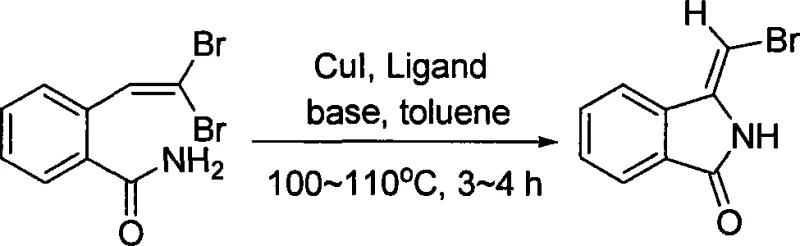
Advanced Cu-Catalyzed Synthesis of (Z)-3-(bromomethylene)isoindolin-1-one for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN101074209A introduces a significant advancement in this domain by detailing a novel synthetic methodology for (Z)-3-(bromomethylene)isoindolin-1-one. This specific isoindolinone derivative represents a high-value intermediate, possessing a unique bromomethylene functionality that serves as a versatile handle for further diversification. The patent highlights a streamlined approach utilizing copper catalysis, which stands in stark contrast to traditional multi-step sequences often required to construct such densely functionalized nitrogen-containing heterocycles. By leveraging a one-pot cyclization strategy, this technology addresses key pain points in process chemistry, including step economy and operational simplicity, thereby offering a compelling value proposition for manufacturers aiming to optimize their supply chains for active pharmaceutical ingredients (APIs) and advanced intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted isoindolin-1-ones has relied on methodologies that are often fraught with inefficiencies and environmental burdens. Traditional routes frequently involve the use of expensive noble metal catalysts, such as palladium or rhodium, which not only inflate raw material costs but also introduce stringent requirements for residual metal removal to meet regulatory standards for pharmaceutical products. Furthermore, conventional strategies often necessitate harsh reaction conditions, including strong acids or bases and elevated temperatures that can compromise the integrity of sensitive functional groups, leading to lower overall yields and complex impurity profiles. The requirement for multiple protection and deprotection steps to achieve regioselectivity further exacerbates the problem, resulting in prolonged production cycles and increased waste generation. These factors collectively hinder the ability of procurement teams to secure cost-effective and reliable supplies of high-purity intermediates, creating bottlenecks in the development of new drug candidates.
The Novel Approach
The methodology disclosed in CN101074209A offers a transformative solution by employing a copper-catalyzed intramolecular coupling reaction that proceeds under relatively mild conditions. This innovative route utilizes readily available 2-(2,2-dibromovinyl)benzamide as the starting material, which undergoes a concerted cyclization and functionalization in the presence of cuprous iodide (CuI), a nitrogen-containing ligand, and an inorganic base. The use of toluene as a solvent at reflux temperatures of 100-110°C ensures excellent solubility and reaction kinetics without the need for exotic or hazardous reagents. Crucially, this process demonstrates high stereoselectivity, predominantly yielding the (Z)-isomer, which is often the biologically relevant configuration. By eliminating the need for noble metals and reducing the number of synthetic steps, this approach significantly simplifies the manufacturing workflow, offering a direct path to cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards.
Mechanistic Insights into CuI-Catalyzed Intramolecular Cyclization
The core of this synthetic breakthrough lies in the efficient activation of the carbon-bromine bond by the copper catalyst, facilitating an intramolecular nucleophilic attack by the amide nitrogen. The reaction mechanism likely involves the formation of an active copper-ligand complex that coordinates with the dibromovinyl moiety, promoting oxidative addition or a similar activation step. The presence of the nitrogen ligand, such as 1,2-cyclohexanediamine or TMEDA, is critical for stabilizing the copper species and enhancing its catalytic turnover number. The inorganic base plays a dual role: it neutralizes the hydrogen bromide generated during the cyclization and assists in the deprotonation of the amide nitrogen, rendering it more nucleophilic. This synergistic interaction between the catalyst, ligand, and base ensures a smooth transformation with minimal side reactions, such as debromination or polymerization, which are common pitfalls in halogenated substrate chemistry.
Furthermore, the control of stereochemistry is a paramount feature of this mechanism. The formation of the (Z)-isomer is governed by the steric and electronic environment of the transition state during the ring-closing step. The specific geometry of the copper intermediate favors the retention of the bromine atom in the Z-configuration relative to the carbonyl group, avoiding the thermodynamic equilibration that might lead to E/Z mixtures. This high level of selectivity is invaluable for R&D directors, as it eliminates the need for costly and yield-reducing chromatographic separations of isomers. The robustness of this catalytic system allows for a broad scope of substrates, suggesting that this methodology could be adapted for the synthesis of various analogues, thereby expanding the chemical space available for drug discovery programs targeting neurodegenerative diseases or hypertension.
