Scalable Synthesis of N-[3-Nitro-4-Methyl-Phenyl]-4-Aldehyde-Benzamide for Oncology Applications
Introduction to Advanced Oncology Intermediate Manufacturing
The landscape of oncology drug manufacturing is constantly evolving, driven by the need for more efficient and safer synthetic routes for complex kinase inhibitors. Patent CN101654416A introduces a pivotal advancement in the synthesis of N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide and its derivatives, which serve as critical precursors for potent anticancer agents like Imatinib. This technology addresses long-standing bottlenecks in the production of 4-substituted-benzamide compounds, specifically targeting the structural motifs found in BCR-ABL tyrosine kinase inhibitors. By shifting away from hazardous reagents and low-yielding multi-step sequences, this methodology offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates. The strategic value of this patent lies not only in the chemical novelty but in its direct applicability to industrial settings where safety, cost, and throughput are paramount concerns for supply chain stability.
![Chemical structure of N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide core scaffold](/insights/img/imatinib-intermediate-synthesis-pharma-supplier-20260309034817-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-[4-methyl-3-(4-(3-pyridyl) pyrimidine-2-amino)-phenyl]-4-substituted-benzamide compounds has been plagued by significant inefficiencies that hinder large-scale production. Prior art, such as European patent EP 564409, describes a route involving the reflux of 3-nitrophenyl guanidine nitrate, which suffers from an overall yield of less than 10% due to the accumulation of losses across multiple steps. Furthermore, key intermediates in these traditional pathways often require purification via column chromatography, a technique that is notoriously difficult to scale and economically prohibitive for tonnage production. Other methods, like those disclosed in Chinese patent CN1630648, rely on trimethylaluminum, a pyrophoric reagent that poses severe safety risks including spontaneous combustion upon contact with air or moisture. Additionally, the use of expensive palladium complexes and phosphine ligands in these legacy routes inflates the cost of goods sold (COGS) and introduces heavy metal contamination risks that require extensive downstream removal processes.
The Novel Approach
In stark contrast, the methodology outlined in CN101654416A streamlines the synthesis into a highly efficient sequence that prioritizes safety and yield. The core innovation involves the direct acylation of 3-nitro-4-methylaniline with 4-formylbenzoyl chloride to form the nitro-benzamide scaffold in high yield. This is followed by a versatile reductive amination step that simultaneously functionalizes the aldehyde moiety and reduces the nitro group, or allows for selective transformation depending on the specific derivative desired. 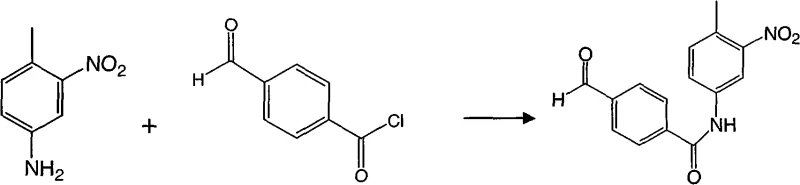 This approach eliminates the need for dangerous organometallic reagents and toxic tin-based reductants. By utilizing common organic solvents like tetrahydrofuran (THF) and dioxane, coupled with readily available bases such as triethylamine, the process achieves isolation yields exceeding 88% for the initial intermediate. This represents a drastic improvement over the sub-50% yields typical of older nitro-intermediate syntheses, thereby providing a reliable foundation for cost reduction in pharmaceutical intermediate manufacturing.
This approach eliminates the need for dangerous organometallic reagents and toxic tin-based reductants. By utilizing common organic solvents like tetrahydrofuran (THF) and dioxane, coupled with readily available bases such as triethylamine, the process achieves isolation yields exceeding 88% for the initial intermediate. This represents a drastic improvement over the sub-50% yields typical of older nitro-intermediate syntheses, thereby providing a reliable foundation for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Reductive Amination and Coupling
The chemical elegance of this process is best understood through the mechanistic details of the reductive amination and subsequent heterocyclic coupling. In the transformation of the aldehyde-benzamide intermediate to the amine-substituted derivative, the reaction proceeds via the formation of an iminium ion intermediate upon the condensation of the aldehyde with the secondary amine (e.g., N-methylpiperazine or morpholine). The use of sodium triacetoxyborohydride as a mild reducing agent ensures high chemoselectivity, reducing the iminium species to the corresponding amine without affecting other sensitive functional groups initially. Subsequent treatment with stronger reducing agents like sodium borohydride or catalytic hydrogenation facilitates the reduction of the aromatic nitro group to the aniline, a crucial step for the final coupling reaction. This tandem or sequential reduction strategy minimizes the number of isolation steps, reducing solvent waste and processing time. The final coupling with 2-chloro-4-(3-pyridyl)pyrimidine is mediated by strong bases such as sodium hydride or potassium tert-butoxide, which deprotonate the newly formed aniline to generate a potent nucleophile capable of displacing the chloride on the pyrimidine ring via an SNAr mechanism.
