Advanced Catalytic Synthesis of (Z)-3-(bromomethylene)isoindolin-1-one for Commercial Scale-up
Advanced Catalytic Synthesis of (Z)-3-(bromomethylene)isoindolin-1-one for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds that serve as pivotal building blocks for drug discovery. Patent CN101074209A introduces a highly efficient synthetic pathway for (Z)-3-(bromomethylene)isoindolin-1-one, a versatile intermediate characterized by its unique bromomethylene functionality attached to the isoindolinone core. This compound is not merely a static molecule but a dynamic synthon capable of undergoing diverse transformations, making it invaluable for the synthesis of bioactive agents targeting neurodegenerative diseases and hypertension. The structural integrity and specific stereochemistry of this molecule are crucial for its downstream reactivity, as evidenced by its defined melting point of 176-178°C and characteristic spectroscopic data. By leveraging a copper-catalyzed cyclization strategy, this technology offers a streamlined alternative to traditional multi-step sequences, directly addressing the industry's demand for reliable pharmaceutical intermediates supplier capabilities that prioritize both quality and efficiency.
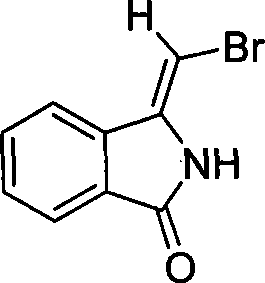
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of substituted isoindolin-1-one derivatives has often relied on harsh reaction conditions that pose significant challenges for large-scale manufacturing and environmental compliance. Traditional routes frequently involve the use of stoichiometric amounts of toxic reagents, extreme temperatures, or prolonged reaction times that degrade sensitive functional groups and lead to complex impurity profiles. Furthermore, conventional methods for introducing halogenated methylene groups onto heterocyclic rings often suffer from poor regioselectivity and stereoselectivity, resulting in difficult-to-separate isomeric mixtures that compromise the purity required for pharmaceutical applications. These inefficiencies translate directly into increased production costs, extended lead times, and substantial waste generation, creating bottlenecks for procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing. The reliance on non-catalytic processes also limits the atom economy of the synthesis, making it less sustainable and economically viable in a competitive global market where green chemistry principles are increasingly mandated.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN101074209A utilizes a catalytic system centered around cuprous iodide (CuI) paired with nitrogen-containing ligands to drive the cyclization under remarkably mild conditions. This innovative approach operates in toluene at moderate temperatures of 100-110°C, significantly reducing energy consumption and thermal stress on the reactants compared to high-temperature pyrolysis or strong acid-mediated cyclizations. The use of a catalytic amount of copper, rather than stoichiometric reagents, enhances the atom economy and simplifies the downstream purification process, as there are fewer metal residues to remove. The reaction demonstrates excellent chemoselectivity, specifically targeting the formation of the (Z)-isomer, which is critical for maintaining the biological activity of downstream derivatives. This operational simplicity, combined with high yields ranging from 67% to 77% across different scales, represents a paradigm shift towards more sustainable and commercially attractive synthetic routes for complex heterocycles.
Mechanistic Insights into CuI-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the intricate interplay between the copper catalyst, the nitrogen ligand, and the inorganic base, which collectively facilitate an intramolecular nucleophilic attack leading to ring closure. The mechanism likely initiates with the coordination of the CuI species to the nitrogen ligand, forming an active catalytic complex that activates the amide nitrogen or the vinyl bromide moiety. Under the basic conditions provided by carbonates or phosphates, the amide nitrogen becomes sufficiently nucleophilic to attack the electron-deficient vinyl carbon, displacing a bromide ion in a concerted or stepwise fashion. The presence of the nitrogen ligand is paramount, as it stabilizes the copper center in the +1 oxidation state, preventing disproportionation and ensuring the catalytic cycle continues efficiently throughout the 3 to 4-hour reaction window. This precise control over the electronic environment of the catalyst allows for the selective formation of the five-membered lactam ring without affecting other sensitive parts of the molecule.
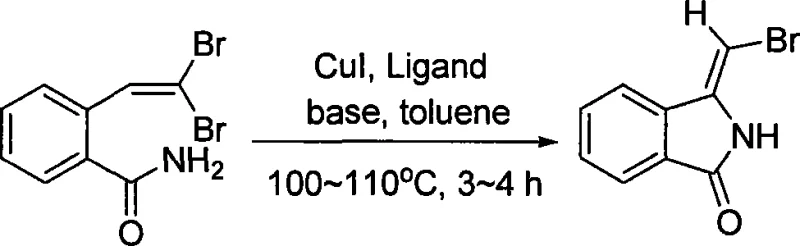
Furthermore, the selectivity observed in this transformation is a direct result of the specific geometric constraints imposed by the catalyst-substrate complex, which favors the formation of the (Z)-isomer over the (E)-isomer. Impurity control is inherently built into this mechanism; by avoiding radical pathways or harsh acidic conditions that typically lead to polymerization or decomposition, the process maintains a clean reaction profile. The choice of inorganic base, such as potassium carbonate or cesium carbonate, plays a dual role in neutralizing the generated hydrogen bromide and facilitating the deprotonation steps necessary for the cyclization to proceed. Understanding these mechanistic nuances is essential for R&D directors aiming to optimize the process further or adapt it for analogous substrates, as slight modifications in ligand sterics or base strength can fine-tune the reaction outcome. This deep mechanistic understanding ensures that the commercial scale-up of complex pharmaceutical intermediates remains robust and reproducible.