How to Synthesize (Z)-3-(bromomethylene)isoindolin-1-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the precise weighing of 2-(2,2-dibromovinyl)benzamide, which is then combined with the copper catalyst and ligand in a molar ratio that maximizes catalytic efficiency while minimizing metal loading. The choice of solvent, specifically toluene, is critical for maintaining the reaction temperature at the optimal reflux range of 100-110°C. It is imperative to maintain anhydrous and anaerobic conditions throughout the setup to prevent catalyst deactivation and oxidation of sensitive intermediates. Following the reaction period of 3 to 4 hours, a straightforward workup procedure involving filtration and solvent removal yields the crude product, which can be further purified to meet stringent purity specifications. For detailed standardized synthesis steps, please refer to the guide below.
- Charge a reactor with 2-(2,2-dibromovinyl)benzamide, inorganic base (e.g., K2CO3), CuI catalyst, and nitrogen ligand in toluene under anhydrous conditions.
- Heat the reaction mixture to reflux at 100-110°C for 3-4 hours to facilitate the intramolecular cyclization and bromination.
- Filter the mixture, wash solids with ethyl acetate, concentrate the filtrate, and purify via silica gel column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed route offers substantial strategic advantages for procurement managers and supply chain heads tasked with securing reliable sources of complex intermediates. The shift from noble metals to base metals like copper represents a fundamental change in the cost structure of the synthesis, removing the volatility associated with precious metal markets. Additionally, the use of commodity chemicals such as toluene and potassium carbonate ensures that the supply chain remains resilient against disruptions, as these materials are globally available in bulk quantities. The simplified operational profile, characterized by a single reaction vessel and standard heating requirements, reduces the capital expenditure needed for specialized equipment, making it easier for contract manufacturing organizations (CMOs) to adopt this technology rapidly. This accessibility translates directly into shorter lead times and more competitive pricing for the final high-purity pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with economical cuprous iodide drastically lowers the direct material costs associated with the synthesis. Moreover, the reduced catalyst loading (0.05-0.1 equivalents) minimizes the burden on downstream purification processes, as there is less metal residue to remove, thereby saving on adsorbents and processing time. The high atom economy of the cyclization reaction ensures that a significant portion of the starting material mass is incorporated into the final product, reducing waste disposal costs. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on the quality or purity of the (Z)-3-(bromomethylene)isoindolin-1-one.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials and reagents mitigates the risk of supply shortages that often plague niche chemical syntheses. Since the reaction conditions are mild and utilize standard solvents, the process can be easily transferred between different manufacturing sites globally, ensuring business continuity. The robustness of the reaction, evidenced by consistent yields across different scales (from 0.1 mmol to 10 mmol in the examples), suggests that scaling up to multi-kilogram or tonne levels will encounter fewer technical hurdles. This predictability allows supply chain planners to forecast inventory needs more accurately and maintain optimal stock levels, ensuring a steady flow of materials to downstream API synthesis units.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by minimizing the use of hazardous reagents and reducing the overall E-factor (mass of waste per mass of product). The absence of toxic heavy metals simplifies the treatment of effluent streams, helping manufacturers comply with increasingly stringent environmental regulations. The ability to run the reaction at atmospheric pressure in a closed reflux system enhances operational safety, reducing the risk of accidents associated with high-pressure reactors. These factors collectively make the technology highly scalable, supporting the commercial scale-up of complex heterocycles from gram-scale R&D batches to industrial production volumes while maintaining a sustainable environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of (Z)-3-(bromomethylene)isoindolin-1-one. These insights are derived directly from the experimental data and technical specifications provided in patent CN101074209A, ensuring that stakeholders have access to accurate and actionable information. Understanding the nuances of this catalytic system is essential for evaluating its fit within existing manufacturing portfolios and for assessing its potential to drive innovation in drug development pipelines. We encourage technical teams to review these details carefully to fully appreciate the operational benefits and versatility of this synthetic route.
Q: What is the typical yield for this Cu-catalyzed cyclization?
A: According to patent CN101074209A, the process achieves yields ranging from 67% to 77% depending on the specific ligand and base combination used, demonstrating robust efficiency for industrial application.
Q: Can this process be scaled for commercial production?
A: Yes, the use of common solvents like toluene and standard reflux temperatures (100-110°C) makes this highly amenable to large-scale manufacturing without requiring specialized high-pressure equipment.
Q: Why is the Z-isomer significant in this synthesis?
A: The high stereoselectivity for the (Z)-isomer is crucial for downstream biological activity and further functionalization, eliminating the need for difficult separation of geometric isomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-3-(bromomethylene)isoindolin-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating the drug discovery and development timeline. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify identity and assay. By partnering with us, you gain access to a supply chain that is not only robust and compliant but also optimized for cost-efficiency, allowing you to focus on your core competencies in therapeutic innovation.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can be integrated into your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new candidates, we are ready to provide the support you need. Contact us today to request a quote and discover how NINGBO INNO PHARMCHEM can serve as your strategic partner in bringing life-saving medicines to market faster and more efficiently.
