. This dual functionality ensures that the reaction proceeds smoothly at moderate temperatures (80°C), avoiding the thermal degradation often seen in harsher basic conditions.
Moreover, the mechanistic pathway effectively suppresses the formation of regioisomers and bis-substituted by-products. In conventional methods, controlling the selectivity between the 4-chloro and 6-chloro positions on the pyrimidine ring is challenging, often leading to mixtures that are difficult to separate. The ionic liquid system creates a specific solvation shell around the reactants, kinetically favoring the desired substitution sequence. First, the more reactive 4-chloro position is displaced by N-hydroxyethyl piperazine, followed by the displacement of the 6-chloro position by the thiazole amine. This controlled reactivity is crucial for maintaining high purity profiles. Additionally, the use of copper bromide in the initial step provides a cleaner bromination profile compared to radical bromination agents, ensuring that the thiazole precursor is free from poly-brominated impurities that could carry through to the final API.
How to Synthesize Dasatinib Efficiently
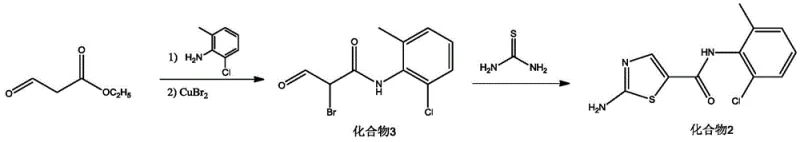
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for manufacturing Dasatinib intermediates and the final API. The process is designed to be operationally simple, requiring standard glass-lined or stainless steel reactors without the need for exotic metallurgy or cryogenic cooling systems. The initial formation of the brominated ketone intermediate sets the stage for high purity, while the aqueous cyclization step demonstrates the feasibility of green chemistry in industrial settings. The final coupling step is particularly noteworthy for its telescoped nature, allowing operators to add reagents sequentially in a single vessel. Detailed standardized synthetic steps see the guide below.
- React ethyl 3-oxopropionate with 2-chloro-6-methylaniline under alkaline conditions, followed by copper bromide addition to obtain Compound 3.
- Cyclize Compound 3 with thiourea in water at room temperature to synthesize 2-amino-N-(2-chloro-6-methylphenyl)thiazole-5-formamide.
- Perform a one-pot reaction of 4,6-dichloro-2-methylpyrimidine, N-hydroxyethyl piperazine, and the thiazole intermediate using K3PO4 and an ionic liquid catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of the CN109678853B process offers tangible strategic benefits beyond mere technical elegance. The elimination of cryogenic reagents like n-butyllithium removes a significant bottleneck in production scheduling and safety compliance. Facilities no longer need to allocate resources for maintaining ultra-low temperature infrastructure, thereby freeing up capital for other capacity expansions. Furthermore, the substitution of expensive palladium catalysts with abundant copper salts and recyclable ionic liquids results in substantial cost savings on raw materials. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates supply risk associated with fluctuating precious metal markets.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by removing the need for costly noble metal catalysts and hazardous volatile intermediates. By utilizing water as a solvent for the cyclization step, the expense associated with purchasing, recovering, and disposing of large volumes of organic solvents is drastically reduced. The high yields reported in the patent examples, often exceeding 90% for intermediates and nearly 90% for the final step, mean that less raw material is wasted per kilogram of product. This improved atom economy directly correlates to a lower cost base, allowing for more competitive pricing in the generic pharmaceutical market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Relying on stable, commodity-grade starting materials like ethyl 3-oxopropionate and 2-chloro-6-methylaniline ensures a robust supply chain unaffected by the shortages often seen with specialized reagents. The avoidance of thermally unstable intermediates means that materials can be stored safely for longer periods, providing a buffer against demand spikes. Additionally, the simplified workflow reduces the number of discrete unit operations, shortening the overall manufacturing cycle time. This agility allows suppliers to respond more rapidly to market demands, reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent availability for downstream formulation partners.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as the use of water and the reduction of hazardous waste, align perfectly with increasingly stringent global environmental regulations. The absence of heavy metal residues simplifies the purification process and reduces the burden on wastewater treatment facilities. Scalability is inherently supported by the mild reaction conditions; the exothermic risks associated with lithiation or hydride reductions are eliminated, making the process safer to run in large-volume reactors. This ease of scale-up from pilot to commercial production ensures that supply continuity can be maintained as market volume grows, without the need for complex process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this novel Dasatinib synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or licensing.
Q: How does the ionic liquid catalyst improve dasatinib synthesis yield?
A: The use of 1-butyl-3-methylimidazole glycinate ([Bmim][Gly]) combined with K3PO4 facilitates a highly efficient one-pot nucleophilic substitution, eliminating the need for harsh conditions and improving overall yield to over 89%.
Q: What are the safety advantages of this new process compared to traditional routes?
A: This process avoids the use of volatile and hazardous intermediates like (E)-3-ethoxyacryloyl chloride and eliminates the need for cryogenic conditions (-78°C) or expensive palladium catalysts, significantly enhancing operational safety.
Q: Is water used as a solvent in any stage of this synthesis?
A: Yes, the cyclization step to form the thiazole ring utilizes water as a green, non-toxic solvent, replacing traditional organic solvents and reducing environmental impact while maintaining high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dasatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ionic liquid-catalyzed synthesis described in CN109678853B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this green chemistry process, including the safe handling of ionic liquids and the efficient management of aqueous workups. We are committed to delivering Dasatinib and its key intermediates with stringent purity specifications, ensuring that every batch meets the rigorous quality standards required for oncology therapeutics. Our rigorous QC labs employ advanced analytical techniques to verify the absence of genotoxic impurities and residual metals, guaranteeing product safety.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By optimizing this route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate the availability of high-quality, affordable Dasatinib to patients worldwide while adhering to the highest standards of sustainability and operational excellence.