How to Synthesize (Z)-3-(bromomethylene)isoindolin-1-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the specified molar ratios and environmental controls to achieve the reported high yields and purity. The process begins with the precise weighing of 2-(2,2-dibromovinyl)benzamide, which serves as the key starting material, along with the catalytic system components. It is imperative to maintain anhydrous and anaerobic conditions throughout the setup, typically achieved through nitrogen purging or the use of Schlenk line techniques, to prevent catalyst deactivation by moisture or oxygen. The detailed standardized synthesis steps outlined below provide a clear roadmap for technical teams to replicate the success of the patent examples, ensuring consistency from gram-scale experiments to multi-kilogram production runs.
- Dissolve 2-(2,2-dibromovinyl)benzamide, inorganic base, cuprous iodide, and nitrogen-containing ligand in toluene under anhydrous and anaerobic conditions.
- Heat the reaction mixture to reflux at 100-110°C and maintain stirring for 3 to 4 hours to ensure complete cyclization.
- Filter the reaction mixture, wash solids with ethyl acetate, remove solvent via rotary evaporation, and purify the residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic methodology offers tangible strategic advantages that extend beyond mere technical feasibility. The simplification of the synthetic route directly correlates with a reduction in the number of unit operations required, which minimizes equipment occupancy time and labor costs associated with batch processing. By eliminating the need for exotic or hazardous reagents that require special handling and disposal protocols, the overall operational expenditure is significantly lowered, aligning with corporate sustainability goals. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in raw material quality or environmental parameters, thereby enhancing supply chain reliability and reducing the risk of batch failures. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of inexpensive and readily available catalysts like cuprous iodide and common inorganic bases, replacing costly stoichiometric reagents. The high selectivity of the reaction reduces the burden on purification steps, meaning less solvent consumption and lower waste treatment costs, which are major contributors to the final cost of goods. Additionally, the ability to run the reaction at moderate temperatures reduces energy consumption for heating and cooling, further contributing to substantial cost savings in pharmaceutical intermediates manufacturing. The simplified workup procedure, involving simple filtration and evaporation, streamlines the production workflow, allowing for faster turnover of batches and improved asset utilization.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, potassium carbonate, and cuprous iodide ensures that the supply chain is not vulnerable to the volatility associated with specialized or single-source reagents. This diversification of raw material sources mitigates the risk of supply disruptions, ensuring a steady flow of materials to the production line. The short reaction time of 3 to 4 hours allows for high throughput, enabling manufacturers to respond quickly to changes in market demand without the need for extensive inventory buffering. This agility is a key factor in reducing lead time for high-purity pharmaceutical intermediates, providing a competitive edge in a fast-paced industry.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the use of standard reactor materials and conditions that do not require specialized high-pressure or cryogenic equipment. The process generates minimal hazardous waste, as the primary byproducts are inorganic salts that can be easily separated and disposed of in accordance with environmental regulations. This alignment with green chemistry principles not only reduces regulatory compliance costs but also enhances the corporate image of the manufacturer as a responsible producer. The scalability of this route ensures that it can meet the growing demand for this intermediate as it moves through the drug development pipeline, from preclinical studies to commercial API production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of (Z)-3-(bromomethylene)isoindolin-1-one, derived directly from the experimental data and claims of the underlying patent. These insights are designed to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their supply chains. Understanding these specifics helps in making informed decisions regarding process adoption and vendor selection.
Q: What are the critical reaction conditions for this synthesis?
A: The process requires anhydrous and anaerobic conditions using toluene as a solvent, with a reaction temperature maintained between 100-110°C for 3-4 hours.
Q: Which ligands are compatible with the CuI catalyst system?
A: Effective nitrogen-containing ligands include 2,2'-bipyridine, TMEDA, 1,2-cyclohexanediamine, and 1,10-phenanthroline, which stabilize the copper species.
Q: What is the expected purity and yield of the final product?
A: The patented method demonstrates high selectivity and yields ranging from 67% to 77%, producing a pale yellow solid suitable for further pharmaceutical derivatization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-3-(bromomethylene)isoindolin-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our expertise in copper-catalyzed transformations allows us to optimize this specific route for maximum yield and minimal impurity formation, providing our partners with a consistent and reliable source of this valuable building block.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this catalytic method for your supply chain. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your requirements for high-purity pharmaceutical intermediates. Let us collaborate to accelerate your drug development timeline with superior chemical solutions.