Impurity control is inherently built into this design through the choice of reagents and purification methods. Unlike previous methods that generated stanniferous waste or required chromatographic separation of closely related byproducts, this route allows for the precipitation of products by adjusting pH and pouring into water. The high purity of the intermediates, confirmed by elemental analysis and mass spectrometry in the patent examples, suggests that side reactions such as over-alkylation or ester hydrolysis are effectively suppressed. The ability to tune the reaction temperature between -20°C and 150°C allows process chemists to optimize the kinetic profile, ensuring that the exothermic nature of the acylation and reduction steps is managed safely. This level of control is essential for maintaining the high-purity pharmaceutical intermediates required for regulatory compliance in drug substance manufacturing.
How to Synthesize N-[3-Nitro-4-Methyl-Phenyl]-4-Aldehyde-Benzamide Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of stoichiometry and temperature control. The process begins with the dissolution of 3-nitro-4-methylaniline in anhydrous THF, followed by the slow addition of triethylamine to scavenge the hydrochloric acid generated during the acylation. The detailed standardized synthesis steps see the guide below for exact parameters regarding addition rates and workup procedures.
- Perform nucleophilic acyl substitution between 3-nitro-4-methylaniline and 4-formylbenzoyl chloride using triethylamine in THF to form the nitro-benzamide core.
- Execute reductive amination on the aldehyde group using secondary amines and sodium triacetoxyborohydride, followed by nitro group reduction to yield the diamino intermediate.
- Couple the resulting aniline intermediate with 2-chloro-4-(3-pyridyl)pyrimidine using strong bases like sodium hydride in DMF to finalize the kinase inhibitor scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible operational benefits that extend beyond simple chemistry. The elimination of hazardous reagents like trimethylaluminum and tin chloride removes significant liability and waste disposal costs associated with traditional methods. Furthermore, the reliance on commodity chemicals such as 3-nitro-4-methylaniline and 4-formylbenzoyl chloride ensures a stable supply chain, as these materials are produced by multiple global vendors, mitigating the risk of single-source bottlenecks. The high yields reported in the embodiments, particularly the 88.5% yield for the primary intermediate, directly correlate to lower raw material consumption per kilogram of final product, driving down the variable costs of production.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal catalysts and chiral ligands that were mandatory in prior art. By replacing column chromatography with simple crystallization and filtration, the manufacturing cycle time is drastically shortened, and solvent recovery becomes more efficient. This simplification of the downstream processing unit operations leads to substantial cost savings in both labor and utilities, making the final API intermediate much more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of temperatures and use stable organic solvents, ensures consistent batch-to-batch quality. This reliability is critical for maintaining continuous supply to downstream API manufacturers. The avoidance of pyrophoric reagents also simplifies logistics and storage requirements, allowing for safer transportation and handling of materials within the facility, thereby reducing the risk of production shutdowns due to safety incidents.
- Scalability and Environmental Compliance: From an environmental perspective, this route is superior as it avoids the generation of heavy metal waste streams that require specialized treatment. The aqueous workup procedures described allow for easier separation of organic and inorganic phases, facilitating solvent recycling. The scalability is evidenced by the successful execution of reactions on multi-gram scales in the patent examples without loss of efficiency, indicating a smooth path to multi-kilogram and ton-scale production suitable for meeting commercial demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the feasibility and advantages for potential partners.
Q: What are the safety advantages of this synthesis route compared to prior art?
A: Unlike previous methods utilizing pyrophoric trimethylaluminum or toxic tin chloride reductants, this patent employs standard organic bases and borohydride reductants, significantly enhancing operational safety and waste management profiles.
Q: How does this method improve purification efficiency?
A: The process eliminates the need for column chromatography, which was required in older methods with yields below 20%. Instead, the products are isolated via crystallization or simple filtration, drastically reducing solvent consumption and processing time.
Q: Can this route be adapted for different amine substituents?
A: Yes, the reductive amination step is highly versatile, allowing for the introduction of various cyclic and acyclic amines such as morpholine, piperidine, and methylpiperazine, facilitating the rapid generation of diverse compound libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-[3-Nitro-4-Methyl-Phenyl]-4-Aldehyde-Benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team has extensively analyzed the route described in CN101654416A and possesses the expertise to implement this chemistry with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of N-[3-nitro-4-methyl-phenyl]-4-aldehyde-benzamide meets the highest industry standards for impurity profiles and physical properties.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your overall budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from clinical trials to commercial launch with superior chemical solutions.
