Scalable Synthesis of N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide for Advanced Kinase Inhibitor Manufacturing
Scalable Synthesis of N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide for Advanced Kinase Inhibitor Manufacturing
The pharmaceutical landscape for oncology treatments continues to evolve, driven by the demand for robust and scalable synthetic routes for tyrosine kinase inhibitors. Patent CN101654416B introduces a pivotal advancement in the preparation of N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide, a critical intermediate for a class of compounds exhibiting extensive anti-tumor activity, including analogs of the blockbuster drug Imatinib. This technology addresses long-standing inefficiencies in the supply chain for pharmaceutical intermediates, offering a pathway that balances high chemical fidelity with industrial practicality. By leveraging a novel amidation strategy followed by versatile functionalization, this method provides a reliable foundation for the commercial production of complex heterocyclic therapeutics.
![Chemical structure of N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide (Formula I)](/insights/img/n-3-nitro-4-methyl-phenyl-4-aldehyde-benzamide-synthesis-pharma-supplier-20260308085940-01.png)
The core innovation lies in the structural integrity of the intermediate shown in Formula I, which serves as a versatile precursor. Unlike previous iterations that struggled with low overall yields and hazardous reagents, this approach utilizes readily available starting materials to construct the benzamide backbone efficiently. For R&D directors focused on impurity profiles, the controlled reaction conditions minimize the formation of difficult-to-remove byproducts, ensuring that the resulting high-purity pharmaceutical intermediate meets stringent regulatory standards required for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-[4-methyl-3-(4-(3-pyridyl) pyrimidine-2-amino)-phenyl]-4-substituted-benzamide compounds has been plagued by significant technical and economic hurdles. Early methods, such as those described in European patent EP 564409, suffered from excessively long reaction sequences and dismal overall yields often falling below 10 percent. A critical bottleneck in these traditional routes was the reliance on palladium-catalyzed hydrogenation and subsequent acylation steps that required rigorous column chromatography purification, a technique that is notoriously difficult to translate to multi-ton commercial scale-up of complex pharmaceutical intermediates. Furthermore, alternative routes disclosed in Chinese patent CN1630648 utilized trimethylaluminum, a pyrophoric reagent that poses severe safety risks upon exposure to air or moisture, creating substantial liability and infrastructure costs for manufacturing facilities.
The Novel Approach
The methodology outlined in CN101654416B represents a paradigm shift towards safer and more efficient API intermediate manufacturing. By initiating the synthesis with the direct condensation of 3-nitro-4-methylaniline and 4-formylbenzoyl chloride, the process bypasses the need for dangerous organometallic reagents in the early stages. This novel approach not only streamlines the workflow but also drastically simplifies the purification protocol. Instead of relying on silica gel chromatography, the product can be isolated through straightforward filtration and washing procedures. This transition from complex purification to crystallization-based isolation is a key driver for cost reduction in pharmaceutical intermediate manufacturing, as it significantly reduces solvent consumption and processing time while enhancing the safety profile of the entire production line.
Mechanistic Insights into Amidation and Reductive Functionalization
The chemical elegance of this process is rooted in its stepwise construction of the molecular architecture. The initial step involves a nucleophilic acyl substitution where the amine group of 3-nitro-4-methylaniline attacks the carbonyl carbon of 4-formylbenzoyl chloride. The presence of the nitro group deactivates the aromatic ring, necessitating careful control of basicity to facilitate the reaction without promoting hydrolysis of the acid chloride. The use of organic bases like triethylamine in aprotic solvents such as tetrahydrofuran creates an optimal environment for this transformation, ensuring high conversion rates while maintaining the integrity of the sensitive aldehyde moiety for subsequent modifications.
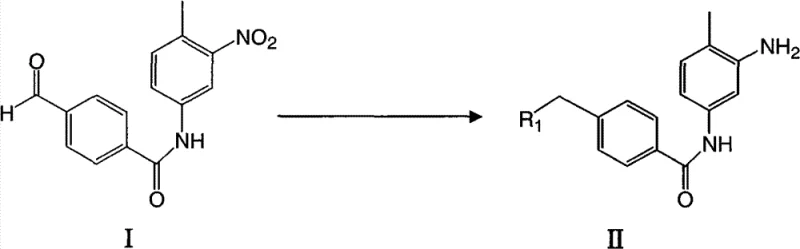
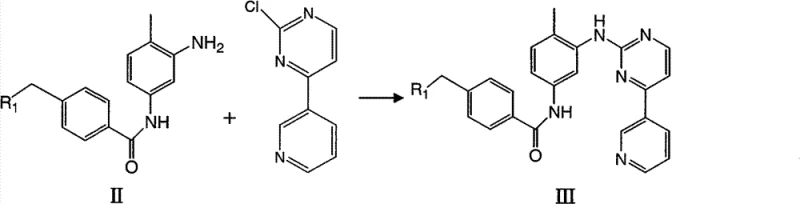
Following the formation of the nitro-benzamide core, the process leverages the reactivity of the aldehyde group through reductive amination. As illustrated in the reaction scheme, the aldehyde functionality serves as a handle for introducing diverse amine side chains, such as N-methylpiperazine or morpholine. This step typically employs mild reducing agents like sodium triacetoxyborohydride, which selectively reduces the iminium intermediate formed in situ without affecting the nitro group or the amide bond. This chemoselectivity is crucial for maintaining a clean impurity profile. The subsequent reduction of the nitro group to an amine, followed by coupling with the pyrimidine fragment, completes the assembly of the pharmacophore. This modular approach allows for the rapid generation of derivative libraries, accelerating the drug discovery timeline for new kinase inhibitors.
How to Synthesize N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide Efficiently
The synthesis protocol detailed in the patent provides a robust framework for producing this key intermediate with high reproducibility. The procedure emphasizes the importance of temperature control during the exothermic amidation step and the stoichiometric balance of reagents during the reductive amination phase to maximize yield. Operators are advised to utilize anhydrous conditions to prevent the hydrolysis of the acid chloride starting material. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the guide below to ensure consistent quality across batches.
- Perform amidation of 3-nitro-4-methylaniline with 4-formylbenzoyl chloride in THF using triethylamine as a base to form the nitro-benzamide core.
- Execute reductive amination on the aldehyde group using secondary amines (e.g., N-methylpiperazine) and sodium triacetoxyborohydride to introduce the solubilizing side chain.
- Conduct nucleophilic aromatic substitution with 2-chloro-4-(3-pyridyl)pyrimidine under basic conditions to finalize the pharmacophore assembly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical yield. The elimination of exotic and hazardous reagents like trimethylaluminum and tin chloride translates directly into reduced operational overhead and lower waste disposal costs. By utilizing commodity chemicals such as 3-nitro-4-methylaniline and 4-formylbenzoyl chloride, manufacturers can secure a more resilient supply chain that is less susceptible to the volatility associated with specialized catalyst markets. This stability is essential for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by removing the need for expensive transition metal catalysts and chiral ligands that characterized earlier methods. The avoidance of column chromatography purification further drives down costs by reducing solvent usage and labor intensity. Additionally, the high atom economy of the amidation step ensures that raw material inputs are converted effectively into the desired product, minimizing waste generation and maximizing the return on investment for every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the key starting materials are bulk commodities available from multiple global suppliers. This diversification mitigates the risk of single-source bottlenecks that can halt production lines. Furthermore, the simplified reaction conditions reduce the dependency on highly specialized equipment or extreme operating parameters, allowing for greater flexibility in manufacturing site selection and ensuring that production can be maintained even during regional supply disruptions or logistical challenges.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns with green chemistry principles by avoiding the generation of heavy metal waste streams associated with tin or palladium residues. The ability to isolate products via crystallization rather than chromatography significantly lowers the volume of organic waste, facilitating easier compliance with increasingly stringent environmental regulations. This scalability ensures that the process can be seamlessly expanded from pilot plant quantities to full commercial production volumes without encountering the technical barriers often faced when scaling complex batch processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical capabilities of the described method for potential partners and stakeholders evaluating this technology for their supply chains.
Q: What are the primary safety advantages of this synthesis route compared to traditional methods?
A: This method eliminates the use of pyrophoric trimethylaluminum and toxic tin chloride reagents found in older patents, significantly reducing hazardous waste generation and operational risk during scale-up.
Q: Does this process require complex purification techniques like column chromatography?
A: No, the patented process is designed for industrial viability, utilizing simple filtration and recrystallization techniques to achieve high purity, thereby avoiding the bottlenecks associated with column chromatography.
Q: Can this intermediate be used for derivatives other than Imatinib?
A: Yes, the aldehyde functionality allows for versatile reductive amination with various amines (morpholine, piperidine, etc.), making it a flexible scaffold for a broad range of anticancer tyrosine kinase inhibitors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to the manufacturing floor. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and potency at every stage of the manufacturing process.
We invite you to collaborate with us to optimize your supply chain for kinase inhibitor production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance the efficiency and reliability of your pharmaceutical manufacturing operations.
